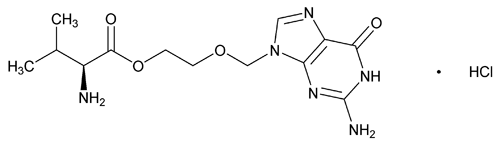
Valacyclovir Hydrochloride
C13H20N6O4·HCl 360.80
l-Valine, 2-[(2-amino-1,6-dihydro-6-oxo-9H-purin-9-yl)methoxy] ethyl ester, monohydrochloride;
l-Valine, ester with 9-[(2-hydroxyethoxy)methyl]guanine, monohydrochloride

 [124832-27-5].
[124832-27-5].
(val'' ay sye' kloe vir hye'' droe klor' ide).
C13H20N6O4·HCl 360.80
l-Valine, 2-[(2-amino-1,6-dihydro-6-oxo-9H-purin-9-yl)methoxy] ethyl ester, monohydrochloride;
l-Valine, ester with 9-[(2-hydroxyethoxy)methyl]guanine, monohydrochloride
DEFINITION
Valacyclovir Hydrochloride contains NLT 95.0% and NMT 102.0% of C13H20N6O4·HCl, calculated on the anhydrous and solvent-free basis.
IDENTIFICATION
• B.
The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
• C. Identification Tests—General, Chloride  191
191
Sample solution:
50 mg/mL in water
Acceptance criteria:
Meets the requirements
ASSAY
• Procedure
Mobile phase:
Methanol, water, and perchloric acid (1: 19: 0.1)
Standard solution:
0.5 mg/mL of USP Valacyclovir Hydrochloride RS in 0.05 M hydrochloric acid. [Note—USP Valacyclovir Hydrochloride RS contains a detectable quantity of d-valacyclovir. ]
Sample solution:
0.5 mg/mL of Valacyclovir Hydrochloride in 0.05 M hydrochloric acid
Chromatographic system
Mode:
LC
Detector:
UV 254 nm
Column:
4-mm × 15-cm; 5-µm packing L66
Column temperature:
10
Flow rate:
0.75 mL/min
Injection size:
10 µL
System suitability
Sample:
Standard solution
Suitability requirements
Resolution:
NLT 2.0 between valacyclovir hydrochloride and d-valacyclovir
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of valacyclovir hydrochloride (C13H20N6O4·HCl) in the portion of Valacyclovir Hydrochloride taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response of valacyclovir from the Sample solution |
| rS | = | = peak response of valacyclovir from the Standard solution |
| CS | = | = concentration of valacyclovir hydrochloride in the Standard solution (mg/mL) |
| CU | = | = concentration of Valacyclovir Hydrochloride in the Sample solution (mg/mL) |
Acceptance criteria:
95.0%–102.0% on the anhydrous and solvent-free basis
IMPURITIES
• Residue on Ignition  281
281 :
NMT 0.1% on a 2-g sample
:
NMT 0.1% on a 2-g sample
• Heavy Metals, Method II  231
231 :
NMT 20 ppm
:
NMT 20 ppm
• Limit of Palladium (if present)
(See Plasma Spectrochemistry  730
730 .)
.)
Diluent:
Dimethyl sulfoxide and hydrochloric acid (98:2)
Blank solution:
Diluent
Standard solutions:
Dilute with Diluent any commercially available standard stock solution of 1 mg/mL of palladium to prepare the following two solutions: 1 µg/mL of palladium and 10 µg/mL of palladium.
Sample solution:
10 mg/mL of Valacyclovir Hydrochloride in Diluent
Analytical wavelength:
340.458 nm
Spectrophotometer system:
Use a suitable standard inductively coupled plasma–optical emission spectrophotometric system, and construct a calibration curve.
System suitability
Samples:
Blank solution and Standard solutions
Suitability requirements
Relative standard deviation:
NMT 2.0%, Standard solutions
Correlation coefficient:
NLT 0.999, Blank solution and Standard solution
Analysis
Samples:
Blank solution and Sample solution
Calculate the concentration of palladium using the calibration curve corrected for the emission response of the Blank solution and sample weight. Calculate the amount of palladium in the Valacyclovir Hydrochloride taken to prepare the Sample solution.
Acceptance criteria:
NMT 10 ppm
• Organic Impurities, Procedure 1 (for related compounds E, F, and G)
Developing solvent:
Methylene chloride, methanol, tetrahydrofuran, and ammonia solution (54:34:12:3)
Standard stock solution:
Transfer 5 mg each of USP Valacyclovir Related Compound D RS, USP Valacyclovir Related Compound E RS, and USP Valacyclovir Related Compound G RS, and 8.4 mg of USP Valacyclovir Related Compound F RS into a 10-mL volumetric flask. Add 2 mL of water with swirling, followed by 6 mL of alcohol, and sonicate for 20 min. Allow to cool, and dilute with alcohol to volume.
