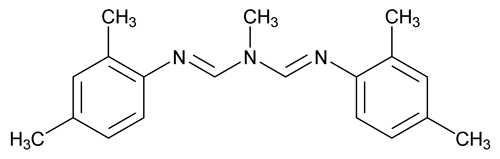
Amitraz
(a' mi traz).
Methanimidamide, N¢-(2,4-dimethylphenyl)-[[N-(2,4-dimethylphenyl)imino]methyl-N]-methyl-.
N-Methyl-N¢-2,4-xylyl-N-(N-2,4-xylylformimidoyl)formamidine.
N-Methylbis(2,4-xylyliminomethyl)amine
» Amitraz contains not less than 95.0 percent and not more than 101.5 percent of C19H23N, calculated on the anhydrous basis.
Packaging and storage—
Preserve in well-closed containers.
Labeling—
Label it to indicate that it is for veterinary use only.
Identification—
B:
Proceed as directed in the test for Related compounds, except to prepare a test solution of Amitraz in toluene containing 2 mg per mL and a Standard solution of USP Amitraz RS in toluene containing 2 mg per mL: the RF value of the principal spot in the chromatogram obtained from the test solution corresponds to that in the chromatogram obtained from the Standard solution.
C:
The retention time of the major peak in the chromatogram of the Reference solution corresponds to that in the chromatogram of the Standard preparation, as obtained in the Assay.
Water, Method I  921
921 :
not more than 0.1%, anhydrous pyridine being used in place of methanol in the titration vessel.
:
not more than 0.1%, anhydrous pyridine being used in place of methanol in the titration vessel.
Residue on ignition  281
281 :
not more than 0.2%.
:
not more than 0.2%.
Related compounds—
Prepare a test solution of Amitraz in toluene containing 100 mg per mL. Prepare a solution of USP Amitraz RS in toluene having a concentration of 2.0 mg per mL (Standard solution 1). Prepare a solution of 2,4-dimethylaniline in toluene having a concentration of 0.30 mg per mL (Standard solution 2). Prepare a thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of chromatographic silica gel mixture as follows. Stand the plate to a depth of 3.5 cm in a solution prepared by dissolving 35 g of acetamide in 100 mL of methanol, adding 100 mL of triethylamine, and diluting to 250 mL with methanol. Allow to stand the wet plate in a current of cold air for about 30 seconds. Immediately apply separately to the plate, at a level about 1 cm below the top of the impregnated zone, 2 µL each of the test solution, Standard solution 1, and Standard solution 2. Promptly develop the chromatogram in a solvent system consisting of a mixture of cyclohexane, ethyl acetate, and triethylamine (5:3:2) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, allow it to air-dry, and examine it under short-wavelength UV light. Any secondary spot in the chromatogram obtained from the test solution is not more intense than the spot in the chromatogram obtained from Standard solution 1 (2.0%). Expose the plate to the vapor of hydrochloric acid for about 10 minutes, then expose it to the vapor of nitrogen dioxide (prepared by the reaction of nitric acid and zinc) for 10 minutes, remove any excess nitric oxide by air exhaust, and spray the plate with a 0.5% solution of N-(1-naphthyl)ethylenediamine dihydrochloride in methanol, and examine the plate. Any secondary spot in the chromatogram obtained from the test solution corresponding to 2,4-dimethylaniline is not more intense than the spot in the chromatogram obtained from Standard solution 2 (0.30%).
) coated with a 0.25-mm layer of chromatographic silica gel mixture as follows. Stand the plate to a depth of 3.5 cm in a solution prepared by dissolving 35 g of acetamide in 100 mL of methanol, adding 100 mL of triethylamine, and diluting to 250 mL with methanol. Allow to stand the wet plate in a current of cold air for about 30 seconds. Immediately apply separately to the plate, at a level about 1 cm below the top of the impregnated zone, 2 µL each of the test solution, Standard solution 1, and Standard solution 2. Promptly develop the chromatogram in a solvent system consisting of a mixture of cyclohexane, ethyl acetate, and triethylamine (5:3:2) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, allow it to air-dry, and examine it under short-wavelength UV light. Any secondary spot in the chromatogram obtained from the test solution is not more intense than the spot in the chromatogram obtained from Standard solution 1 (2.0%). Expose the plate to the vapor of hydrochloric acid for about 10 minutes, then expose it to the vapor of nitrogen dioxide (prepared by the reaction of nitric acid and zinc) for 10 minutes, remove any excess nitric oxide by air exhaust, and spray the plate with a 0.5% solution of N-(1-naphthyl)ethylenediamine dihydrochloride in methanol, and examine the plate. Any secondary spot in the chromatogram obtained from the test solution corresponding to 2,4-dimethylaniline is not more intense than the spot in the chromatogram obtained from Standard solution 2 (0.30%).
Assay—
Internal standard solution—
Prepare a solution of squalane in methyl acetate containing 10 mg per mL.
Standard preparation—
Quantitatively dissolve an accurately weighed quantity of USP Amitraz RS in Internal standard solution to obtain a solution having a known concentration of about 8 mg of USP Amitraz RS per mL.
Assay preparation—
Transfer about 200 mg of Amitraz, accurately weighed, to a 25-mL volumetric flask, add about 20 mL of Internal standard solution, and swirl to dissolve. Dilute with Internal standard solution to volume, and mix.
Reference solution—
Prepare a solution of Amitraz in methyl acetate containing about 8 mg per mL.
Chromatographic system (see Chromatography  621
621 )—
The gas chromatograph is equipped with a flame-ionization detector and a 4-mm × 1.5-m column packed with 3% liquid phase G1 on support S1A. The column and detector block temperatures are maintained at about 250
)—
The gas chromatograph is equipped with a flame-ionization detector and a 4-mm × 1.5-m column packed with 3% liquid phase G1 on support S1A. The column and detector block temperatures are maintained at about 250 . Dry nitrogen is used as the carrier gas at a flow rate of about 60 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the resolution, R, between squalane and amitraz is not less than 3.0; and the relative standard deviation for replicate injections is not more than 2.0%.
. Dry nitrogen is used as the carrier gas at a flow rate of about 60 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the resolution, R, between squalane and amitraz is not less than 3.0; and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 5 µL) of the Standard preparation, the Assay preparation, and the Reference solution into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the quantity, in mg, of C19H23N3 in the portion of Amitraz taken by the formula:
25C(RU / RS)
in which C is the concentration, in mg per mL, of USP Amitraz RS in the Standard preparation; and RU and RS are the ratios of the response of the amitraz peak to the response of the squalane peak obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Morgan Puderbaugh, B.S.
Associate Scientific Liaison 1-301-998-6833 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2182