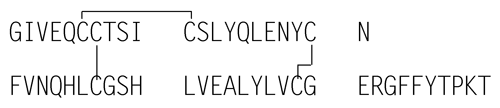Insulin Human
(in' su lin hue' man).
» Insulin Human is a protein corresponding to the active principle elaborated in the human pancreas that affects the metabolism of carbohydrate (particularly glucose), fat, and protein. It is derived by enzymatic modification of insulin from pork pancreas in order to change its amino acid sequence appropriately, or produced by microbial synthesis via a recombinant DNA process. Its potency, calculated on the dried basis, is not less than 27.5 USP Insulin Human Units in each mg. The proinsulin content of Insulin Human derived from pork, determined by a validated method, is not more than 10 ppm. The host cell derived proteins content of Insulin Human derived from a recombinant DNA process, determined by an appropriate and validated method, is not more than 10 ppm. The host cell or vector derived DNA content and limit of Insulin Human derived from a recombinant DNA process that utilizes eukaryotic host cells are determined by a validated method.
[note—One USP Insulin Human Unit is equivalent to 0.0347 mg of pure Insulin Human. ]
Packaging and storage—
Preserve in tight containers. Store in a freezer, and protect from light.
Labeling—
Label it to indicate that it has been prepared by microbial synthesis or that it is derived by enzymatic modification of insulin from pork pancreas.
Identification—
A:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of the Standard preparation, as obtained in the Assay.
B:
Determine the peptide fragments, using the following peptide mapping procedure.
Sulfate buffer—
Mix equal volumes of 2.0 M ammonium sulfate and 0.5 M sulfuric acid, and filter.
Enzyme solution—
Prepare a solution of Staphylococcus aureus V-8 protease in water having an activity of 500 units per mL.
HEPES buffer—
Dissolve 2.38 g of HEPES (N-2-hydroxyethylpiperazine-N¢-2-ethanesulfonic acid) in about 90 mL of water in a 100-mL volumetric flask. Adjust with 5 M sodium hydroxide to a pH of 7.5, dilute with water to volume, and mix.
Solution A—
Prepare a filtered and degassed mixture of 100 mL of acetonitrile, 700 mL of water, and 200 mL of Sulfate buffer.
Solution B—
Prepare a filtered and degassed mixture of 400 mL of acetonitrile, 400 mL of water, and 200 mL of Sulfate buffer.
Mobile phase—
Use variable mixtures of Solution A and Solution B as directed for Chromatographic system. Make adjustments, if necessary (see System Suitability under Chromatography  621
621 ).
).
Standard digest solution—
Dissolve about 6 mg of USP Insulin Human RS in 3 mL of 0.01 N hydrochloric acid, and transfer 500 µL of the resulting solution to a clean vial. Add 2.0 mL of HEPES buffer and 400 µL of Enzyme solution, and incubate at 25 for 6 hours. Quench the digestion by adding 2.9 mL of Sulfate buffer.
for 6 hours. Quench the digestion by adding 2.9 mL of Sulfate buffer.
Test digest solution—
To 1 mg of Insulin Human add 500 µL of 0.01 N hydrochloric acid, and mix to dissolve. Proceed as directed for Standard digest solution, beginning with “Add 2.0 mL of HEPES buffer”.
Chromatographic system (see Chromatography  621
621 )—
A liquid chromatograph is equipped with a 214-nm detector and a 4.6-mm × 10-cm column that contains packing L1. The flow rate is about 1 mL per minute. The column temperature is maintained at 40
)—
A liquid chromatograph is equipped with a 214-nm detector and a 4.6-mm × 10-cm column that contains packing L1. The flow rate is about 1 mL per minute. The column temperature is maintained at 40 . The chromatograph is programmed as follows.
. The chromatograph is programmed as follows.
Chromatograph the Standard digest solution, and record the peak responses as directed for Procedure: the chromatogram of the Standard digest solution corresponds to that of the standard chromatogram provided with USP Insulin Human RS. For the chromatogram of the Standard digest the tailing factor is not greater than 1.5; and the resolution, R, is not less than 3.4 for digest fragments II and III.
[note* —Fragment I elutes at the same time in insulin derived from pork and Insulin Human; Fragment II elutes at the same time in all insulins; and Fragment III elutes at the same time in insulin derived from beef and pork. ]
| Time (minutes) |
Solution A
(%) |
Solution B
(%) |
Elution |
|---|---|---|---|
| 0 | 90 | 10 | equilibration |
| 0–60 | 90®30 | 10®70 | linear gradient |
| 60–65 | 30®0 | 70®100 | linear gradient |
| 65–70 | 0 | 100 | isocratic |
| 70–71 | 0®90 | 100®10 | linear gradient |
| 71–86 | 90 | 10 | re-equilibration |
Procedure—
Using the gradient program, run a blank. Inject equal volumes of the Standard digest solution and the Test digest solution into the chromatograph, and record the chromatograms. The chromatographic profile of the Test digest solution corresponds to that of the Standard digest solution.
Bioidentity—
It meets the requirements of the Bioidentity test under Insulin.
Microbial enumeration tests  61
61 and Tests for specified microorganisms
and Tests for specified microorganisms  62
62 —
The total bacterial count does not exceed 300 cfu per g, the test being performed on a portion of about 0.2 g, accurately weighed.
—
The total bacterial count does not exceed 300 cfu per g, the test being performed on a portion of about 0.2 g, accurately weighed.
Bacterial endotoxins  85
85 —
It contains not more than 10 USP Endotoxin Units in each mg.
—
It contains not more than 10 USP Endotoxin Units in each mg.
Loss on drying  731
731 —
Dry about 200 mg, accurately weighed, at 105
—
Dry about 200 mg, accurately weighed, at 105 for 16 hours: it loses not more than 10.0% of its weight.
for 16 hours: it loses not more than 10.0% of its weight.
Related compounds—
Proceed as directed for the Related compounds test under Insulin except to use the following gradient elution program. The program initially calls for isocratic elution for about 36 minutes with a Mobile phase consisting of a mixture of 78% Solution A and 22% Solution B. Following the gradient elution phase, the system is returned to the initial conditions of 78% Solution A and 22% Solution B. Adjust the composition of the Mobile phase so that the retention time of the main insulin human peak is between 15 and 25 minutes. The content of A-21 desamido insulin and of other insulin related compounds is not more than 2.0% each of the total amount of insulin and total related compounds.
Limit of high molecular weight proteins—
Proceed as directed in the test for Limit of high molecular weight proteins under Insulin. Not more than 1.0% is found.
Other requirements—
It meets the requirements for Zinc content under Insulin.
Assay—
Mobile phase, Standard preparation, Assay preparation, System suitability solution, Chromatographic system, and Procedure—
Proceed as directed in the Assay under Insulin except to use USP Insulin Human RS and otherwise substitute Insulin Human for Insulin throughout.
*
Fragment I consists of amino acids A5 to A17 and B1 to B13. Fragment II consists of amino acids A18 to A21 and B14 to B21. Fragment III consists of amino acids B22 to B30. Fragment IV consists of amino acids A1 to A4. A refers to the A-chain of Insulin Human, and B refers to the B-chain of Insulin Human.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Thomas A. Sigambris, M.S.
Scientific Liaison 1-301-998-6789 |
(BIO12010) Monographs - Biologics and Biotechnology 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
|
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology |
USP35–NF30 Page 3506
Pharmacopeial Forum: Volume No. 31(5) Page 1375