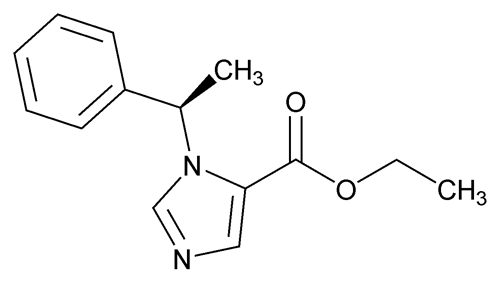
Etomidate
C14H16N2O2 244.29
1H-Imidazole-5-carboxylic acid, 1-(1-phenylethyl)-, ethyl ester, (+)-;
(+)-Ethyl 1-( -methylbenzyl)imidazole-5-carboxylate
-methylbenzyl)imidazole-5-carboxylate


 [33125-97-2].
[33125-97-2].
(e tom' i date).
C14H16N2O2 244.29
1H-Imidazole-5-carboxylic acid, 1-(1-phenylethyl)-, ethyl ester, (+)-;
(+)-Ethyl 1-(
DEFINITION
Etomidate contains NLT 98.0% and NMT 102.0% of C14H16N2O2, calculated on the dried basis.
IDENTIFICATION
• B.
The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
ASSAY
• Procedure
Buffer:
0.7 g/L of monobasic sodium phosphate in water
Mobile phase:
Acetonitrile and Buffer (2:3)
Standard solution:
0.16 mg/mL of USP Etomidate RS in acetonitrile
Sample solution:
0.16 mg/mL of Etomidate in acetonitrile
Chromatographic system
Mode:
LC
Detector:
UV 254 nm
Column:
3.9-mm × 30-cm; packing L1
Flow rate:
2.3 mL/min
Injection size:
20 µL
System suitability
Sample:
Standard solution
Suitability requirements
Tailing factor:
NMT 2.0
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C14H16N2O2 in the portion of Etomidate taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Etomidate RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Etomidate in the Sample solution (mg/mL) |
Acceptance criteria:
98.0%–102.0% on the dried basis
IMPURITIES
Organic Impurities
• Procedure
Solution A:
Dissolve 6 g of sodium citrate dihydrate and 4 g of anhydrous citric acid in 1 L of water.
Solution B:
Acetonitrile
Mobile phase:
See the gradient table below.
| Time (min) |
Solution A (%) |
Solution B (%) |
|---|---|---|
| 0 | 95 | 5 |
| 20 | 30 | 70 |
| 21 | 95 | 5 |
| 30 | 95 | 5 |
Diluent:
Weigh 6 g of sodium citrate dihydrate and 4 g of anhydrous citric acid into a 1000-mL volumetric flask. Add 500 mL of water, and shake to dissolve. Add 110 mL of acetonitrile and 50 mL of methanol, and dilute with water to volume.
Standard solution:
4 µg/mL of USP Metomidate Hydrochloride RS in Diluent
Sensitivity solution:
0.8 µg/mL of USP Metomidate Hydrochloride RS in Diluent, from the Standard solution
System suitability solution:
0.02 mg/mL of USP Etomidate RS and 0.02 mg/mL of USP Metomidate Hydrochloride RS in Diluent
Sample solution:
0.8 mg/mL of Etomidate in Diluent
Chromatographic system
Mode:
LC
Detector:
UV 254 nm
Column:
3.9-mm × 30-cm; packing L1
Flow rate:
2.0 mL/min
Injection size:
50 µL
System suitability
Samples:
Standard solution, Sensitivity solution, and System suitability solution
Suitability requirements
Resolution:
NLT 2.0 between etomidate and metomidate, System suitability solution
Signal-to-noise ratio:
NLT 10, Sensitivity solution
Relative standard deviation:
NMT 3.0%, Standard solution
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of each impurity in the portion of Etomidate taken:
Result = (rU/rS) × (CS/CU) × (Mr1/Mr2) × 100
| rU | = | = peak response of each individual impurity from the Sample solution |
| rS | = | = peak response of metomidate from the Standard solution |
| CS | = | = concentration of metomidate hydrochloride in the Standard solution (mg/mL) |
| CU | = | = concentration of Etomidate in the Sample solution (mg/mL) |
| Mr1 | = | = molecular weight of metomidate free base, 230.26 |
| Mr2 | = | = molecular weight of metomidate hydrochloride, 266.72 |
Acceptance criteria
Individual impurities:
See Impurity Table 1. [Note—Disregard any impurity peaks less than 0.05%. ]
SPECIFIC TESTS
• Loss on Drying, Method Ic  731
731 :
Dry 1 g of sample over phosphorus pentoxide for 16 h: it loses NMT 0.5% of its weight.
:
Dry 1 g of sample over phosphorus pentoxide for 16 h: it loses NMT 0.5% of its weight.
• Optical Rotation, Specific Rotation  781S
781S :
67.0
:
67.0 to 70.0
to 70.0 (t = 20
(t = 20 ), calculated on the dried basis
), calculated on the dried basis
Sample solution:
10 mg/mL in dehydrated alcohol
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in well closed, light-resistant containers, and store at room temperature.
• USP Reference Standards  11
11
USP Metomidate Hydrochloride RS
Methyl 1-(1-phenylethyl)-1H-imidazole-5-carboxylate hydrochloride.
C13H14N2O2·HCl 266.72
Methyl 1-(1-phenylethyl)-1H-imidazole-5-carboxylate hydrochloride.
C13H14N2O2·HCl 266.72
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3143
Pharmacopeial Forum: Volume No. 36(4) Page 918