Glyceryl Monooleate
Oleic acid, 2,3-dihydroxypropyl ester, (±);
(RS)-1-Glyceryl oleate 356.54

 [25496-72-4].
[25496-72-4].
(glis' er il mon'' oh oh' lee ate).
Oleic acid, 2,3-dihydroxypropyl ester, (±);
(RS)-1-Glyceryl oleate 356.54
DEFINITION
Glyceryl Monooleate is a mixture of monoglycerides, mainly glyceryl monooleate, together with variable quantities of diglycerides and triglycerides. It is obtained by partial glycerolysis of vegetable oil that consists mainly of triglycerides of oleic acid, or by esterification of glycerol with oleic acid of vegetable or animal origin. It is defined by the nominal content of monoglycerides. The assay requirements differ as set forth in the accompanying table. A suitable antioxidant may be added.
| Nominal Content of Monoglycerides (%) | |||
|---|---|---|---|
| 40 | 60 | 90 | |
| Monoglycerides | 32.0–52.0 | 55.0–65.0 | 90.0–101.0 |
| Diglycerides | 30.0–50.0 | 15.0–35.0 | <10.0 |
| Triglycerides | 5.0–20.0 | 2.0–10.0 | <2.0 |
IDENTIFICATION
• A. Thin-Layer Chromatographic Identification Test  201
201
Standard solution:
50 mg/mL of USP Glyceryl Monooleate 40% RS or USP Glyceryl Monooleate 90% RS in methylene chloride
Sample solution:
50 mg/mL of Glyceryl Monooleate in methylene chloride
Application volume:
10 µL
Developing solvent system:
Ether and hexane (7:3)
Spray reagent:
0.1 mg/mL of rhodamine B in alcohol
Analysis:
Proceed as directed in the chapter. Spray with the Spray reagent, and locate the spots on the plate by examination under UV light at a wavelength of 365 nm.
Acceptance criteria:
The principal spot of the Sample solution corresponds in color, size, and RF value to that of the Standard solution.
• B.
It meets the requirements in Specific Tests for Fats and Fixed Oils, Iodine Value  401
401 .
.
ASSAY
• Procedure
Mobile phase:
Tetrahydrofuran
Sample solution:
40 mg/mL of Glyceryl Monooleate in tetrahydrofuran
Chromatographic system
Mode:
LC
Detector:
Refractive index
Column:
7.5-mm × 60-cm; 5-µm 100-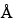 packing L21
packing L21
[Note—Two or three 7.5-mm × 30-cm L21 columns may be used in place of the one 60-cm column, provided that the system suitability requirements are met. The column temperature may be lowered to ambient temperature, although working at 40 provides stable separation conditions and ensures better sample solubility. ]
provides stable separation conditions and ensures better sample solubility. ]
Temperature
Column:
40
Detector:
40
Flow rate:
1 mL/min
Injection size:
40 µL
System suitability
Sample:
Sample solution
[Note—The relative retention times for triglycerides, diglycerides, monoglycerides, and glycerin are about 0.76, 0.79, 0.85, and 1.0, respectively. ]
Suitability requirements
Resolution:
NLT 1.0 between the diglycerides and monoglycerides
Relative standard deviation:
NMT 2.0%, determined from the monoglycerides peak
Analysis
Sample:
Sample solution
Calculate the percentage of monoglycerides, diglycerides, and triglycerides in the portion of Glyceryl Monooleate taken:
Result = (rU/rT) × 100
| rU | = | = individual peak responses for the monoglycerides, diglycerides, and triglycerides, as appropriate |
| rT | = | = sum of all the glyceride peak responses |
Acceptance criteria:
See the table in the Definition.
IMPURITIES
• Limit of Free Glycerin
Mobile phase, Sample solution, and Chromatographic system:
Proceed as directed in the Assay.
Standard solutions:
Prepare four solutions by dissolving glycerin in tetrahydrofuran, and diluting each with tetrahydrofuran as necessary, to obtain solutions having known concentrations of 0.4, 1.0, 2.0, and 4.0 mg/mL.
Standard curve
Samples:
Standard solutions
Plot:
Record the chromatograms, and measure the responses for the glycerin peaks. Plot the glycerin peak responses obtained versus the concentration, in mg/mL, of glycerin in the Standard solutions.
Analysis
Sample:
Sample solution
From the Standard curve, determine the glycerin concentration, in mg/mL, in the Sample solution.
Calculate the percentage of free glycerin in the portion of Glyceryl Monooleate taken:
Result = (CS/CU) × 100
| CS | = | = concentration of glycerin in the Sample solution from the Standard curve (mg/mL) |
| CU | = | = concentration of Glyceryl Monooleate in the Sample solution (mg/mL) |
Acceptance criteria:
NMT 6.0%
SPECIFIC TESTS
• Fats and Fixed Oils, Acid Value  401
401 :
NMT 6.0, determined on 1.0 g
:
NMT 6.0, determined on 1.0 g
• Fats and Fixed Oils, Iodine Value  401
401 :
65.0–95.0
:
65.0–95.0
• Fats and Fixed Oils, Peroxide Value  401
401 :
NMT 12.0, determined on 2.0 g
:
NMT 12.0, determined on 2.0 g
• Fats and Fixed Oils, Saponification Value  401
401 :
150–175, determined on 2.0 g
:
150–175, determined on 2.0 g
• Fats and Fixed Oils, Fatty Acid Composition  401
401 :
Glyceryl Monooleate exhibits the following composition profile of fatty acids (see Table 1), determined as directed in the chapter.
:
Glyceryl Monooleate exhibits the following composition profile of fatty acids (see Table 1), determined as directed in the chapter.
Table 1
| Carbon-Chain Length | Number of Double Bonds |
Percentage, NMT (%) |
|---|---|---|
| 16 | 0 | 12.0 |
| 18 | 0 | 6.0 |
| 18 | 1 | 60.0 |
| 18 | 2 | 35.0 |
| 18 | 3 | 2.0 |
| 20 | 0 | 2.0 |
| 20 | 1 | 2.0 |
• Water Determination, Method I  921
921 :
NMT 1.0%, using a mixture of methanol and methylene chloride (1:1) in place of methanol in the titration vessel
:
NMT 1.0%, using a mixture of methanol and methylene chloride (1:1) in place of methanol in the titration vessel
• Articles of Botanical Origin, Total Ash  561
561 :
NMT 0.1%
:
NMT 0.1%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers. No storage requirements specified.
• Labeling:
The labeling indicates the nominal content of monoglycerides and the name and the concentration of any added antioxidant.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 1814
Pharmacopeial Forum: Volume No. 33(6) Page 1249
