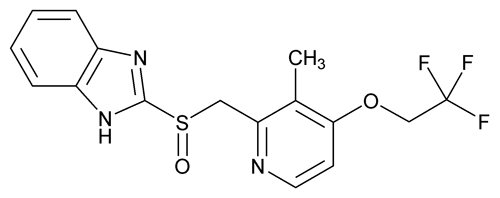
Lansoprazole
C16H14F3N3O2S 369.36
1H-Benzimidazole, 2-[[[3-methyl-4-(2,2,2-trifluoroethoxy)-2-pyridinyl]methyl]sulfinyl]-;
2-[[[3-Methyl-4-(2,2,2-trifluoroethoxy)-2-pyridyl]-methyl]sulfinyl]benzimidazole

 [103577-45-3].
[103577-45-3].

 USP35
USP35
 11
11
 USP Lansoprazole Related Compound B RS
USP Lansoprazole Related Compound B RS
2-[[[3-Methyl-4-(2,2,2-trifluoroethoxy)-pyridin-2-yl]methyl]sulfanyl]-1H-benzimidazole.
C16H14F3N3OS 353.36
 USP35
USP35
(lan soe' pra zole).
C16H14F3N3O2S 369.36
1H-Benzimidazole, 2-[[[3-methyl-4-(2,2,2-trifluoroethoxy)-2-pyridinyl]methyl]sulfinyl]-;
2-[[[3-Methyl-4-(2,2,2-trifluoroethoxy)-2-pyridyl]-methyl]sulfinyl]benzimidazole
DEFINITION
Lansoprazole contains NLT 98.0% and NMT 102.0% of C16H14F3N3O2S.
IDENTIFICATION
• B. Ultraviolet Absorption  197U
197U
Sample solution:
10 µg/mL in methanol
Acceptance criteria:
Meets the requirements
ASSAY
Change to read:
• Procedure
Mobile phase:
Acetonitrile, water, and triethylamine (40:60:1). Adjust with phosphoric acid to a pH of 7.0.
Diluent:
Acetonitrile, water, and triethylamine (40:60:1). Adjust with phosphoric acid to a pH of 10.0.
System suitability solution:
0.1 mg/mL of USP Lansoprazole RS and 0.1 mg/mL of USP Lansoprazole Related Compound A RS in Diluent
Standard solution:
 0.1 mg/mL of USP Lansoprazole RS in Diluent
0.1 mg/mL of USP Lansoprazole RS in Diluent USP35
USP35
Sample solution:
 0.1 mg/mL of Lansoprazole in Diluent
0.1 mg/mL of Lansoprazole in Diluent USP35
USP35
Chromatographic system
Mode:
LC
Detector:
UV 285 nm
Column:
4.6-mm × 25-cm; 5-µm packing L1
Flow rate:
1 mL/min
Injection size:
10 µL
System suitability
Samples:
System suitability solution and Standard solution
Suitability requirements
Resolution:
NLT 5 between lansoprazole and lansoprazole related compound A, System suitability solution
Relative standard deviation:
NMT 1.0%, Standard solution
Analysis
 Result = (rU/rS) × (CS/CU) × 100
Result = (rU/rS) × (CS/CU) × 100
Samples:
Standard solution and Sample solution
Calculate the percentage of lansoprazole (C16H14F3N3O2S) in the portion of Lansoprazole taken:
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Lansoprazole RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Lansoprazole in the Sample solution (mg/mL) |
Acceptance criteria:
98.0%–102.0%
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.1%
:
NMT 0.1%
Change to read:
Organic Impurities
• Procedure

