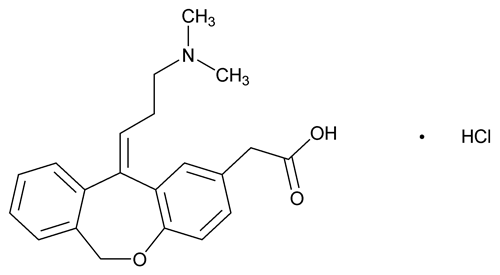
Olopatadine Hydrochloride
C21H23NO3·HCl 373.87
Dibenz[b,e]oxepin-2-acetic acid, 11-[3-(dimethylamino)propylidene]-6,11-dihydro-, hydrochloride, (Z)-;
11-[(Z)-3-(Dimethylamino)propylidene]-6,11-dihydrodibenz[b,e]oxepin-2-acetic acid, hydrochloride

 [140462-76-6].
[140462-76-6].
(oh'' loe pa' ta deen hye'' droe klor' ide).
C21H23NO3·HCl 373.87
Dibenz[b,e]oxepin-2-acetic acid, 11-[3-(dimethylamino)propylidene]-6,11-dihydro-, hydrochloride, (Z)-;
11-[(Z)-3-(Dimethylamino)propylidene]-6,11-dihydrodibenz[b,e]oxepin-2-acetic acid, hydrochloride
DEFINITION
Olopatadine Hydrochloride contains NLT 98.0% and NMT 102.0% of C21H23NO3·HCl, calculated on the dried basis.
IDENTIFICATION
• B.
The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
• C. Identification Tests—General, Chloride  191
191 :
Meets the requirements
:
Meets the requirements
ASSAY
• Procedure
[Note—Protect solutions from light. ]
Buffer:
Dissolve 13.6 g of monobasic potassium phosphate in 1 L of water, add 1 mL of triethylamine, and mix. Adjust with phosphoric acid to a pH of 3.0.
Mobile phase:
Acetonitrile and Buffer (7:18)
Standard solution:
0.1 mg/mL of USP Olopatadine Hydrochloride RS in Mobile phase
Sample solution:
0.1 mg/mL of Olopatadine Hydrochloride in Mobile phase
Chromatographic system
Mode:
LC
Detector:
UV 299 nm
Column:
4.6-mm × 15-cm; 5-µm packing L7
Flow rate:
1 mL/min
Injection size:
30 µL
System suitability
Sample:
Standard solution
Suitability requirements
Column efficiency:
NLT 2000 theoretical plates based on Olopatadine peak
Tailing factor:
NMT 2.0
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C21H23NO3·HCl in the portion of Olopatadine Hydrochloride taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response of the Sample solution |
| rS | = | = peak response of the Standard solution |
| CS | = | = concentration of USP Olopatadine Hydrochloride RS in the Standard solution (mg/mL) |
| CU | = | = nominal concentration of Olopatadine Hydrochloride in the Sample solution (mg/mL) |
Acceptance criteria:
98.0%–102.0% on the dried basis
IMPURITIES
Organic Impurities
• Procedure
[Note—Protect solutions from light. ]
Mobile phase:
Proceed as directed in the Assay.
Blank solution:
Mobile phase
System suitability solution:
0.2 mg/mL of USP Olopatadine Hydrochloride RS and 0.02 mg/mL of USP Olopatadine Related Compound B RS in Mobile phase
Sample solution:
0.2 mg/mL of Olopatadine Hydrochloride in Mobile phase
Chromatographic system
Mode:
LC
Detector:
UV 299 nm
Column:
4.6-mm × 15-cm; 5-µm packing L7
Flow rate:
1 mL/min
Injection size:
30 µL
Run time:
At least 2.5 times the retention time of the major peak
System suitability
Sample:
System suitability solution
[Note—The relative retention times for olopatadine and olopatadine related compound B are 1.0 and 1.2, respectively. ]
Suitability requirements
Resolution:
NLT 2.0 between olopatadine and olopatadine related compound B
Column efficiency:
NLT 2000 theoretical plates, olopatadine peak
Tailing factor:
NMT 2.0, olopatadine peak
Relative standard deviation:
NMT 2.0%, olopatadine peak
Analysis
Sample:
Sample solution
Calculate the percentage of each impurity in the portion of Olopatadine Hydrochloride taken:
Result = (rU/rT) × (1/F) × 100
| rU | = | = peak response of each individual impurity from the Sample solution |
| rT | = | = sum of all the peak responses from the Sample solution |
| F | = | = relative response factor for each individual impurity (see Impurity Table 1) |
[Note—Disregard any peaks corresponding to those of the Blank solution. ]
Acceptance criteria
Individual impurities:
See Impurity Table 1.
Total impurities:
NMT 0.25%
Impurity Table 1
| Name | Relative Retention Time |
Relative Response Factor |
Acceptance Criteria, NMT (%) |
|---|---|---|---|
| 0.4 | 1.0 | 0.2 | |
| Olopatadine E-isomerb | 0.7 | 1.3 | 0.1 |
| Olopatadine | 1.0 | — | — |
| Any other individual impurity | — | 1.0 | 0.1 |
|
a
(Z)-2-{11-[3-(Dimethylamino)propylidene]-6,11-dihydrodibenz[b,e]oxepin-2-yl}-2-hydroxyacetic acid.
b
11-[(E)-3-(Dimethylamino)propylidene]-6,11-dihydrodibenz[b,e]oxepin-2-acetic acid.
|
|||
SPECIFIC TESTS
• pH  791
791 :
Between 2.0 and 4.0, in a solution (1 in 100)
:
Between 2.0 and 4.0, in a solution (1 in 100)
• Loss on Drying  731
731 :
Dry a sample at 105
:
Dry a sample at 105 for 3 h: it loses NMT 0.3% of its weight.
for 3 h: it loses NMT 0.3% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight, light-resistant containers and store at room temperature.
• USP Reference Standards  11
11
USP Olopatadine Related Compound B RS
(Z)-3-{2-(Carboxymethyl)dibenzo[b,e]oxepin-11(6H)-ylidene}-N,N-dimethylpropan-1-amine oxide.
C21H23NO4 353.41
(Z)-3-{2-(Carboxymethyl)dibenzo[b,e]oxepin-11(6H)-ylidene}-N,N-dimethylpropan-1-amine oxide.
C21H23NO4 353.41
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Feiwen Mao, M.S.
Senior Scientific Liaison 1-301-816-8320 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4106
Pharmacopeial Forum: Volume No. 35(3) Page 567