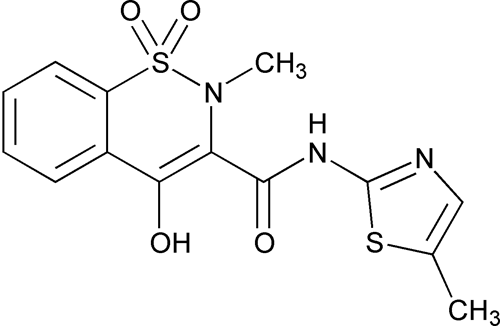
Meloxicam
C14H13N3O4S2 351.40
4-Hydroxy-2-methyl-N-(5-methyl-2-thiazolyl)-2H-1,2-benzothiazine-3-carboxamide 1,1-dioxide

 [71125-38-7].
[71125-38-7].
(mel ox' i kam).
C14H13N3O4S2 351.40
4-Hydroxy-2-methyl-N-(5-methyl-2-thiazolyl)-2H-1,2-benzothiazine-3-carboxamide 1,1-dioxide
DEFINITION
Meloxicam contains NLT 99.0% and NMT 100.5% of C14H13N3O4S2, calculated on the dried basis.
IDENTIFICATION
ASSAY
• Procedure
Solution A:
Mixture of a 0.1% (w/v) solution of ammonium acetate adjusted with 10% ammonia solution to a pH of 9.1
Mobile phase:
Methanol and Solution A (21:29)
Diluent:
Methanol and 1 N sodium hydroxide (250:1)
System suitability solution:
Dissolve 4 mg each of USP Meloxicam RS and USP Meloxicam Related Compound A RS in 25 mL of Diluent, and complete the volume to 50 mL with water.
Standard solution:
Dissolve 20 mg of USP Meloxicam RS in 50 mL of Diluent, and complete the volume to 100 mL with water.
Sample solution:
Dissolve 20 mg of Meloxicam in 50 mL of Diluent, and complete the volume to 100 mL with water.
Chromatographic system
Mode:
LC
Detector:
UV 360 nm
Column:
4.6-mm × 15-cm; packing L1
Column temperature:
45
Flow rate:
1.0 mL/min
Injection size:
10 µL
System suitability
Sample:
System suitability solution
[Note—The relative retention times for meloxicam related compound A and meloxicam are 0.7 and 1.0, respectively. ]
Suitability requirements
Resolution:
NLT 3.0 between meloxicam related compound A and meloxicam
Tailing factor:
NMT 2.0 for meloxicam peak
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C14H13N3O4S2 in the portion of Meloxicam taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response of Meloxicam from the Sample solution |
| rS | = | = peak response of meloxicam from the Standard solution |
| CS | = | = concentration of USP Meloxicam RS in the Standard solution (mg/mL) |
| CU | = | = concentration of the Sample solution (mg/mL) |
Acceptance criteria:
99.0%–100.5% on the dried basis
IMPURITIES
Organic Impurities
• Procedure 1
[Note—Perform either Procedure 1 or Procedure 2, depending on the manufacturing process used. ]
Solution A:
0.1% (w/v) solution of monobasic potassium phosphate adjusted with 1 N sodium hydroxide to a pH of 6.0
Solution B:
Methanol
Diluent:
Methanol and 1 N sodium hydroxide (50:3)
Mobile phase:
Use the gradient table below.
| Time (min) |
Solution A (%) |
Solution B (%) |
|---|---|---|
| 0 | 60 | 40 |
| 2 | 60 | 40 |
| 10 | 30 | 70 |
| 15 | 30 | 70 |
| 15.1 | 60 | 40 |
| 18 | 60 | 40 |
System suitability solution:
Dissolve 4 mg each of USP Meloxicam RS, USP Meloxicam Related Compound A RS, and USP Meloxicam Related Compound B RS in 5 mL of Diluent, and complete the volume to 50 mL with methanol.
Standard solution:
Dissolve 12 mg of USP Meloxicam RS in 5 mL of Diluent, complete the volume to 20 mL with methanol, and dilute 2 mL to 100 mL with methanol.
