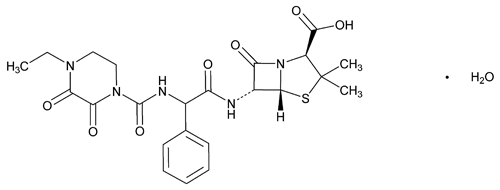
Piperacillin
C23H27N5O7S·H2O 535.57
C23H27N5O7S 517.56
4-Thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, 6-[[[[(4-ethyl-2,3-dioxo-1-piperazinyl)carbonyl]amino]phenylacetyl]amino]-3,3-dimethyl-7-oxo-, monohydrate, [2S-2 ,5
,5 ,6
,6 (S*)]];
(S*)]];
(2S,5R,6R)-6-[(R)-2-(4-Ethyl-2,3-dioxo-1-piperazinecarboxamido)-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid monohydrate

 [66258-76-2].
[66258-76-2].
Anhydrous

 [61477-96-1].
[61477-96-1].
(pi'' per a sil' in).
C23H27N5O7S·H2O 535.57
C23H27N5O7S 517.56
4-Thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, 6-[[[[(4-ethyl-2,3-dioxo-1-piperazinyl)carbonyl]amino]phenylacetyl]amino]-3,3-dimethyl-7-oxo-, monohydrate, [2S-2
(2S,5R,6R)-6-[(R)-2-(4-Ethyl-2,3-dioxo-1-piperazinecarboxamido)-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid monohydrate
Anhydrous
DEFINITION
Piperacillin contains NLT 960 µg/mg and NMT 1030 µg/mg of piperacillin (C23H27N5O7S), calculated on the anhydrous basis.
IDENTIFICATION
ASSAY
• Procedure
Mobile phase:
Methanol, water, 0.2 M monobasic sodium phosphate, and 0.4 M tetrabutylammonium hydroxide (450:447:100:3). Adjust with phosphoric acid to a pH of 5.50 ± 0.02.
System suitability solution:
0.1 mg/mL of USP Ampicillin RS and 0.2 mg/mL of USP Piperacillin RS in Mobile phase
Standard solution:
0.4 mg/mL of USP Piperacillin RS in Mobile phase. Dissolve in a few drops of methanol, and dilute with Mobile phase to volume. Use this solution within 1 h.
Sample solution:
0.4 mg/mL of Piperacillin in Mobile phase. Dissolve in a few drops of methanol, and dilute with Mobile phase to volume. Use this solution within 1 h.
Chromatographic system
Mode:
LC
Detector:
UV 220 nm
Column:
4.6-mm × 25-cm; packing L1
Flow rate:
1 mL/min
Injection size:
10 µL
System suitability
Samples:
System suitability solution and Standard solution
[Note—See Table 1 for relative retention times. ]
Suitability requirements
Resolution:
NLT 16 between ampicillin and piperacillin, System suitability solution
Tailing factor:
NMT 1.2 for the piperacillin peak, System suitability solution
Relative standard deviation:
NMT 2%, Standard solution
Analysis
Samples:
Standard solution and Sample solution
Calculate the potency of piperacillin (C23H27N5O7S) in the portion of Piperacillin taken:
Result = (rU/rS) × (CS/CU) × P
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Piperacillin RS in the Standard solution (mg/mL) |
| CU | = | = concentration of the Sample solution (mg/mL) |
| P | = | = potency of piperacillin in USP Piperacillin RS (µg/mg) |
Acceptance criteria:
960–1030 µg/mg on the anhydrous basis
IMPURITIES
• Heavy Metals, Method II  231
231 :
NMT 20 ppm
:
NMT 20 ppm
• Ampicillin and Piperacillin Related Compounds A, B, and C
Mobile phase, Sample solution, Chromatographic system, and System suitability:
Proceed as directed in the Assay.
Standard solution 1:
0.08 mg/mL of USP Ampicillin RS in Mobile phase
Standard solution 2:
0.04 mg/mL of USP Piperacillin RS in Mobile phase. Dissolve in a few drops of methanol, and dilute with Mobile phase to volume. Use this solution within 1 h.
Analysis
Samples:
Standard solution 1, Standard solution 2, and Sample solution
Calculate the percentage of ampicillin in the portion of Piperacillin taken:
Result = (rU/rS) × (CS/CU) × P × F × 100
| rU | = | = peak response of ampicillin from the Sample solution |
| rS | = | = peak response of ampicillin from Standard solution 1 |
| CS | = | = concentration of USP Ampicillin RS in Standard solution 1 (mg/mL) |
| CU | = | = concentration of the Sample solution (mg/mL) |
| P | = | = potency of ampicillin in USP Ampicillin RS (µg/mg) |
| F | = | = conversion factor, 0.001 mg/µg |
Calculate the percentages of piperacillin related compounds A, B, and C in the portion of Piperacillin taken:
Result = (rU/rS) × (CS/CU) × P × F1 × F2 × 100
| rU | = | = response of piperacillin related compound A, B, or C from the Sample solution |
| rS | = | = peak response of piperacillin from Standard solution 2 |
| CS | = | = concentration of USP Piperacillin RS in Standard solution 2 (mg/mL) |
| CU | = | = concentration of the Sample solution (mg/mL) |
| P | = | = potency of piperacillin in USP Piperacillin RS (µg/mg) |
| F1 | = | = relative response factor (see Table 1) |
| F2 | = | = conversion factor, 0.001 mg/µg |
Acceptance criteria:
See Table 1.