Standard solutions:
Transfer 1.0 and 0.5 mL of Standard stock solution into two separate 10-mL volumetric flasks. Dilute the solution in both flasks with alcohol to volume.
Sample solution:
Transfer 250 mg of Valacyclovir Hydrochloride into a 5-mL volumetric flask. Add 2 mL of water, and sonicate for 20 min to dissolve. Add alcohol to about 95% volume of the flask. Cool, and dilute with alcohol to volume. Pass through a suitable filter of 0.45-µm pore size.
Chromatographic system
Mode:
TLC
Detector:
UV, long and short wavelength
Plate:
TLC plate coated with 0.25-mm layer of chromatographic silica gel mixture. Prewash the plate with methanol.
Developing distance:
NLT 7 cm from the origin
Application size:
4 µL
Analysis
Samples:
Standard solutions and Sample solution
Develop the plate to the specified distance. Remove the plate from the solvent chamber, and allow to dry. Examine the plate under short-wavelength UV light, and visually estimate the valacyclovir related compounds E and G in the sample using the appropriate standard spots. The chromatograms obtained with the Standard solutions each show three clearly separated spots due to valacyclovir related compounds D, E, and G. Spray the plate with 0.01% fluorescamine in ethylene dichloride, and examine the sprayed plate under long-wavelength UV to estimate the level of valacyclovir related compound F in the sample using the appropriate standard spot. The relative RF values and limits for each impurity are provided in Table 1.
Acceptance criteria:
See Table 1.
Table 1
| Name | Relative RF Value |
Acceptance Criteria, NMT (%) |
|---|---|---|
| Valacyclovir hydrochloride | 1 | — |
| Valacyclovir related compound Da | 1.1 | — |
| Valacyclovir related compound Eb | 1.3 | 0.2 |
| Valacyclovir related compound Fc | 1.8 | 0.1 |
| Valacyclovir related compound Gd | 1.9 | 0.05 |
|
a
This impurity is quantitated using Procedure 2.
b
2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy]ethyl N-[(benzyloxy)carbonyl]-l-valinate.
c
2-Hydroxyethyl-l-valinate.
d
N,N-Dimethylpyridin-4-amine.
|
||
• Organic Impurities, Procedure 2
Solution A:
0.3% w/w trifluoroacetic acid solution in water
Solution B:
0.3% w/w trifluoroacetic acid solution in methanol
Diluent:
Alcohol and water (1:4)
Mobile phase:
See Table 2.
Table 2
| Time (min) |
Solution A (%) |
Solution B (%) |
|---|---|---|
| 0 | 90 | 10 |
| 5 | 90 | 10 |
| 35 | 60 | 40 |
| 35.01 | 90 | 10 |
| 45 | 90 | 10 |
System suitability solution:
0.4 mg/mL of USP Valacyclovir Hydrochloride RS, 0.8 µg/mL of USP Valacyclovir Related Compound C RS, and 1.6 µg/mL of USP Acyclovir Related Compound A RS in Diluent
Sample solution:
0.4 mg/mL of Valacyclovir Hydrochloride in Diluent
Chromatographic system
Mode:
LC
Detector:
UV 254 nm
Column:
4.6-mm × 25-cm; 5-µm packing L11
Column temperature:
15
Flow rate:
0.8 mL/min
Injection size:
10 µL
System suitability
Sample:
System suitability solution
Resolution:
NLT 1.5 between valacyclovir and valacyclovir related compound C, and NLT 1.5 between valacyclovir related compound C and acyclovir related compound A
Tailing factor:
NMT 1.5 for the valacyclovir hydrochloride peak
Analysis
Sample:
Sample solution
Calculate the percentage of each individual impurity in the portion of Valacyclovir Hydrochloride taken:
Result = (rU/rT) × 100
| rU | = | = peak response of any impurity in the Sample solution |
| rT | = | = sum of all the peak responses from the Sample solution |
Acceptance criteria:
See Table 3.
Table 3
| Name | Relative Retention Time |
Acceptance Criteria, NMT (%) |
|---|---|---|
| Guanine (near solvent front)a,b | 0.31 | — |
| Acyclovira,c | 0.42 | — |
| Acyclovir alaninated | 0.54 | 0.2 |
| Valacyclovir | 1.00 | — |
| Valacyclovir related compound Ce | 1.06 | 0.3 |
| Acyclovir related compound Aa,f | 1.09 | — |
| Valacyclovir related compound Dg | 1.17 | 0.5 |
| Acyclovir isoleucinateh | 1.30 | 0.2 |
| N-Formyl valacycloviri | 1.61 | 0.8 |
| Guaninyl valacyclovirj | 1.66 | 0.2 |
| Bis valacyclovirk | 2.0 | 0.3 |