Sample solution:
Dissolve 80 mg of Meloxicam in 5 mL of Diluent, and complete the volume to 20 mL with methanol.
Chromatographic system
Mode:
LC
Detector:
UV 260–350 nm (variable wavelength or multi-wavelength detector)
Column:
4.6-mm × 15-cm; 5-µm packing L1
Column temperature:
45
Flow rate:
1 mL/min
Injection size:
5 µL
System suitability
Samples:
Standard solution and System suitability solution
[Note—Relative retention times are listed in Impurity Table 1. ]
Suitability requirements
Resolution:
NLT 3.0 between meloxicam related compound A and meloxicam at 350 nm, and NLT 3.0 between meloxicam related compound B and meloxicam at 260 nm, System suitability solution
Relative standard deviation:
NMT 10%, Standard solution
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of each impurity in the portion of Meloxicam taken:
Result = (rU/rS) × (CS/CU) × (1/F) × 100
| rU | = | = peak response of each impurity from the Sample solution |
| rS | = | = peak response of meloxicam at 350 nm from the Standard solution |
| CS | = | = concentration of USP Meloxicam RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Meloxicam in the Sample solution (mg/mL) |
| F | = | = relative response factor (see Impurity Table 1) |
[Note—For the specified impurities, calculate the percentage content of each impurity, using the peak responses from the Sample solution recorded at the detection wavelength given in Impurity Table 1. For an unknown impurity, calculate the percentage content, using peak responses recorded at the wavelength that gives the greater response. ]
Acceptance criteria
Individual impurity:
See Impurity Table 1.
Total impurities:
NMT 0.3%
Impurity Table 1
| Name | Relative Retention Time |
Wavelength (nm) |
Relative Response Factor |
Acceptance Criteria, NMT (%) |
|---|---|---|---|---|
| Meloxicam related compound Ba | 0.4 | 260 | 1.0 | 0.1 |
| Meloxicam | 1.0 | 260/350 | — | — |
| Meloxicam related compound Ab | 1.4 | 350 | 0.5 | 0.1 |
| Methyl-meloxicamc | 1.7 | 350 | 1.0 | 0.05 |
| Ethyl-meloxicamd | 1.9 | 350 | 1.0 | 0.05 |
| Individual unknown impurity | — | 260/350 | 1.0 | 0.1 |
|
a
2-Amino-5-methyl-thiazole.
b
4-Hydroxy-2-methyl-2H-1,2-benzothiazine-3-carboxylic acid ethyl ester 1,1-dioxide.
c
N-(3,5-Dimethylthiazol-2(3H)-ylidene)-4-hydroxy-2-methyl-2H-benzo[e][1,2]thiazine-3-carboxamide 1,1-dioxide.
d
N-(3-Ethyl-5-methylthiazol-2(3H)-ylidene)-4-hydroxy-2-methyl-2H-benzo[e][1,2]thiazine-3-carboxamide 1,1-dioxide.
|
||||
• Procedure 2:
If an article complies with this test, the labeling indicates that it meets the requirements under Organic Impurities, Procedure 2.
Solution A, Solution B, and Mobile phase:
Proceed as directed in Procedure 1.
Mobile phase:
See the gradient table below.
| Time (min) |
Solution A (%) |
Solution B (%) |
|---|---|---|
| 0 | 45 | 55 |
| 25 | 45 | 55 |
| 30 | 30 | 70 |
| 40 | 30 | 70 |
| 45 | 45 | 55 |
| 50 | 45 | 55 |
Diluent A:
Diluent B and 0.4 N sodium hydroxide (50:3)
Diluent B:
Methanol and water (2:3)
Standard stock solution A:
50 µg/mL of USP Meloxicam RS in Diluent A. Dilute 2 mL of this solution with Diluent B to 10 mL.
Standard stock solution B:
Transfer 5 mg each of USP Meloxicam Related Compound B RS, USP Meloxicam Related Compound C RS, and USP Meloxicam Related Compound D RS into a 100-mL volumetric flask. Add 6 mL of 0.4 N sodium hydroxide, and sonicate for 2 min. Add 40 mL of methanol to the resulting solution, sonicate for 2 min, and dilute with water to volume.
