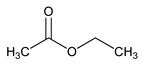
Ethyl Acetate
(eth' il as' e tate).
DEFINITION
Ethyl Acetate contains NLT 99.0% and NMT 100.5% of ethyl acetate (C4H8O2).
IDENTIFICATION
• A.
It is readily volatilized, even at low temperatures, and is flammable. When burned, a yellow flame and acetous odor are produced.
ASSAY
• Procedure
Sample:
Weigh 1.5 g of Ethyl Acetate in a stoppered weighing bottle.
Titrimetric system
(See Titrimetry  541
541 .)
.)
Mode:
Residual titration
Titrant:
0.5 N sodium hydroxide VS
Back-titrant:
0.5 N hydrochloric acid VS
Blank:
50 mL of 0.5 N sodium hydroxide VS, accurately measured
Endpoint detection:
Visual
Analysis:
Transfer the Sample to a suitable flask. Add 50.0 mL of 0.5 N sodium hydroxide VS, and heat on a steam bath under a reflux condenser for 1 h. Allow to cool, and add phenolphthalein TS. Titrate the excess sodium hydroxide with 0.5 N hydrochloric acid VS. Perform a blank determination.
Calculate the percentage of ethyl acetate (C4H8O2) in the Sample taken:
Result = {[(VB  VS) × N × F ]/W} × 100
VS) × N × F ]/W} × 100
| VB | = | = Back-titrant volume consumed by the Blank (mL) |
| VS | = | = Back-titrant volume consumed by the Sample (mL) |
| N | = | = actual normality of the Back-titrant (mEq/mL) |
| F | = | = equivalency factor for ethyl acetate, 88.1 mg/mEq |
| W | = | = Sample weight (mg) |
Acceptance criteria:
99.0%–100.5%
IMPURITIES
• Limit of Nonvolatile Residue
Sample:
Ethyl Acetate
Analysis:
Evaporate the Sample in a tared porcelain dish on a steam bath, and dry at 105 for 1 h.
for 1 h.
Acceptance criteria:
NMT 0.02%
• Limit of Methyl Compounds
Sample:
20 mL of Ethyl Acetate
Analysis:
Place the Sample in a 500-mL separator. Add a solution of 20 g of sodium hydroxide in 50 mL of water, and insert the stopper in the separator. Wrap it securely in a towel for protection against the heat of the reaction. Shake the mixture vigorously for about 5 min, cautiously opening the stopcock from time to time to permit the escape of air. Continue shaking vigorously until a homogeneous liquid results, then distill, and collect about 25 mL of the distillate. To 0.05 mL of the distillate add 1 drop of dilute phosphoric acid (1 in 20) and 1 drop of potassium permanganate solution (1 in 20). Mix, allow to stand for 1 min, and add sodium bisulfite solution (1 in 20), dropwise, until the permanganate color is discharged. If a brown color remains, add 1 drop of the dilute phosphoric acid. To the colorless solution add 5 mL of freshly prepared chromotropic acid TS, and heat on a steam bath at 60 for 10 min.
for 10 min.
Acceptance criteria:
No violet color appears.
• Chromatographic Purity
System suitability solution:
Chloroform, ethyl acetate, isobutyl acetate, and n-butyl acetate (3:1:1:1)
Chromatographic system
Mode:
GC
Detector:
Flame ionization
Column:
1.8-m × 4-mm; support S11
Column temperature:
See Table 1.
Table 1
| Initial Temperature ( |
Ramp ( |
Final Temperature ( |
Hold Time at Final Temperature (min) |
|---|---|---|---|
| 115 | — | 115 | 6 |
| 115 | 16 | 200 | 15 |
System suitability
Sample:
0.1 µL of System suitability solution
[Note—The retention times for chloroform, isobutyl acetate, and n-butyl are 0.9, 2.7, and 2.8, respectively, relative to ethyl acetate as 1.0. ]
Suitability requirements
Resolution:
NLT 1.3 between the chloroform and ethyl acetate peaks; NLT 1.5 between the isobutyl acetate and n-butyl acetate peaks
Tailing factor:
NMT 1.5 for the ethyl acetate peak
Analysis
Sample:
Ethyl Acetate (neat)
Inject about 0.06 µL using a 1-µL syringe.
Acceptance criteria:
The area of the ethyl acetate peak is NLT 99.5% of the sum of the areas of all the peaks.
SPECIFIC TESTS
• Acidity
Sample solution:
2.0 mL of Ethyl Acetate in 10 mL of neutralized alcohol
Analysis:
Add 2 drops of phenolphthalein TS to the Sample solution. Neutralize with 0.10 N sodium hydroxide.
Acceptance criteria:
NMT 0.10 mL of 0.10 N sodium hydroxide is required.
• Readily Carbonizable Substances Test  271
271
Sample:
2 mL of Ethyl Acetate
Analysis:
Add the Sample carefully to 10 mL of sulfuric acid to form separate layers.
Acceptance criteria:
No dark zone is developed within 15 min.
• Specific Gravity  841
841 :
0.894–0.898
:
0.894–0.898
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers, and avoid exposure to excessive heat.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
USP35–NF30 Page 1792
Pharmacopeial Forum: Volume No. 34(5) Page 1223