| Any unspecified impurity | — | 0.1 |
|
a
This impurity is quantitated by the Procedure 3 method.
b
2-Amino-1H-purin-6(9H)-one (guanine).
c
9-[(2-Hydroxyethoxy)methyl]guanine (acyclovir).
d
9-[(2-Hydroxyethoxy)methyl]guanine l-alaninate.
e
2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy]ethyl N-methyl-l-valinate.
f
2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy]ethyl acetate.
g
2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy]ethyl N-ethyl-l-valinate.
h
9-[(2-Hydroxyethoxy)methyl]guanine l-isoleucinate.
i
9-[(2-Hydroxyethoxy)methyl]guanine N-formyl-l-valinate.
j
[N2-(guanine-N2-yl)methyl]-9-[(2-hydroxyethoxy)methyl]guanine l-valinate.
k
2,2¢-[Methylenebis[imino(6-oxo-1,6-dihydro-9H-purine-9,2-diyl)methylene-oxy]]diethyl di(l-valinate).
|
||
• Organic Impurities, Procedure 3
Mobile phase, Standard solution, Sample solution, and Chromatographic system:
Proceed as directed in the Assay.
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of each individual impurity in the portion of Valacyclovir Hydrochloride taken:
Result = (rU/rS) × (CS/CU) × (1/F) × 100
| rU | = | = peak response of guanine plus acyclovir or acyclovir acetate or d-valacyclovir from the Sample solution |
| rS | = | = peak response of valacyclovir from the Standard solution |
| CS | = | = concentration of USP Valacyclovir Hydrochloride RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Valacyclovir Hydrochloride in the Sample solution (mg/mL) |
| F | = | = relative response factor (see Table 4) |
Acceptance criteria:
See Table 4.
Table 4
| Name | Relative Retention Time |
Relative Response Factor |
Acceptance Criteria, NMT (%) |
|
|---|---|---|---|---|
| Guanine and acyclovira,b | 0.18 | 1.51 | 2.0 | |
| Acyclovir related compound Ac | 0.42 | 1.12 | 0.2 | |
| d-Valacyclovird | 0.55 | 1.0 | 3.0 | |
| Valacyclovir | 1.0 | — | — | |
|
a
2-Amino-1H-purin-6(9H)-one (guanine).
b
9-[(2-Hydroxyethoxy)methyl]guanine (acyclovir).
c
2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy]ethyl acetate.
d
d-Valine, 2-[(2-amino-1,6-dihydro-6-oxo-9H-purin-9-yl)methoxy] ethyl
ester, monohydrochloride. |
||||
Total organic impurities:
NMT 5.0% for the sum of all impurities from Organic Impurities, Procedures 1, 2, and 3
SPECIFIC TESTS
• Water Determination, Method I  921
921 :
For the anhydrous form: NMT 2.0% (200 mg of sample); if labeled as the hydrous form: 5.0%–11.0%
:
For the anhydrous form: NMT 2.0% (200 mg of sample); if labeled as the hydrous form: 5.0%–11.0%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers, and store at a temperature below 30 .
.
• Labeling:
Where it is the hydrous form, the label so indicates.
• USP Reference Standards  11
11
USP Acyclovir Related Compound A RS 
2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy]ethyl acetate.
C10H13N5O4 267.24

[Note—USP Acyclovir Related Compound A AS is equivalent. ]
2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy]ethyl acetate.
C10H13N5O4 267.24
USP Valacyclovir Related Compound C RS 
2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy]ethyl N-methyl-l-valinate hydrochloride.
C14H22N6O4·HCl 374.82

2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy]ethyl N-methyl-l-valinate hydrochloride.
C14H22N6O4·HCl 374.82
USP Valacyclovir Related Compound D RS 
2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9yl)methoxy]ethyl N-ethyl-l-valinate.
C15H24N6O4 352.39

2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9yl)methoxy]ethyl N-ethyl-l-valinate.
C15H24N6O4 352.39
USP Valacyclovir Related Compound E RS 
2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy]ethyl N-benzyloxy)carbonyl]-l-valinate.
C21H26N6O6 458.47

2-[(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy]ethyl N-benzyloxy)carbonyl]-l-valinate.
C21H26N6O6 458.47
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Leonel M. Santos, Ph.D.
Senior Scientific Liaison 1-301-816-8168 |
(SM12010) Monographs - Small Molecules 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4983
Pharmacopeial Forum: Volume No. 35(3) Page 589