Table 1
| Name | Relative Retention Time |
Relative Response Factor |
Acceptance Criteria, NMT (%) |
|---|---|---|---|
| Piperacillin related compound Ba | 0.24 | 0.41 | 0.2 |
| Ampicillin | 0.31 | 1.0 | 0.2 |
| Piperacillin related compound Cb | 0.37 | 0.93 | 0.4 |
| Piperacillin related compound Ac | 0.62 | 1.4 | 1.0 |
| Piperacillin | 1.0 | — | — |
|
a
1-Ethyl-2,3-piperazinedione.
b
2-(3-Acetyl-4-carboxy-5,5-dimethyl-2-thiazolidinyl)-N-[N-[(4-ethyl-2,3-dioxo-1-piperazinyl)-carbonyl]-2-phenylglycyl]glycine.
c
4-Carboxy-
|
|||
• Piperacillin Related Compound D
Mobile phase:
Methanol, water, 0.2 M monobasic sodium phosphate, and 0.4 M tetrabutylammonium hydroxide (615:282:100:3). Adjust with phosphoric acid to a pH of 5.50 ± 0.02.
Standard solution:
0.04 mg/mL of USP Piperacillin RS in Mobile phase. Dissolve in a few drops of methanol, and dilute with Mobile phase to volume. Use this solution within 1 h.
Sample solution:
0.4 mg/mL of Piperacillin in Mobile phase. Dissolve in a few drops of methanol, and dilute with Mobile phase to volume. Use this solution within 1 h.
Chromatographic system
Mode:
LC
Detector:
UV 220 nm
Column:
4.6-mm × 25-cm; packing L1
Flow rate:
1 mL/min
Injection size:
10 µL
System suitability
Suitability requirements
Relative standard deviation:
NMT 2%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of piperacillin related compound D in the portion of Piperacillin taken:
Result = (rU/rS) × (CS/CU) × P × F1 × F2 × 100
| rU | = | = peak response of piperacillin related compound D from the Sample solution |
| rS | = | = peak response of piperacillin from the Standard solution |
| CS | = | = concentration of USP Piperacillin RS in the Standard solution (mg/mL) |
| CU | = | = concentration of the Sample solution (mg/mL) |
| P | = | = potency of piperacillin in USP Piperacillin RS (µg/mg) |
| F1 | = | = relative response factor (see Table 2) |
| F2 | = | = conversion factor, 0.001 mg/µg |
Acceptance criteria
See Table 2.
Table 2
| Name | Relative Retention Time |
Relative Response Factor |
Acceptance Criteria, NMT (%) |
|---|---|---|---|
| Piperacillin | 1.0 | — | — |
| Piperacillin related compound Da | 2.55 | 1.47 | 2.0 |
| Total impuritiesb | — | — | 3.8 |
|
a
6-[2-[6-[2-(4-Ethyl-2,3-dioxo-1-piperazinecarboxamido)-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxamido]-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid.
b
Total impurities is the sum of all impurities reported in the tests for Ampicillin and Piperacillin Related Compounds A, B, and C and Piperacillin Related Compound D.
|
|||
SPECIFIC TESTS
• Water Determination, Method I  921
921 :
2.0%–4.0%
:
2.0%–4.0%
• Optical Rotation, Specific Rotation  781S
781S
Sample solution:
40 mg/mL, in methanol
Acceptance criteria:
+155 to +175
to +175
• Bacterial Endotoxins Test  85
85 :
Where the label states that Piperacillin is sterile or that it must be subjected to further processing during the preparation of injectable dosage forms, it contains NMT 0.07 USP Endotoxin Unit/mg of piperacillin.
:
Where the label states that Piperacillin is sterile or that it must be subjected to further processing during the preparation of injectable dosage forms, it contains NMT 0.07 USP Endotoxin Unit/mg of piperacillin.
• Sterility Tests  71
71 :
Where the label states that Piperacillin is sterile or that it must be subjected to further processing during the preparation of injectable dosage forms, it meets the requirements when tested as directed under Test for Sterility of the Product to Be Examined, Membrane Filtration.
:
Where the label states that Piperacillin is sterile or that it must be subjected to further processing during the preparation of injectable dosage forms, it meets the requirements when tested as directed under Test for Sterility of the Product to Be Examined, Membrane Filtration.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in well-closed containers.
• Labeling:
Where it is intended for use in preparing injectable dosage forms, the label states that it is sterile or must be subjected to further processing during the preparation of injectable dosage forms.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ahalya Wise, M.S.
Senior Scientific Liaison 1-301-816-8161 |
(SM12010) Monographs - Small Molecules 1 |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4332
Pharmacopeial Forum: Volume No. 28(4) Page 1182