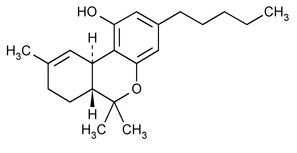
Dronabinol
(droe nab' i nol).
6H-Dibenzo[b,d]pyran-1-ol, 6a,7,8,10a-tetrahydro-6,6,9-trimethyl-3-pentyl-, (6aR-trans)-.
(6aR,10aR)-6a,7,8,10a-Tetrahydro-6,6,9-trimethyl-3-pentyl-6H-dibenzo[b,d]-pyran-1-ol
» Dronabinol is D9-tetrahydrocannabinol. It contains not less than 95.0 percent of C21H30O2.
Packaging and storage—
Preserve in tight, light-resistant glass containers in inert atmosphere. Store as per labeling instructions.
Identification—
A:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of the Standard preparation, as obtained in the Assay.
B:
Visualizing agent—Transfer about 100 mg of Fast Blue B salt to a suitable flask containing about 100 mL of methanol, stir for about 5 minutes, and allow to settle. Decant the clear liquid into the sprayer reservoir. [note—Prepare fresh daily. ]
Identification solution—
Use the Standard preparation, prepared as directed in the Assay.
Test solution—
Use the Assay preparation.
Procedure—
Apply separately 10 µL each of the Identification solution and the Test solution to a suitable thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of chromatographic silica gel. Allow the spots to dry, and develop the plate in a chromatographic chamber that has been equilibrated (for about 2 minutes) with vapors from a solvent mixture of n-hexane and methylene chloride (1:1) until the solvent front has moved about 10 cm. Remove the plate from the developing chamber, quickly mark the solvent front, and allow the plate to dry at room temperature for about 5 minutes. Spray the plate with the Visualizing agent until it is uniformly damp (not saturated). Heat the plate at about 80
) coated with a 0.25-mm layer of chromatographic silica gel. Allow the spots to dry, and develop the plate in a chromatographic chamber that has been equilibrated (for about 2 minutes) with vapors from a solvent mixture of n-hexane and methylene chloride (1:1) until the solvent front has moved about 10 cm. Remove the plate from the developing chamber, quickly mark the solvent front, and allow the plate to dry at room temperature for about 5 minutes. Spray the plate with the Visualizing agent until it is uniformly damp (not saturated). Heat the plate at about 80 until the spots are developed: the color and RF value of the spots from the Test solution correspond to those obtained from the Identification solution.
until the spots are developed: the color and RF value of the spots from the Test solution correspond to those obtained from the Identification solution.
Related compounds—
[note—Minimize exposure to air and light for the Standard solution and the Test solution. Analyze all samples within 24 hours. ]
Mobile phase, System suitability solution, and Standard preparation—
Proceed as directed in the Assay.
Standard solution—
Dilute an accurately measured volume of the Standard preparation quantitatively, and stepwise if necessary, with dehydrated alcohol to obtain a solution having a known concentration of about 0.004 mg per mL.
Sensitivity solution—
Quantitatively dilute an accurately measured volume of the Standard solution with dehydrated alcohol to obtain a solution having a concentration of about 0.2 µg per mL.
Test solution—
Use the Assay preparation.
Chromatographic system—
Proceed as directed in the Assay. In addition, chromatograph the Sensitivity solution, and calculate the signal-to-noise ratio, S/N, by the formula:
(2H)/h
in which H is the measured height of the peak, and h is the amplitude of the average measured baseline noise; the signal-to-noise ratio is not less than 10.
Procedure—
Separately inject equal volumes (about 10 µL) of the Standard solution and the Test solution into the chromatograph, record the chromatograms, and measure the responses for all of the peaks.
Table 1
| Name | Relative Retention Time |
Relative Response Factor |
Limit (%) |
|---|---|---|---|
| Cannabinol | 0.78 | 2.7 | 1.5 |
| D9-Tetrahydrocannabinol | 1.00 | 1.0 | — |
| Exo-tetrahydrocannabinol1 | 1.07 | 0.92 | 0.5 |
| D8-Tetrahydrocannabinol | 1.18 | 0.90 | 2.0 |
| Any other individual impurity | — | 1.0 | 1.0 |
|
1
(6aR, 10aR)-6,6-Dimethyl-9-methylene-3-pentyl-6a,7,8,9,10,10a-hexahydro-6H-benzo[c]chromen-1-ol.
|
|||
Calculate the percentage of each impurity in the portion of Dronabinol taken by the formula:
100(1/F)(CV/W)(rU / rS)
in which F is the relative response factor for each impurity (see Table 1); C is the concentration, in mg per mL, of D9-tetrahydrocannabinol in the Standard solution; V is the volume, in mL, of the Test solution; W is the weight, in mg, of Dronabinol taken to prepare the Test solution; rU is the peak area response of each impurity in the Test solution; and rS is the peak area response of D9-tetrahydrocannabinol in the Standard solution. In addition to not exceeding the limits in Table 1, not more than 5.0% of total impurities is found.
Assay—
[note—Minimize exposure to air and light for the Standard preparation and the Assay preparation. Analyze all samples within 24 hours. ]
Mobile phase—
Prepare a filtered and degassed mixture of methanol, water, tetrahydrofuran, and acetonitrile (45:25:20:10), making adjustments, if necessary (see System Suitability under Chromatography  621
621 ).
).
System suitability solution—
Transfer accurately measured volumes of USP D9-Tetrahydrocannabinol RS and USP Exo-tetrahydrocannabinol RS to a suitable volumetric flask, and dilute with dehydrated alcohol to prepare a solution that contains about 200 µg of D9-tetrahydrocannabinol and about 10 µg of exo-tetrahydrocannabinol per mL.
Standard preparation—
Quantitatively dilute an accurately measured volume of USP D9-Tetrahydrocannabinol RS with dehydrated alcohol to obtain a solution having a known concentration of about 0.2 mg per mL.
Assay preparation—
Transfer about 20 mg of Dronabinol, accurately weighed, to a 100-mL volumetric flask, dissolve in and dilute with dehydrated alcohol to volume, and mix.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 228-nm detector and a 4.6-mm × 15-cm analytical column that contains 4-µm packing L1. The flow rate is about 1 mL per minute. The column temperature is maintained at 20
)—
The liquid chromatograph is equipped with a 228-nm detector and a 4.6-mm × 15-cm analytical column that contains 4-µm packing L1. The flow rate is about 1 mL per minute. The column temperature is maintained at 20 . Chromatograph the System suitability solution, and record the peak responses as directed for Procedure: the resolution, R, between D9-tetrahydrocannabinol and exo-tetrahydrocannabinol is not less than 1.5; and the tailing factor of D9-tetrahydrocannabinol is not more than 2.0. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the relative standard deviation for replicate injections is not more than 2.0%.
. Chromatograph the System suitability solution, and record the peak responses as directed for Procedure: the resolution, R, between D9-tetrahydrocannabinol and exo-tetrahydrocannabinol is not less than 1.5; and the tailing factor of D9-tetrahydrocannabinol is not more than 2.0. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 10 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for all of the peaks. Calculate the quantity, in mg, of C21H30O2 in the portion of Dronabinol taken by the formula:
CV(rU / rS)
in which C is the concentration, in mg per mL, of D9-tetrahydrocannabinol in the Standard preparation; V is the volume, in mL, of the Assay preparation; and rU and rS are the D9-tetrahydrocannabinol peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Elena Gonikberg, Ph.D.
Principal Scientific Liaison 1-301-816-8251 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2991
Pharmacopeial Forum: Volume No. 34(1) Page 90