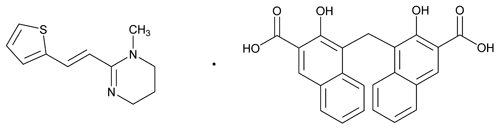
Pyrantel Pamoate
C11H14N2S·C23H16O6 594.68
Pyrimidine, 1,4,5,6-tetrahydro-1-methyl-2-[2-(2-thienyl)ethenyl]-, (E)-, compd. with 4,4'-methylenebis[3-hydroxy-2-naphthalenecarboxylic acid] (1:1);
(E)-1,4,5,6-Tetrahydro-1-methyl-2-[2-(2-thienyl)vinyl]pyrimidine 4,4'-methylenebis[3-hydroxy-2-naphthoate] (1:1)

 [22204-24-6].
[22204-24-6].
(pi ran' tel pam' oh ate).
C11H14N2S·C23H16O6 594.68
Pyrimidine, 1,4,5,6-tetrahydro-1-methyl-2-[2-(2-thienyl)ethenyl]-, (E)-, compd. with 4,4'-methylenebis[3-hydroxy-2-naphthalenecarboxylic acid] (1:1);
(E)-1,4,5,6-Tetrahydro-1-methyl-2-[2-(2-thienyl)vinyl]pyrimidine 4,4'-methylenebis[3-hydroxy-2-naphthoate] (1:1)
DEFINITION
Pyrantel Pamoate contains NLT 97.0% and NMT 103.0% of C34H30N2O6S, calculated on the dried basis.
IDENTIFICATION
• B. Ultraviolet Absorption  197U
197U
Solution:
16 µg/mL in methanol
• C.
The retention time of the major peaks due to pyrantel base and pamoic acid of the Sample solution correspond to those of the Standard solution, as obtained in the Assay.
ASSAY
• Procedure
[Note—Use low-actinic glassware in preparing solutions of pyrantel pamoate, or otherwise protect the solutions from unnecessary exposure to bright light. Complete the Assay without prolonged interruption. ]
Mobile phase:
Acetonitrile, acetic acid, diethylamine, and water (92.8:3:1.2:3)
[Note—Increasing the amount of acetonitrile in the Mobile phase increases retention times. Increasing the amount of acetic acid, water, and diethylamine decreases retention times. Should the Mobile phase need to be adjusted, maintain the ratios among acetic acid, diethylamine, and water (1:0.4:1). ]
Standard solution:
80 µg/mL of USP Pyrantel Pamoate RS in Mobile phase
Sample solution:
80 µg/mL of Pyrantel Pamoate in Mobile phase
Chromatographic system
Mode:
LC
Detector:
UV 288 nm
Column:
4.6-mm × 25-cm; packing L3
Flow rate:
1 mL/min
Injection size:
20 µL
System suitability
Sample:
Standard solution
[Note—The relative retention times for pamoic acid and pyrantel are 0.6 and 1.0, respectively. ]
Suitability requirements
Resolution:
NLT 10.0 between pyrantel and pamoic acid
Column efficiency:
NLT 8000 theoretical plates
Tailing factor:
NMT 1.3 for the pyrantel peak
Relative standard deviation:
NMT 1.0% for the pyrantel peak
Analysis
Samples:
Standard solution and Sample solution
Record the chromatograms obtained for a period of NLT 2.5 times the retention times of pyrantel, and measure the responses for the major peaks.
Calculate the percentage of C34H30N2O6S in the portion of Pyrantel Pamoate taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response for pyrantel from the Sample solution |
| rS | = | = peak response for pyrantel from the Standard solution |
| CS | = | = concentration of USP Pyrantel Pamoate RS in the Standard solution (µg/mL) |
| CU | = | = concentration of the Sample solution (µg/mL) |
Acceptance criteria:
97.0%–103.0% on the dried basis
OTHER COMPONENTS
• Content of Pamoic Acid
Mobile phase:
Acetonitrile, acetic acid, diethylamine, and water (92.8:3:1.2:3)
[Note—Increasing the amount of acetonitrile in the Mobile phase increases retention times. Increasing the amount of acetic acid, water, and diethylamine decreases retention times. Should the Mobile phase need to be adjusted, maintain the ratios among acetic acid, diethylamine, and water (1:0.4:1). ]
Standard solution:
52 µg/mL of USP Pamoic Acid RS in Mobile phase
Sample solution:
80 µg/mL of Pyrantel Pamoate in Mobile phase
Chromatographic system
Mode:
LC
Detector:
UV 288 nm
Column:
4.6-mm × 25-cm; packing L3
Flow rate:
1 mL/min
Injection size:
20 µL
System suitability
Sample:
Standard solution
Suitability requirements
Resolution:
NLT 10.0 between pyrantel and pamoic acid
Column efficiency:
NLT 8000 theoretical plates
Tailing factor:
NMT 1.3 for the pyrantel peak
Relative standard deviation:
NMT 1.0% for the pyrantel peak
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C23H16O6 in the portion of Pyrantel Pamoate taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response for pamoic acid from the Sample solution |
| rS | = | = peak response for pamoic acid from the Standard solution |
| CS | = | = concentration of USP Pamoic Acid RS in the Standard solution (µg/mL) |
| CU | = | = concentration of the Sample solution (µg/mL) |
Acceptance criteria:
63.4%–67.3% of pamoic acid, calculated on the dried basis
IMPURITIES
Inorganic Impurities
• Limit of Iron  241
241
Analysis:
To the residue obtained in the test for Residue on Ignition, add hydrochloric acid and nitric acid (3:2), and evaporate on a steam bath to dryness. Dissolve the residue in 2 mL of hydrochloric acid with the aid of gentle heat. Add 18 mL of hydrochloric acid, dilute with water to 50 mL, and mix. Dilute 5 mL of this solution with water to 47 mL.
