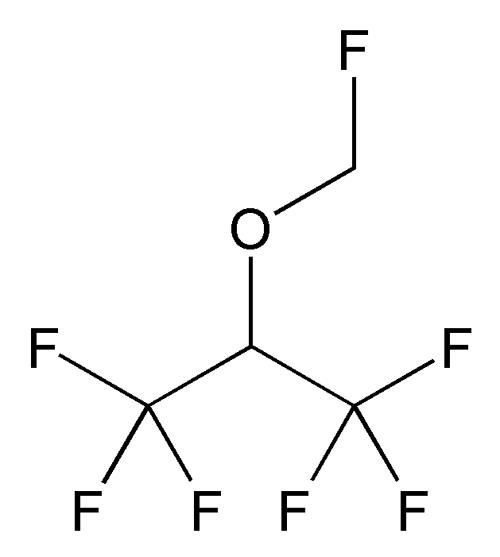
Sevoflurane
C4H3F7O 200.05
Propane, 1,1,1,3,3,3-hexafluoro-2-(fluoromethoxy)-;
Fluoromethyl 2,2,2-trifluoro-1-(trifluoromethyl)ethyl ether

 [28523-86-6].
[28523-86-6].
(see'' voe floo' rane).
C4H3F7O 200.05
Propane, 1,1,1,3,3,3-hexafluoro-2-(fluoromethoxy)-;
Fluoromethyl 2,2,2-trifluoro-1-(trifluoromethyl)ethyl ether
DEFINITION
Sevoflurane contains NLT 99.97% and NMT 100.00% of C4H3F7O.
IDENTIFICATION
• Infrared Absorption:
The IR absorption spectrum of Sevoflurane, obtained using a gas cell, exhibits maxima only at the same wavelengths as that of a similar preparation of USP Sevoflurane RS.
ASSAY
• Procedure
Analysis:
Using the results from Organic Impurities, Procedure 2, calculate the percentage of C4H3F7O in the volume of Sevoflurane taken by subtracting the sum of percentages for all impurities found from 100.00%.
Acceptance criteria:
99.97%–100.00% of C4H3F7O
IMPURITIES
Inorganic Impurities
• Limit of Fluoride
[Note—Use plastic utensils throughout this test. ]
Buffer solution:
Transfer 110 g of sodium chloride and 1 g of sodium citrate to a 2000-mL volumetric flask. Dissolve in 700 mL of water. Carefully add 150 g of sodium hydroxide, and shake to dissolve. Cool to room temperature, and carefully add 450 mL of glacial acetic acid while stirring. Cool, add 600 mL of isopropyl alcohol, and dilute with water to volume. [Note—The pH of this solution is 5.0–5.5. This solution may be used for 6 weeks when stored at room temperature. ]
Solution A:
Transfer 221 mg of sodium fluoride, previously dried at 150 for 4 h, to a 100-mL volumetric flask. Add about 20 mL of water, and mix to dissolve. Add 1.0 mL of 0.01 N sodium hydroxide, and dilute with water to volume. Each mL of this solution contains 1 mg of fluoride. Store in a tightly closed, plastic container. [Note—This solution may be used for 2 weeks when stored in a refrigerator. ]
for 4 h, to a 100-mL volumetric flask. Add about 20 mL of water, and mix to dissolve. Add 1.0 mL of 0.01 N sodium hydroxide, and dilute with water to volume. Each mL of this solution contains 1 mg of fluoride. Store in a tightly closed, plastic container. [Note—This solution may be used for 2 weeks when stored in a refrigerator. ]
System suitability solution 1:
0.2 µg/mL of fluoride from Solution A in water
System suitability solution 2:
0.5 µg/mL of fluoride from Solution A in water
System suitability solution 3:
2 µg/mL of fluoride from Solution A in water
System suitability solution 4:
5 µg/mL of fluoride from Solution A in water
Standard solution 1:
0.10 µg/mL of fluoride from System suitability solution 1 in Buffer solution
Standard solution 2:
0.25 µg/mL of fluoride from System suitability solution 2 in Buffer solution
Standard solution 3:
1.0 µg/mL of fluoride from System suitability solution 3 in Buffer solution
Standard solution 4:
2.5 µg/mL of fluoride from System suitability solution 4 in Buffer solution
Sample solution:
Pipet 50.0 mL of Sevoflurane and 50.0 mL of water into a separatory funnel, shake vigorously for 3 min, and allow the liquids to separate completely. Transfer 25.0 mL of the aqueous top layer to a 50-mL volumetric flask, and dilute with Buffer solution to volume.