 USP35
USP35
[Note—Store and inject the lansoprazole solutions at or below 5 using a cooled autosampler. The solutions are stable for about 24 h when stored at 5
using a cooled autosampler. The solutions are stable for about 24 h when stored at 5 . ]
. ]
Solution A:
Water
Solution B:
Acetonitrile, water, and triethylamine (160:40:1). Adjust with phosphoric acid to a pH of 7.0.
Diluent:
Methanol and 0.1 N sodium hydroxide (1:3)
Mobile phase:
See Table 1.
Table 1
| Time (min) |
Solution A (%) |
Solution B (%) |
|---|---|---|
| 0 | 90 | 10 |
| 40 | 20 | 80 |
| 50 | 20 | 80 |
| 51 | 90 | 10 |
| 60 | 90 | 10 |
System suitability solution:
Prepare a solution containing 25 µg/mL of USP Lansoprazole RS and 25 µg/mL of USP Lansoprazole Related Compound A RS in methanol. Transfer 1 mL of this solution into a 10-mL volumetric flask, and dilute with Diluent to volume.
Standard solution:
Prepare a solution containing 25 µg/mL of USP Lansoprazole RS  and 25 µg/mL of USP Lansoprazole Related Compound B RS
and 25 µg/mL of USP Lansoprazole Related Compound B RS USP35 in methanol. Transfer 1 mL of this solution into a
USP35 in methanol. Transfer 1 mL of this solution into a  100-mL
100-mL USP35 volumetric flask, and dilute with Diluent to volume.
USP35 volumetric flask, and dilute with Diluent to volume.
Sample solution:
2.5 mg/mL of Lansoprazole in methanol. Transfer 1 mL of this solution into a 10-mL volumetric flask, and dilute with Diluent to volume.
Blank:
Methanol and Diluent (1:9)
Chromatographic system
Mode:
LC
Detector:
UV 285 nm
Column:
4.6-mm × 15-cm; 5-µm packing L1
Flow rate:
0.8 mL/min
Injection size:
40 µL
System suitability
Sample:
System suitability solution 
 USP35
USP35
Suitability requirements
Resolution:
NLT 6 between lansoprazole and lansoprazole related compound A 
 USP35
USP35
Relative standard deviation:
NMT 3% 
 USP35
USP35
Analysis
 Calculate the percentage of lansoprazole related compound B in the portion of Lansoprazole taken:
Calculate the percentage of lansoprazole related compound B in the portion of Lansoprazole taken:
 USP35
USP35
Samples:
Standard solution, Sample solution, and Blank
Identify the lansoprazole peak and the peaks due to the impurities listed in Table 2. Measure the areas for the major peaks, excluding peaks obtained from the Blank.
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response for lansoprazole related compound B from the Sample solution |
| rS | = | = peak response for lansoprazole related compound B from the Standard solution |
| CS | = | = concentration of USP Lansoprazole Related Compound B RS in the Standard solution (µg/mL) |
| CU | = | = concentration of Lansoprazole in the Sample solution (µg/mL) |
Calculate the percentage of  lansoprazole N-oxide, lansoprazole sulfone, and any other individual
lansoprazole N-oxide, lansoprazole sulfone, and any other individual USP35 impurity in the portion of Lansoprazole taken:
USP35 impurity in the portion of Lansoprazole taken:
Result = (rU/rS) × (CS/CU) × (1/F) × 100
| rU | = | = peak response for each impurity from the Sample solution |
| rS | = | = peak response for lansoprazole from the Standard solution |
| CS | = | = concentration of USP Lansoprazole RS in the Standard solution (µg/mL) |
| CU | = | = concentration of Lansoprazole in the Sample solution (µg/mL) |
| F | = | = relative response factor for each impurity (see Table 2) |
Acceptance criteria
Individual impurities:
See Table 2.  Disregard any peak below 0.05%.
Disregard any peak below 0.05%. USP35
USP35
Table 2
| Name | Relative Retention Time |
Relative Response Factor |
Acceptance Criteria, NMT (%) |
|---|---|---|---|
| Lansoprazole N-oxidea |
0.8 | 1.3 | 0.1 |
| Lansoprazole | 1.0 | — | — |
| Lansoprazole related compound A (lansoprazole sulfone)b | 1.1 | 0.82 | 0.4 |
| 1.2 | 0.1 | ||
| Other individual impurity |
— | 1.00 | 0.1 |
| Total impurities | — | — | 0.6 |
|
a
[[(1H-Benzimidazole-2-yl)sulfinyl]methyl]-3-methyl-4-(2,2,2-trifluoroethoxy)-pyridine 1-oxide.
b
2-[[[3-Methyl-4-(2,2,2-trifluoroethoxy)-2-pyridyl]methyl]sulfonyl]benzimidazole.
c
2-[[[3-Methyl-4-(2,2,2-trifluoroethoxy)-pyridin-2-yl]methyl]sulfanyl]-1H-benzimidazole.
|
|||
SPECIFIC TESTS
• Water Determination, Method Ia  921
921
Sample:
1.0 g
[Note—Use 50 mL of a dehydrated mixture of pyridine and ethylene glycol (9:1 to 8:2) as the solvent. ]
Acceptance criteria:
NMT 0.1%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight, light-resistant containers. Store at room temperature, and protect from excessive heat.
Change to read:
• USP Reference Standards
USP Lansoprazole Related Compound A RS
2-[[[3-Methyl-4-(2,2,2-trifluoroethoxy)-2-pyridyl]methyl]sulfonyl]benzimidazole.
C16H14F3N3O3S 385.36
2-[[[3-Methyl-4-(2,2,2-trifluoroethoxy)-2-pyridyl]methyl]sulfonyl]benzimidazole.
C16H14F3N3O3S 385.36
2-[[[3-Methyl-4-(2,2,2-trifluoroethoxy)-pyridin-2-yl]methyl]sulfanyl]-1H-benzimidazole.
C16H14F3N3OS 353.36
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Elena Gonikberg, Ph.D.
Principal Scientific Liaison 1-301-816-8251 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3642
Pharmacopeial Forum: Volume No. 36(6) Page 1547