Acceptance criteria:
The limit is 0.0075%.
• Heavy Metals, Method II  231
231 :
50 ppm
:
50 ppm
Organic Impurities
• Procedure
Diluent:
Glacial acetic acid, diethylamine, and water (5:2:5)
Mobile phase:
Acetonitrile and Diluent (92.8:7.2). [Note—Prepare the solutions immediately before use, and protect from light at all stages. ]
Stock standard solution:
0.8 mg/mL of USP Pyrantel Pamoate RS. Dissolve in Diluent, using 7% of the final volume, and diluting with acetonitrile to volume.
Standard solution:
4 µg/mL of USP Pyrantel Pamoate RS in Mobile phase from the Stock standard solution
System suitability solution:
Dissolve a quantity of USP Pyrantel Related Compound A RS first in the Stock standard solution, using 5% of the final volume. Dilute with Diluent to volume to obtain a solution with a concentration of 0.2 mg/mL of pyrantel related compound A and 40 µg/mL of pyrantel pamoate. Transfer a suitable portion of this solution, and dilute quantitatively with Diluent to obtain a solution with a final concentration of about 4 µg/mL of pyrantel related compound A and 0.8 µg/mL of pyrantel pamoate.
Sample solution:
0.8 mg/mL of pyrantel pamoate prepared by dissolving in Diluent, using 7% of the final volume, and diluting with acetonitrile to volume
Chromatographic system
Mode:
LC
Detector:
UV 288 nm
Column:
4.6-mm × 25-cm; packing L3
Flow rate:
1.0 mL/min
Injection size:
20 µL
System suitability
Sample:
System suitability solution
Suitability requirements
Resolution:
NLT 4.0 between pyrantel and pyrantel related compound A
Tailing factor:
NMT 1.5 for the pyrantel peak
Relative standard deviation:
NMT 10.0% for the pyrantel peak
Analysis
Samples:
Standard solution, System suitability solution, and Sample solution
[Note—Record the chromatograms for about 4 times the retention time of pyrantel. Disregard any peak in the Sample solution having an area less than one-tenth the principal peak in the Standard solution. ]
Calculate the percentage of pyrantel related compound A in the portion of Pyrantel Pamoate taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response for pyrantel related compound A from the Sample solution |
| rS | = | = peak response for pyrantel related compound A from the System suitability solution |
| CS | = | = concentration of USP Pyrantel Related Compound A RS in the System suitability solution (µg/mL) |
| CU | = | = concentration of pyrantel pamoate in the Sample solution (µg/mL) |
Calculate the percentage of Impurity B or any other unspecified impurity in the portion of Pyrantel Pamoate taken:
Result = (rU/rS) × (CS/CU) × (1/F) × 100
| rU | = | = peak response for impurity B or any individual unspecified impurity from the Sample solution |
| rS | = | = peak response for pyrantel from the Standard solution |
| CS | = | = concentration of USP Pyrantel Pamoate RS in the Standard solution (µg/mL) |
| CU | = | = concentration of pyrantel pamoate in the Sample solution (µg/mL) |
| F | = | = relative response factor (see Impurity Table 1) |
Acceptance criteria
Individual impurities:
See Impurity Table 1.
Total impurities:
NMT 1.0%
Impurity Table 1
| Name | Relative Retention Time |
Relative Response Factor |
Acceptance Criteria, NMT (%) |
|---|---|---|---|
| Pamoic acida | 0.5 | — | — |
| Pyrantel(E-isomer) | 1.0 | — | — |
| Pyrantel related compound Ab |
1.3 | — | 0.5 |
| Impurity Bc | 1.8 | 2.5 | 0.2 |
| Individual unspecified impurities |
— | 1.0 | 0.1 |
| Total unspecified impurities |
— | — | 0.3 |
|
a
For identification purposes only.
b
1-Methyl-2-[(Z)-2-(thiophen-2-yl)ethenyl]-1,4,5,6-tetrahydropyrimidine.
c
(E)-N-[3-(Methylamino)propyl]-3-(thiophen-2-yl)prop-2-enamide.
|
|||
SPECIFIC TESTS
• Loss on Drying  731
731 :
Dry a sample in a vacuum at 60
:
Dry a sample in a vacuum at 60 for 3 h: it loses NMT 2.0% of its weight.
for 3 h: it loses NMT 2.0% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in well-closed, light-resistant containers.
• USP Reference Standards  11
11
USP Pamoic Acid RS
C23H16O6 388.38
C23H16O6 388.38
USP Pyrantel Related Compound A RS
(Z)-1-Methyl-2-(2-(thiophen-2-yl)vinyl)-1,4,5,6-tetrahydropyrimidine.
C34H30N2O6S 594.69
(Z)-1-Methyl-2-(2-(thiophen-2-yl)vinyl)-1,4,5,6-tetrahydropyrimidine.
C34H30N2O6S 594.69
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Leonel M. Santos, Ph.D.
Senior Scientific Liaison 1-301-816-8168 |
(SM12010) Monographs - Small Molecules 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4485
Pharmacopeial Forum: Volume No. 34(6) Page 1482