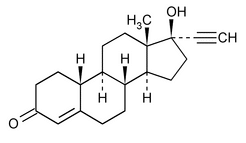
Norethindrone
(nor eth' in drone).
19-Norpregn-4-en-20-yn-3-one, 17-hydroxy-, (17
17-Hydroxy-19-nor-17
» Norethindrone contains not less than 97.0 percent and not more than 102.0 percent of C20H26O2, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
Completeness of solution—
The solution called for in the test for Specific rotation is clear and free from undissolved solid.
Melting range  741
741 :
between 202
:
between 202 and 208
and 208 .
.
Loss on drying  731
731 —
Dry it in vacuum at 105
—
Dry it in vacuum at 105 for 3 hours: it loses not more than 0.5% of its weight.
for 3 hours: it loses not more than 0.5% of its weight.
Limit of ethynyl group—
Dissolve 200 mg in about 40 mL of tetrahydrofuran. Add 10 mL of silver nitrate solution (1 in 10), and titrate with 0.1 N sodium hydroxide VS, either a glass-calomel or a silver–silver chloride electrode system with potassium nitrate filling solution. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N sodium hydroxide is equivalent to 2.503 mg of ethynyl group (–C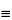 CH). Not less than 8.18% and not more than 8.43% of ethynyl group is found.
CH). Not less than 8.18% and not more than 8.43% of ethynyl group is found.
Chromatographic purity—
Test solution—
Prepare a solution of Norethindrone in chloroform to contain 10 mg per mL.
Standard solutions—
Prepare a solution of USP Norethindrone RS in chloroform to contain 10 mg per mL (Standard stock solution). Dilute accurately measured volumes of Standard stock solution with chloroform to obtain Standard solutions A, B, C, and D having known concentrations of 150, 50, 30, and 10 µg per mL, respectively.
Procedure—
Separately apply 10 µL of the Test solution and 10 µL of each Standard solution to a thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of chromatographic silica gel mixture. Develop the chromatogram in a solvent system consisting of a mixture of chloroform and methanol (95:5) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the chamber, mark the solvent front, and allow the solvent to evaporate. Spray the plate with a mixture of methanol and sulfuric acid (7:3), then heat the plate at 100
) coated with a 0.25-mm layer of chromatographic silica gel mixture. Develop the chromatogram in a solvent system consisting of a mixture of chloroform and methanol (95:5) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the chamber, mark the solvent front, and allow the solvent to evaporate. Spray the plate with a mixture of methanol and sulfuric acid (7:3), then heat the plate at 100 for 5 minutes: the RF value of the principal spot from the Test solution corresponds to that of the principal spot from Standard solution A. Compare the intensities of any secondary spots observed in the chromatogram of the Test solution with those of the principal spots in the chromatograms of the Standard solution: no secondary spot from the chromatogram of the Test solution is larger or more intense than the principal spot obtained from Standard solution B (0.5%), and the sum of the intensities of the secondary spots obtained from the Test solution is not more intense than the principal spot obtained from Standard solution A (1.5%).
for 5 minutes: the RF value of the principal spot from the Test solution corresponds to that of the principal spot from Standard solution A. Compare the intensities of any secondary spots observed in the chromatogram of the Test solution with those of the principal spots in the chromatograms of the Standard solution: no secondary spot from the chromatogram of the Test solution is larger or more intense than the principal spot obtained from Standard solution B (0.5%), and the sum of the intensities of the secondary spots obtained from the Test solution is not more intense than the principal spot obtained from Standard solution A (1.5%).
Assay—
Dissolve about 100 mg of Norethindrone, accurately weighed, in alcohol, and dilute quantitatively and stepwise with alcohol to obtain a solution containing about 10 µg per mL. Dissolve an accurately weighed quantity of USP Norethindrone RS in alcohol, and dilute quantitatively and stepwise with alcohol to obtain a Standard solution having a known concentration of about 10 µg per mL. Concomitantly determine the absorbances of both solutions in 1-cm cells at the wavelength of maximum absorbance at about 240 nm, using alcohol as the blank. Calculate the quantity, in mg, of C20H26O2 in the portion of Norethindrone taken by the formula:
10C(AU / AS)
in which C is the concentration, in µg per mL, of USP Norethindrone RS in the Standard solution, and AU and AS are the absorbances of the solution of Norethindrone and the Standard solution, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Mary S. Waddell
Scientific Liaison 1-301-816-8124 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4075