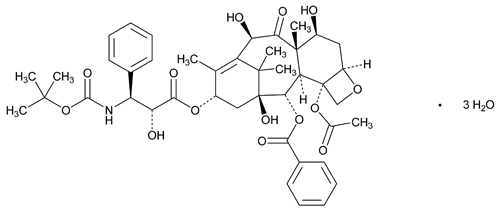
Docetaxel
C43H53NO14·3H2O 861.93
Anhydrous 807.88
Benzenepropanoic acid, -[[(1,1-dimethylethoxy)carbonyl]amino]-
-[[(1,1-dimethylethoxy)carbonyl]amino]- -hydroxy-, 12b-(acetyloxy)-12-(benzoyloxy)-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-4,6,11-trihydroxy-4a,8,13,13-tetramethyl-5-oxo-7,11-methano-1H-cyclodeca[3,4]benz[1,2-b]oxet-9-yl ester trihydrate, [2aR-[2a
-hydroxy-, 12b-(acetyloxy)-12-(benzoyloxy)-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-4,6,11-trihydroxy-4a,8,13,13-tetramethyl-5-oxo-7,11-methano-1H-cyclodeca[3,4]benz[1,2-b]oxet-9-yl ester trihydrate, [2aR-[2a ,4
,4 ,4a
,4a ,6
,6 ,9
,9 (
( R*,
R*, S*),11
S*),11 ,12
,12 ,12a
,12a ,12b
,12b ]]-;(2R,3S)-N-Carboxy-3-phenylisoserine, N-tert-butyl ester, 13-ester with 5
]]-;(2R,3S)-N-Carboxy-3-phenylisoserine, N-tert-butyl ester, 13-ester with 5 ,20-epoxy-1,2
,20-epoxy-1,2 ,4,7
,4,7 ,10
,10 ,13
,13 -hexahydroxytax-11-en-9-one 4-acetate 2-benzoate, trihydrate
-hexahydroxytax-11-en-9-one 4-acetate 2-benzoate, trihydrate


 [148408-66-6].
[148408-66-6].
(doe'' se tax' el).
C43H53NO14·3H2O 861.93
Anhydrous 807.88
Benzenepropanoic acid,
DEFINITION
Docetaxel contains NLT 97.5% and NMT 102.0% of C43H53NO14, calculated on the anhydrous and solvent-free basis. [Caution—Docetaxel is cytotoxic. Great care should be taken to prevent inhaling particles of Docetaxel and exposing the skin to it.
]
IDENTIFICATION
• A. Infrared Absorption  197
197
[Note—Methods described under Infrared Absorption  197K
197K ,
,  197M
197M , or
, or  197S
197S may be used. Use solution containing 60 mg/mL of Docetaxel in methylene chloride for
may be used. Use solution containing 60 mg/mL of Docetaxel in methylene chloride for  197S
197S . ]
. ]
• B.
The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
ASSAY
• Procedure
Solution A:
Water
Solution B:
Acetonitrile
Mobile phase:
See the gradient table below.
| Time (min) |
Solution A (%) |
Solution B (%) |
|---|---|---|
| 0 | 72 | 28 |
| 9.0 | 72 | 28 |
| 39.0 | 28 | 72 |
| 39.1 | 72 | 28 |
| 50 | 72 | 28 |
Diluent:
Acetonitrile, water, and acetic acid (100:100:0.1)
Standard solution:
1.0 mg/mL made by transferring a quantity of USP Docetaxel RS to a suitable volumetric flask, dissolving in alcohol, equivalent to about 5% of the final volume, and diluting with Diluent to volume
System suitability solution:
1 mg/mL of USP Docetaxel Identification RS in Diluent. [Note—USP Docetaxel Identification RS contains docetaxel and a small amount of 2-debenzoxyl 2-pentenoyl docetaxel, 6-oxodocetaxel, 4-epidocetaxel, and 4-epi-6-oxodocetaxel. See Impurity Table 1. ]
Sample solution:
1.0 mg/mL made by transferring a quantity of Docetaxel to a suitable volumetric flask, dissolving in alcohol, equivalent to about 5% of the final volume, and diluting with Diluent to volume
Chromatographic system
Mode:
LC
Detector:
UV 232 nm
Refrigerated autosampler temperature:
10
Column:
4.6-mm × 15-cm; 3.5-µm packing L1
Column temperature:
45
Flow rate:
1.2 mL/min
Injection size:
10 µL
System suitability
Samples:
Standard solution and System suitability solution
Suitability requirements
Resolution:
NLT 4 between 2-debenzoxyl 2-pentenoyl docetaxel and docetaxel, System suitability solution
Relative standard deviation:
NMT 1.0%, Standard solution
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C43H53NO14 in the portion of Docetaxel taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of docetaxel in the Standard solution (mg/mL) |
| CU | = | = concentration of Docetaxel in the Sample solution (mg/mL) |
Acceptance criteria:
97.5%–102.0% on the anhydrous and solvent-free basis
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.1%
:
NMT 0.1%
• Heavy Metals, Method I  231
231
Sample solution:
Dissolve 1 g in 20 mL of a mixture of dimethylformamide and water (17:3). To 12 mL of this solution, add 2 mL of pH 3.5 Acetate Buffer and mix. Add 1.2 mL of thioacetamide–glycerin base TS and mix.
