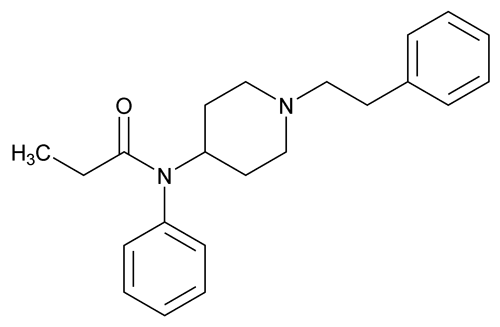
Fentanyl
C22H28N2O 336.47
Propanamide, N-phenyl-N-[1-(2-phenylethyl)-4-piperidinyl];
N-(1-Phenethylpiperidin-4-yl)-N-phenylpropionamide

 [437-38-7].
[437-38-7].
(fen' ta nil).
C22H28N2O 336.47
Propanamide, N-phenyl-N-[1-(2-phenylethyl)-4-piperidinyl];
N-(1-Phenethylpiperidin-4-yl)-N-phenylpropionamide
DEFINITION
Fentanyl contains NLT 98.0% and NMT 102.0% of the labeled amount of C22H28N2O, calculated on the dried basis.
[Caution—Great care should be taken to prevent inhaling particles of Fentanyl and exposing the skin to it.
]
IDENTIFICATION
• B.
The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
ASSAY
• Procedure
Solution A:
Add 3 mL of triethylamine to about 950 mL of water in a 1000-mL volumetric flask. Adjust with perchloric acid to a pH of 2.62 ± 0.02, and dilute with water to volume.
Solution B:
Acetonitrile
Diluent:
Solution A and Solution B (9:1)
Mobile phase:
See Table 1.
Table 1
| Time (min) |
Solution A (%) |
Solution B (%) |
|---|---|---|
| 0 | 90 | 10 |
| 2 | 90 | 10 |
| 3.5 | 85 | 15 |
| 5.1 | 82 | 18 |
| 7.6 | 72 | 28 |
| 11.5 | 63 | 37 |
| 15 | 40 | 60 |
| 19 | 40 | 60 |
| 20 | 90 | 10 |
| 25 | 90 | 10 |
System suitability stock solution:
0.4 mg/mL of USP Fentanyl Related Compound A RS, and about 0.1 mg/mL each of USP Fentanyl Related Compound B RS, USP Fentanyl Related Compound D RS, USP Fentanyl Related Compound E RS, and USP Fentanyl Related Compound G RS, prepared by dissolving in a suitable volumetric flask 50% filled with acetonitrile and diluting with water to volume
Standard stock solution:
1 mg/mL of USP Fentanyl RS prepared by dissolving in a suitable volumetric flask 40% filled with acetonitrile and diluting with water to volume
Standard solution:
0.1 mg/mL of USP Fentanyl RS from the Standard stock solution in Diluent
System suitability solution:
0.3 µg/mL each of USP Fentanyl Related Compound B RS, USP Fentanyl Related Compound D RS, USP Fentanyl Related Compound G RS, and USP Fentanyl Related Compound E RS; 1.3 µg/mL of USP Fentanyl Related Compound A RS; and 100 µg/mL of USP Fentanyl RS, prepared by diluting an appropriate amount of System suitability stock solution and Standard stock solution in Diluent
Sample stock solution:
Transfer 100 mg of Fentanyl to a 100-mL volumetric flask. Add 25 mL of acetonitrile, dissolve by shaking, and dilute with Diluent to volume.
Sample solution:
0.1 mg/mL of Fentanyl from the Sample stock solution in Diluent
Chromatographic system
Mode:
LC
Detector:
UV 206 nm
Column:
4.6-mm × 15-cm; 3.5-µm packing L7
Column temperature:
35
Flow rate:
1 mL/min
Injection size:
30 µL
System suitability
Sample:
System suitability solution
Suitability requirements
Tailing factor:
NLT 0.5 and NMT 2.0 for fentanyl and all fentanyl related compound peaks
Resolution:
NLT 1.2 between fentanyl and fentanyl related compound E peaks
Relative standard deviation:
NMT 0.7% for fentanyl and NMT 10% for all fentanyl related compounds
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of the labeled amount of fentanyl (C22H28N2O) in the portion of Fentanyl taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response of fentanyl from the Sample solution |
| rS | = | = peak response of fentanyl from the Standard solution |
| CS | = | = concentration of fentanyl in the Standard solution |
| CU | = | = concentration of Fentanyl in the Sample solution |
Acceptance criteria:
98.0%–102.0% on the dried basis
IMPURITIES
• Residue on Ignition  281
281 :
NMT 0.5%
:
NMT 0.5%
• Heavy Metals, Method II  231
231 :
NMT 20 ppm
:
NMT 20 ppm
• Organic Impurities
Solution A, Solution B, Diluent, System suitability solution, Sample stock solution, and Chromatographic system:
Proceed as directed in the Assay.
