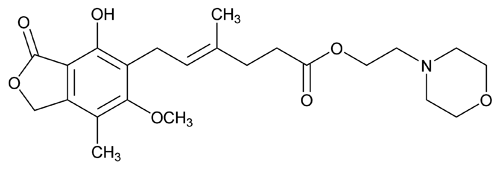
Mycophenolate Mofetil
C23H31NO7 433.49
4-Hexenoic acid, 6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-, 2-(4-morpholinyl)ethyl ester, (E);
2-Morpholinoethyl (E)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-5-phthalanyl)-4-methyl-4-hexenoate

 [115007-34-6].
[115007-34-6].
(mye'' koe fen' oh late moe' fe til).
C23H31NO7 433.49
4-Hexenoic acid, 6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-, 2-(4-morpholinyl)ethyl ester, (E);
2-Morpholinoethyl (E)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-5-phthalanyl)-4-methyl-4-hexenoate
DEFINITION
Mycophenolate Mofetil contains NLT 98.0% and NMT 102.0% of C23H31NO7, calculated on the dried basis.
IDENTIFICATION
• B.
The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
ASSAY
• Procedure
Buffer:
Triethylamine and water (1:325). Adjust with phosphoric acid to a pH of 5.3.
Mobile phase:
Acetonitrile and Buffer (7:13)
Standard solution:
1.0 mg/mL of USP Mycophenolate Mofetil RS in acetonitrile
Sample solution:
1 mg/mL of Mycophenolate Mofetil in acetonitrile
Chromatographic system
Mode:
LC
Detector:
UV 250 nm
Column:
4.6-mm × 15-cm; packing L7
Temperature:
45
Flow rate:
1.5 mL/min
Injection size:
10 µL
System suitability
Sample:
Standard solution
Suitability requirements
Column efficiency:
NLT 8000 theoretical plates
Tailing factor:
NMT 1.5
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C23H31NO7 in the portion of Mycophenolate Mofetil taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Mycophenolate Mofetil RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Mycophenolate Mofetil in the Sample solution (mg/mL) |
Acceptance criteria:
98.0%–102.0% on the dried basis
IMPURITIES
Organic Impurities
• Procedure 1
Buffer, Mobile phase, and Sample solution:
Prepare as directed in the Assay.
System suitability solution:
10 µg/mL each of USP Mycophenolate Mofetil Related Compound A RS and USP Mycophenolate Mofetil Related Compound B RS in acetonitrile
Chromatographic system
Mode:
LC
Detector:
UV 250 nm
Column:
4.6-mm × 25-cm; packing L7
Temperature:
45
Flow rate:
1.5 mL/min
Injection size:
10 µL
System suitability
Sample:
System suitability solution
Suitability requirements
Resolution:
NLT 1.5 between mycophenolate mofetil related compound A and mycophenolate mofetil related compound B
Analysis
Sample:
Sample solution
Calculate the percentage of each impurity in the portion of Mycophenolate Mofetil taken:
Result = (rU/rT) × 100
| rU | = | = peak response for each impurity |
| rT | = | = sum of all the peak responses |
Acceptance criteria
Individual impurities:
See Impurity Table 1.
Total impurities:
NMT 0.70%
[Note—Disregard any unknown peak less than 0.03%. ]
Impurity Table 1
| Name | Relative Retention Time |
Acceptance Criteria, NMT (%) |
|---|---|---|
| Mycophenolic acida | 0.33 | 0.50 |
| Mycophenolate mofetil related compound Ab | 0.45 | 0.10 |
| Mycophenolate mofetil related compound Bc | 0.49 | 0.10 |
| N-Oxide analogd | 0.60 | 0.10 |
| 1-Morpholinoethoxy analoge | 0.86 | 0.10 |
| Mycophenolate mofetil | 1.0 | — |
| O-Methyl analogf | 1.2 | 0.10 |
| Methyl mycophenolateg | 1.5 | 0.10 |
| Any single unspecified impurity | — | 0.10 |
|
a
(E)-6-(1,3-Dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexenoic acid.
b
2-Morpholinoethyl (E)-6-(1,3-dihydro-4,6-dihydroxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexenoate.
c
(RS)-7-Hydroxy-5-methoxy-4-methyl-6-[2-(5-methyl-2-oxo-tetrahydrofuran-5-yl)ethyl]-3H-isobenzofuranyl-1-one.
d
2-Morpholinoethyl (E)-6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexenoate N-oxide.
e
2-Morpholinoethyl (RS)-(E)-6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-1-(2-morpholinoethoxy)-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexenoate.
f
2-Morpholinoethyl (E)-6-(1,3-dihydro-4,6-dimethoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexenoate.
g
Methyl (E)-6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexenoate.
|
||
SPECIFIC TESTS
• Loss on Drying  731
731 :
Dry a sample in a vacuum at 60
:
Dry a sample in a vacuum at 60 for 3 h: it loses NMT 0.5% of its weight.
for 3 h: it loses NMT 0.5% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers, and store at room temperature.
• USP Reference Standards  11
11
USP Mycophenolate Mofetil Related Compound A RS
2-Morpholinoethyl (E)-6-(1,3-dihydro-4,6-dihydroxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexenoate.
C23H31NO7 419.47
2-Morpholinoethyl (E)-6-(1,3-dihydro-4,6-dihydroxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexenoate.
C23H31NO7 419.47
USP Mycophenolate Mofetil Related Compound B RS
(RS)-7-Hydroxy-5-methoxy-4-methyl-6-[2-(5-methyl-2-oxo-tetrahydrofuran-5-yl)ethyl]-3H-isobenzofuranyl-1-one.
C17H20O6 320.34
(RS)-7-Hydroxy-5-methoxy-4-methyl-6-[2-(5-methyl-2-oxo-tetrahydrofuran-5-yl)ethyl]-3H-isobenzofuranyl-1-one.
C17H20O6 320.34
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Feiwen Mao, M.S.
Senior Scientific Liaison 1-301-816-8320 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3967
Pharmacopeial Forum: Volume No. 35(1) Page 89