Analysis:
Concomitantly measure the potentials, in mV, of System suitability solutions 1–4, Standard solutions 1–4, and the Sample solution with a pH meter (see pH  791
791 ) capable of a minimum reproducibility of ±0.2 mV and equipped with a fluoride-specific ion-indicating electrode and a glass-sleeved calomel reference electrode. [Note—When taking measurements, transfer the solution under test to a 100-mL beaker containing a polytef-coated stirring bar, and immerse the electrodes. Allow to stir on a magnetic stirrer having an insulated top until equilibrium is attained in about 2–3 min, and record the potential. Rinse the electrodes with the Buffer solution, and dry, taking care to avoid damaging the crystal of the specific-ion electrode. A satisfactory response is achieved if the difference between the potentials obtained with System suitability solution 4 and System suitability solution 2 is in the range between 50 and 60 mV. ] Plot the logarithms of the fluoride concentrations, in µg/mL, of Standard solutions 1–4 versus potentials, in mV. From the graph and the measured potential of the Sample solution, determine the concentration (µg/mL) of fluoride in the Sample solution.
) capable of a minimum reproducibility of ±0.2 mV and equipped with a fluoride-specific ion-indicating electrode and a glass-sleeved calomel reference electrode. [Note—When taking measurements, transfer the solution under test to a 100-mL beaker containing a polytef-coated stirring bar, and immerse the electrodes. Allow to stir on a magnetic stirrer having an insulated top until equilibrium is attained in about 2–3 min, and record the potential. Rinse the electrodes with the Buffer solution, and dry, taking care to avoid damaging the crystal of the specific-ion electrode. A satisfactory response is achieved if the difference between the potentials obtained with System suitability solution 4 and System suitability solution 2 is in the range between 50 and 60 mV. ] Plot the logarithms of the fluoride concentrations, in µg/mL, of Standard solutions 1–4 versus potentials, in mV. From the graph and the measured potential of the Sample solution, determine the concentration (µg/mL) of fluoride in the Sample solution.
Acceptance criteria:
NMT 2 µg/mL is found.
Organic Impurities
• Procedure 1: Limit of Nonvolatile Residue
Analysis:
Transfer 10.0 mL of Sevoflurane to an evaporating dish, evaporate to dryness on a steam bath, and dry the residue at 105 for 2 h.
for 2 h.
Acceptance criteria:
The weight of the residue does not exceed 1.0 mg.
• Procedure 2
Internal standard solution:
Use dimethoxymethane.
Ethylene dichloride identification solution:
Transfer 2.0 mL of Sevoflurane to a vial, and seal with a cap and septum. Using a microsyringe, add 20 µL of ethylene dichloride through the septum of the vial, and mix thoroughly.
Sevoflurane related compounds stock solution:
Transfer 20 mL of Sevoflurane to a 40-mL vial with a septum lid. Add 20 µL each of USP Sevoflurane Related Compound A RS, USP Sevoflurane Related Compound B RS, and USP Sevoflurane Related Compound C RS to the vial, and mix thoroughly.
Related compounds identification solution:
Transfer 1.0 mL of Ethylene dichloride identification solution to a 10-mL volumetric flask, and dilute with Sevoflurane to volume. Transfer 2 mL of this solution and 5 mL of Sevoflurane related compounds stock solution to a 50-mL volumetric flask, dilute with Sevoflurane to volume, and mix thoroughly.
Standard solutions:
Prepare in duplicate, proceeding for each as follows. Transfer 2.0 mL of ethylene dichloride to a screw-capped vial, immediately seal with a cap and septum, and place on a balance. Using a microsyringe, transfer about 20 µL of USP Sevoflurane RS to the vial by inserting the syringe needle through the septum. Record the quantity, in mg, of USP Sevoflurane RS added. Using the same method, transfer about 20 µL of Internal standard solution to the vial, and record the quantity, in mg, of the solution added.
Control standard solution:
Place a 40-mL vial with a septum lid on an analytical balance, and tare out the weight. Add 30 mL of ethylene dichloride to the vial, and seal tightly. Record the weight of the ethylene dichloride, and tare. Using a microsyringe, add 20 µL of the USP Sevoflurane RS through the septum of the vial, record the weight, and mix thoroughly. Transfer 1.0 mL of this solution to a 100-mL volumetric flask, and dilute with ethylene dichloride to volume.
Sample solution:
Transfer 20.0 mL of Sevoflurane to a vial, and insert the stopper. Using a microsyringe, add 5 µL of Internal standard solution to the vial.
