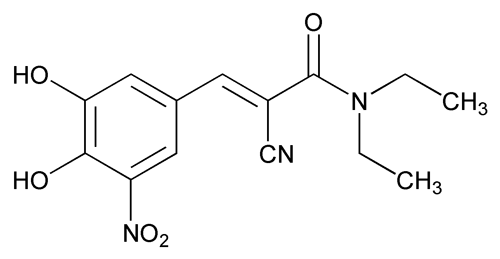
Entacapone
C14H15N3O5 305.29
(E)- -Cyano-N,N-diethyl-3,4-dihydroxy-5-nitrocinnamamide;
-Cyano-N,N-diethyl-3,4-dihydroxy-5-nitrocinnamamide;
2-Propenamide, 2-cyano-3-(3,4-dihydroxy-5-nitrophenyl)-N,N-diethyl-,(E)-

 [130929-57-6].
[130929-57-6].
(en tak' a pone).
C14H15N3O5 305.29
(E)-
2-Propenamide, 2-cyano-3-(3,4-dihydroxy-5-nitrophenyl)-N,N-diethyl-,(E)-
DEFINITION
Entacapone contains NLT 98.0% and NMT 102.0% of C14H15N3O5, calculated on the dried basis.
IDENTIFICATION
• B.
The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
ASSAY
• Procedure
Buffer:
2.34 g/L of monobasic sodium phosphate dihydrate. Adjust with phosphoric acid to a pH of 2.1.
Diluent:
Methanol and tetrahydrofuran (7:3)
Mobile phase:
Methanol, tetrahydrofuran, and Buffer (22:1:27)
System suitability solution:
1 µg/mL each of USP Entacapone Related Compound A RS and USP Entacapone RS in Diluent
Standard solution:
0.1 mg/mL of USP Entacapone RS in Diluent
Sample solution:
0.1 mg/mL of Entacapone in Diluent
Chromatographic system
Mode:
LC
Detector:
UV 300 nm
Column:
4.6-mm × 25-cm; 5-µm packing L11
Flow rate:
1 mL/min
Injection size:
10 µL
System suitability
Samples:
System suitability solution and Standard solution
[Note—The relative retention times for entacapone related compound A and entacapone are 0.8 and 1.0, respectively. ]
Suitability requirements
Resolution:
NLT 2.0 between entacapone related compound A and entacapone, System suitability solution
Tailing factor:
NMT 2.0, Standard solution
Relative standard deviation:
NMT 1.0%, Standard solution
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C14H15N3O5 in the portion of Entacapone taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Entacapone RS in the Standard solution (mg/mL) |
| CU | = | = concentration of entacapone in the Sample solution (mg/mL) |
Acceptance criteria:
98.0%–102.0% on the dried basis
IMPURITIES
Organic Impurities
• Procedure
Buffer, Diluent, and Mobile phase:
Proceed as directed in the Assay.
Standard solution:
Use the System suitability solution prepared as directed in the Assay.
Sample solution:
1.0 mg/mL of Entacapone in Diluent
Chromatographic system:
Prepare as directed in the Assay.
Run time:
2 times the retention time of the entacapone peak
System suitability
Sample:
Standard solution
Suitability requirements
Resolution:
NLT 2.0 between entacapone related compound A and entacapone
Relative standard deviation:
NMT 10.0% based on the entacapone peak
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of any individual impurity in the portion of Entacapone taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response of any impurity from the Sample solution |
| rS | = | = peak response of entacapone from the Standard solution |
| CS | = | = concentration of USP Entacapone RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Entacapone in the Sample solution (mg/mL) |
Acceptance criteria
Individual impurities:
See Impurity Table 1.
Total impurities:
NMT 0.2%
[Note—Do not include entacapone related compound A in the calculation of total impurities. ]
Impurity Table 1
| Name | Relative Retention Time |
Acceptance Criteria, NMT (%) |
|
|---|---|---|---|
| Entacapone related compound Aa | 0.8 | 0.1 | |
| Entacapone | 1.0 | — | |
| Any other unknown individual impurity | — | 0.10 | |
| Total impurities | — | 0.2 | |
|
a
(Z)-2-Cyano-3-(3,4-dihydroxy-5-nitrophenyl)-N,N-diethylacrylamide.
|
|||
SPECIFIC TESTS
• Loss on Drying  731
731
Analysis:
Dry 1.0 g of the sample in vacuum at a pressure not exceeding 49 mm of mercury at 65 to constant weight.
to constant weight.
Acceptance criteria:
NMT 0.5%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers, and store at room temperature.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3047
Pharmacopeial Forum: Volume No. 36(2) Page 396