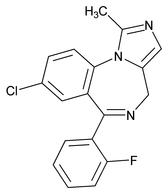
Midazolam
C18H13ClFN3 325.77
4-H-Imidazo[1,5-a][1,4]benzodiazepine, 8-chloro-6-(2-fluorophenyl)-1-methyl;
8-Chloro-6-(o-fluorophenyl)-1-methyl-4H-imidazo[1,5-a][1,4]benzodiazepine

 [59467-70-8].
[59467-70-8].
C18H13ClFN3 325.77
4-H-Imidazo[1,5-a][1,4]benzodiazepine, 8-chloro-6-(2-fluorophenyl)-1-methyl;
8-Chloro-6-(o-fluorophenyl)-1-methyl-4H-imidazo[1,5-a][1,4]benzodiazepine
DEFINITION
Midazolam contains NLT 98.5% and NMT 101.5% of C18H13ClFN3, calculated on the dried basis.
IDENTIFICATION
• B.
The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
ASSAY
• Procedure
Buffer:
7.7 g/L of ammonium acetate in water. Adjust with glacial acetic acid to a pH of 5.5 ± 0.1.
Mobile phase:
Acetonitrile and Buffer (1:2)
Standard solution:
0.04 mg/mL of USP Midazolam RS in Mobile phase
Sample solution:
0.04 mg/mL of Midazolam in Mobile phase
Chromatographic system
Mode:
LC
Detector:
UV 254 nm
Column:
4.6-mm × 25-cm; 5-µm packing L60
Flow rate:
1.5 mL/min
Injection size:
25 µL
System suitability
Sample:
Standard solution
Suitability requirements
Column efficiency:
NLT 10,000 theoretical plates
Tailing factor:
NMT 2.0
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C18H13ClFN3 in the portion of Midazolam taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Midazolam RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Midazolam in the Sample solution (mg/mL) |
Acceptance criteria:
98.5%–101.5% on the dried basis
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.1%
:
NMT 0.1%
Organic Impurities
• Procedure
Buffer, Mobile phase, Standard solution, and Chromatographic system:
Proceed as directed in the Assay.
Sensitivity check solution:
Dilute the Standard solution with Mobile phase to obtain a 0.2-µg/mL solution.
Sample solution:
0.2 mg/mL of Midazolam in Mobile phase
System suitability
Samples:
Standard solution and Sensitivity check solution
Suitability requirements
Column efficiency:
NLT 10,000 theoretical plates, Standard solution
Tailing factor:
NMT 2.0, Standard solution
Relative standard deviation:
NMT 2.0%, Standard solution
Peak ratio:
The ratio of the area of the midazolam peak of the Standard solution to the area of the midazolam peak of the Sensitivity check solution should be within 160–240.
Analysis
Sample:
Sample solution
Calculate the percentage of each impurity in the portion of Midazolam taken:
Result = (rU/F)/[S(rU/F) + rT] × 100
| rU | = | = peak response of each individual impurity from the Sample solution |
| rT | = | = peak response of Midazolam from the Sample solution |
| F | = | = relative response factor (see Impurity Table 1) |
Acceptance criteria:
See Impurity Table 1.
Impurity Table 1
| Name | Relative Retention Time |
Relative Response Factor |
Acceptance Criteria, NMT (%) |
|---|---|---|---|
| Reduced midazolama | 0.20 | 1.0 | 0.1 |
| Reduced reduced midazolamb | 0.24 | 1.0 | 0.1 |
| Amino compoundc | 0.25 | 0.5 | 0.1 |
| Oxide midazolamd | 0.46 | 1.3 | 0.1 |
| Nitromethylene compounde | 0.76 | 1.0 | 0.1 |
| Dihydromidazolamf | 0.83 | 0.5 | 0.1 |
| Midazolam | 1.0 | — | — |
| Desfluoromidazolamg | 1.14 | 1.0 | 0.2 |
| 6H-isomerh | 2.48 | 0.7 | 0.1 |
| Unknown impurity | — | 1.0 | 0.1 |
| Total impurities | — | — | 0.5 |
|
a
8-Chloro-3a,4-dihydro-6-(2-fluorophenyl)-1-methyl-3H-imidazo[1,5-a][1,4]-benzodiazepine.
b
8-Chloro-6-(2-fluorophenyl)-3a,4,5,6-tetrahydro-1-methyl-3H-imidazo[1,5-a][1,4]-benzodiazepine.
c
2-Aminomethyl-7-chloro-2,3-dihydro-5-(2-fluorophenyl)-1H-1,4-benzodiazepine.
d
8-Chloro-6-(2-fluorophenyl)-1-methyl-4H-imidazo[1,5-a][1,4]-benzodiazepine-5-oxide.
e
7-Chloro-1,3-dihydro-2-nitromethylene-5-(2-fluorophenyl)-2H-1,4-benzodiazepine-4-oxide.
f
8-Chloro-6-(2-fluorophenyl)-5,6-dihydro-1-methyl-4H-imidazo[1,5-a][1,4]-benzodiazepine.
g
8-Chloro-6-phenyl-1-methyl-4H-imidazo-[1,5-a][1,4]-benzodiazepine.
h
8-Chloro-6-(2-fluorophenyl)-1-methyl-6H-imidazo[1,5-a][1,4]-benzodiazepine.
|
|||
SPECIFIC TESTS
• Loss on Drying  731
731 :
Dry a sample at 105
:
Dry a sample at 105 for 2 h: it loses NMT 0.5% of its weight.
for 2 h: it loses NMT 0.5% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight, light-resistant containers.
• USP Reference Standards  11
11
USP Midazolam RS
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Mary S. Waddell
Scientific Liaison 1-301-816-8124 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3919
Pharmacopeial Forum: Volume No. 34(4) Page 961