Chromatographic system
Mode:
GC
Detector:
Flame ionization
Column:
0.32-mm × 30-m fused-silica capillary column coated with a 3.0-µm film of liquid phase G43
Temperature:
See the temperature program table. [Note—Before use, condition the column overnight at a temperature of 250 . ]
. ]
| Initial Temperature ( |
Temperature Ramp ( |
Final Temperature ( |
Hold Time at Final Temperature (min) |
|---|---|---|---|
| 40 | 0 | 40 | 10 |
| 40 | 10 | 200 | 14 |
Injector port:
200
Detector:
225
Carrier gas:
Helium
Flow rate:
1 mL/min. [Note—The make-up gas flow rate is 20 mL/min. ]
Injection size:
2 µL
Injection type:
Split ratio 1:20
System suitability
Samples:
Related compounds identification solution and Standard solutions
Suitability requirements
[Note—Identify the peaks using the relative retention times given in Impurity Table 1. ]
Resolution:
NLT 2.0 between sevoflurane related compound C and ethylene dichloride, Related compounds identification solution
Column efficiency:
NLT 6000 theoretical plates, Standard solutions
Relative standard deviation:
NMT 3.0% from the peak area ratio of sevoflurane to the internal standard, Standard solutions.
Analysis
Samples:
Ethylene dichloride identification solution, Control standard solution, Standard solutions, and Sample solution
Calculate the response factor for each of the Standard solutions:
Result = (WI/WS) × RS
| WI | = | = weight of the internal standard in the Standard solutions (mg) |
| WS | = | = weight of USP Sevoflurane RS in the Standard solutions (mg) |
| RS | = | = peak area response ratio of sevoflurane to that of the internal standard from the Standard solutions |
The response factors for the duplicate Standard solutions do not differ by more than 3.0% from their average.
Calculate the quantity, in µg/g, of each impurity in the portion of C4H3F7O taken:
Result = (S1/S2) × (RI/FR) × (1/F) × 250
| S1 | = | = specific gravity of the internal standard, 0.859 |
| S2 | = | = specific gravity of sevoflurane, 1.525 |
| RI | = | = peak area response ratio of the impurity to that of the internal standard from the Sample solution |
| FR | = | = average response factor obtained as directed above |
| F | = | = respective relative response factor for the impurities (see Impurity Table 1) |
Acceptance criteria
[Note—Do not include sevoflurane, the internal standard, or any peak identified as solvent carryover (ethylene dichloride). Also, disregard any peak with an area less than 30% of the average area of the principal peak in the chromatogram obtained with the Control standard solution. ]
Individual impurities:
NMT 25 µg/g of sevoflurane related compound A and NMT 100 µg/g of any other single impurity
Total impurities:
NMT 300 µg/g
Impurity Table 1
| Name | Relative Retention Time |
Relative Response Factor |
|---|---|---|
| Sevoflurane related compound A | 0.78 | 1.0 |
| Sevoflurane related compound B | 0.83 | 1.0 |
| Sevoflurane | 1.0 | — |
| Internal standard (dimethoxymethane) | 1.35 | — |
| Ethylene dichloride | 2.28 | — |
| Sevoflurane related compound C | 2.31 | 0.46 |
| Unknown impurities | — | 1 |
SPECIFIC TESTS
• Refractive Index  831
831 :
1.2745–1.2760 at 20
:
1.2745–1.2760 at 20
• Acidity or Alkalinity:
Transfer 20.0 mL of Sevoflurane and 20.0 mL of carbon dioxide-free water to a separatory funnel, shake for 3 min, and allow the layers to separate.
Acceptance criteria:
The aqueous layer requires NMT 0.10 mL of 0.010 N sodium hydroxide or NMT 0.60 mL of 0.010 N hydrochloric acid for neutralization, bromocresol purple TS being used as the indicator.
• Water Determination, Method I  921
921 :
NMT 0.1%
:
NMT 0.1%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight, light-resistant containers. Store at controlled room temperature. Replace the cap securely after each use.
• USP Reference Standards  11
11
USP Sevoflurane Related Compound A RS
1,1,1,3,3-Pentafluoroisopropenyl fluoromethyl ether.
C4H2F6O 179.97
1,1,1,3,3-Pentafluoroisopropenyl fluoromethyl ether.
C4H2F6O 179.97
USP Sevoflurane Related Compound B RS
1,1,1,3,3,3-Hexafluoro-2-methoxy-propane.
1,1,1,3,3,3-Hexafluoro-2-methoxy-propane.
USP Sevoflurane Related Compound C RS
1,1,1,3,3,3-Hexafluoro-2-propanol.
1,1,1,3,3,3-Hexafluoro-2-propanol.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Mary S. Waddell
Scientific Liaison 1-301-816-8124 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4632
Pharmacopeial Forum: Volume No. 36(1) Page 139