Standard solution:
Transfer 1 mL each of Standard stock solution A and Standard stock solution B into a 10-mL volumetric flask, dilute with Diluent B to volume, and mix.
System suitability stock solution:
2 mg/mL of USP Meloxicam RS in Diluent A
System suitability solution:
Transfer 5 mL of System suitability stock solution and 1 mL of Standard stock solution B into a 10-mL volumetric flask, dilute with Diluent B to volume, and mix.
Sample solution:
Dissolve 20 mg of Meloxicam in 10 mL of Diluent A, and dilute with Diluent B to 20 mL.
Chromatographic system
Mode:
LC
Detector:
UV variable wavelength or multi-wavelength detector at 260 nm and 350 nm
Column:
4.6-mm × 25-cm; 5-µm packing L1
Column temperature:
45
Flow rate:
1 mL/min
Injection size:
20 µL
System suitability
Samples:
Standard solution and System suitability solution
[Note—Relative retention times are listed in Impurity Table 2. ]
Suitability requirements
Resolution:
NLT 5.0 between meloxicam related compound D and meloxicam at 350 nm, System suitability solution
Relative standard deviation:
NMT 5.0% for meloxicam related compound C and for meloxicam related compound D at 350 nm and NMT 5.0% for meloxicam related compound B at 260 nm, Standard solution
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of each impurity in the portion of Meloxicam taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response of each impurity from the Sample solution |
| rS | = | = peak response of the corresponding related compound from the Standard solution |
| CS | = | = concentration of the corresponding USP Related Compound RS in the Standard solution (mg/mL). [Note—Use the concentration of the USP Meloxicam RS for unknown impurities. ] |
| CU | = | = concentration of Meloxicam in the Sample solution (mg/mL) |
[Note—Use the peak response and concentration of USP Meloxicam RS for unknown impurities; for the specified impurities, calculate the percentage content of each impurity using the Sample solution peak responses recorded at the detection wavelength given in Impurity Table 2. For an unknown impurity, calculate the percentage content using peak responses recorded at the wavelength that gives the greater response. ]
Acceptance criteria
Individual impurities:
See Impurity Table 2.
Total impurities:
NMT 0.3%
Impurity Table 2
| Name | Relative Retention Time |
Wavelength (nm) |
Acceptance Criteria, NMT (%) |
|---|---|---|---|
| Meloxicam | 1.0 | 260/350 | — |
| Meloxicam related compound Ba | 0.8 | 260 | 0.1 |
| Meloxicam related compound Cb | 3.2 | 350 | 0.1 |
| Meloxicam related compound Dc | 2.4 | 350 | 0.1 |
| Individual unknown impurity | — | 260/350 | 0.1 |
|
a
2-Amino-5-methyl-thiazole.
b
Isopropyl-4-hydroxy-2-methyl-2H-1,2-benzothiazine-3-carboxylate-1,1-dioxide.
c
4-Methoxy-2-methyl-N-(5-methyl-1,3-thiazol-2yl)-2H-1,2-benzothiazine-3-carboxamide-1,1-dioxide.
|
|||
SPECIFIC TESTS
• Loss on Drying  731
731 :
Dry a sample at 105
:
Dry a sample at 105 for 4 h: it loses NMT 0.5% of its weight.
for 4 h: it loses NMT 0.5% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in well-closed containers. Store at room temperature.
• Labeling:
The labeling states with which Procedure under Organic Impurities the article complies if a test other than Procedure 1 is used.
• USP Reference Standards  11
11
USP Meloxicam Related Compound A RS 
4-Hydroxy-2-methyl-2H-1,2-benzothiazine-3-carboxylic acid ethylester 1,1-dioxide.

4-Hydroxy-2-methyl-2H-1,2-benzothiazine-3-carboxylic acid ethylester 1,1-dioxide.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Clydewyn M. Anthony, Ph.D.
Senior Scientific Liaison 1-301-816-8139 |
(SM22010) Monographs - Small Molecules 2 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3789
Pharmacopeial Forum: Volume No. 35(2) Page 278