Standard stock solution:
1.2 µg/mL in Diluent prepared by dissolving 0.1 mg/mL of USP Fentanyl Related Compound E RS in a suitable volumetric flask 50% filled with acetonitrile and then diluting with water to volume. Dilute an aliquot of this solution with Diluent to achieve the desired concentration.
Standard solution:
0.024 µg/mL of USP Fentanyl Related Compound E RS from the Standard stock solution in Diluent
Sample solution:
250 µg/mL of Fentanyl from the Sample stock solution in Diluent
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of each individual impurity in the portion of Fentanyl taken:
Result = (rU/rS) × (CS/CU) × (1/F) × 100
| rU | = | = peak response of each individual impurity from the Sample solution |
| rS | = | = peak response of fentanyl related compound E from the Standard solution |
| CS | = | = concentration of USP Fentanyl Related Compound E RS in the Standard solution (µg/mL) |
| CU | = | = concentration of Fentanyl in the Sample solution (µg/mL) |
| F | = | = relative response factor of the fentanyl related compounds (see Table 2) |
Acceptance criteria:
See Table 2.
[Note—Identify fentanyl related compound C and fentanyl related compound F by using the relative retention times provided in Table 2. Identify the other impurity peaks in the sample by comparing them to those in the System suitability solution. ]
Table 2
| Name | Relative Retention Timea |
Relative Response Factorb |
Acceptance Criteria, NMT (%) |
|---|---|---|---|
| Fentanyl related compound Bc | 0.28 | 0.67 | 0.015 |
| Fentanyl related compound Cd | 0.56 | 0.67 | 0.25 |
| Fentanyl related compound De | 0.86 | 0.97 | 0.015 |
| Fentanyl related compound Gf | 0.89 | 0.82 | 0.25 |
| Fentanyl related compound Fg | 0.92 | 0.75 | 0.25 |
| Fentanyl related compound Eh | 0.98 | 1.0 | 0.015 |
| Fentanyl | 1.00 | — | — |
| Fentanyl related compound Ai | 1.26 | 0.55 | 0.25 |
| Individual unspecified impurities |
— | — | 0.10 |
| Total impurities | — | — | 0.5 |
|
a
The relative retention time is calculated based on fentanyl.
b
The relative response factor (RRF) is calculated based on fentanyl related compound E.
c
4-Anilinopiperidine.
d
N-Phenyl-N-(4-piperidinyl)propanamide.
e
N-Phenyl-1-(phenylmethyl)-4-piperidinamine.
f
N-Phenyl-N-[1-(2-phenylethyl)-4-piperidinyl]acetanilide hydrochloride, or acetyl fentanyl.
g
N-(1-Benzyl-4-piperidinyl)propionanilide.
h
N-Phenyl-1-(2-phenylethyl)-4-piperidinamine.
i
(2-Bromoethyl)benzene.
|
|||
(Organic Impurities postponed indefinitely)
SPECIFIC TESTS
• Loss on Drying  731
731 :
Dry a sample in a vacuum at 60
:
Dry a sample in a vacuum at 60 for 2 h: it loses NMT 0.5% of its weight.
for 2 h: it loses NMT 0.5% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tightly closed, light-resistant containers. Store at 25 , excursions permitted between 15
, excursions permitted between 15 and 30
and 30 .
.
• USP Reference Standards  11
11
USP Fentanyl Related Compound A RS
USP Fentanyl Related Compound B RS
USP Fentanyl Related Compound C RS
USP Fentanyl Related Compound D RS
USP Fentanyl Related Compound E RS
USP Fentanyl Related Compound F RS
USP Fentanyl Related Compound G RS
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Clydewyn M. Anthony, Ph.D.
Senior Scientific Liaison 1-301-816-8139 |
(SM22010) Monographs - Small Molecules 2 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3168
Pharmacopeial Forum: Volume No. 34(3) Page 602