Acceptance criteria:
NMT 20 ppm
Organic Impurities
• Procedure
Standard solution, System suitability solution, Sample solution, and Chromatographic system:
Proceed as directed in the Assay.
Sensitivity solution:
0.5 µg/mL of USP Docetaxel RS in Diluent, from the Standard solution
System suitability
Samples:
System suitability solution and Sensitivity solution
Suitability requirements
Resolution:
NLT 4 between 2-debenzoxyl 2-pentenoyl docetaxel and docetaxel, System suitability solution
Signal-to-noise ratio:
NLT 10 for the docetaxel peak, Sensitivity solution
Analysis
Sample:
Sample solution
Calculate the percentage of each impurity in the portion of Docetaxel taken:
Result = (rU/rT) × (1/F) × 100
| rU | = | = peak response of each individual impurity from the Sample solution |
| rT | = | = sum of the responses of all peaks from the Sample solution |
| F | = | = relative response factor for each individual impurity (see Impurity Table 1) |
Acceptance criteria
Individual impurities:
See Impurity Table 1. [Note—Disregard any impurity peaks less than 0.05%. ]
Total impurities:
NMT 1.0%
Impurity Table 1
| Name | Relative Retention Time |
Relative Response Factor |
Acceptance Criteria, NMT (%) |
|---|---|---|---|
| 2-Debenzoxyl 2-pentenoyl docetaxela | 0.97 | 0.63 | 0.5 |
| Docetaxel | 1.00 | — | — |
| 6-Oxodocetaxelb | 1.08 | 1.0 | 0.3 |
| 4-Epidocetaxelc | 1.13 | 1.0 | 0.3 |
| 4-Epi-6-oxodocetaxeld | 1.18 | 1.0 | 0.2 |
| Any unspecified impurity |
— | 1.0 | 0.10 |
|
a
(2aR,4S,4aS,6R,9S,11S,12S,12aR,12bS)-1,2a,3,4,4a,6,9,10,11,12,12a,12b-Dodecahydro-4,6,9,11,12,12b-hexahydroxy-4a,8,13,13-tetramethyl-7,11-methano-5H-cyclodeca[3,4]benz[1,2-b]oxet-5-one 12b-acetate, 12-[(E)-2-methylbut-2-enoate], 9-ester with (2R,3S)-N-tert-butoxycarbonyl-3-phenylisoserine.
b
(2aR,4S,4aS,9S,11S,12S,12aR,12bS)-1,2a,3,4,4a,6,9,10,11,12,12a,12b-Dodecahydro-4, 9,11,12,12b-pentahydroxy-4a,8,13,13-tetramethyl-7,11-methano-5H-cyclodeca[3,4]benz[1,2-b]oxet-5,6-dione 12b-acetate, 12-benzoate, 9-ester with (2R,3S)-N-tert-butoxycarbonyl-3-phenylisoserine.
c
(2aR,4R,4aS,6R,9S,11S,12S,12aR,12bS)-1,2a,3,4,4a,6,9,10,11,12,12a,12b-Dodecahydro-4,6,9,11,12,12b-hexahydroxy-4a,8,13,13-tetramethyl-7,11-methano-5H-cyclodeca[3,4]benz[1,2-b]oxet-5-one 12b-acetate, 12-benzoate, 9-ester with (2R,3S)-N-tert-butoxycarbonyl-3-phenylisoserine.
d
(2aR,4R,4aS,9S,11S,12S,12aR,12bS)-1,2a,3,4,4a,6,9,10,11,12,12a,12b-Dodecahydro-4, 9,11,12,12b-pentahydroxy-4a,8,13,13-tetramethyl-7,11-methano-5H-cyclodeca[3,4]benz[1,2-b]oxet-5,6-dione 12b-acetate, 12-benzoate, 9-ester with (2R,3S)-N-tert-butoxycarbonyl-3-phenylisoserine.
|
|||
SPECIFIC TESTS
• Microbial Enumeration Tests  61
61 and Tests for Specified Microorganisms
and Tests for Specified Microorganisms  62
62 :
The total aerobic microbial limit does not exceed 100 cfu/g. The total yeast and mold count does not exceed 10 cfu/g.
:
The total aerobic microbial limit does not exceed 100 cfu/g. The total yeast and mold count does not exceed 10 cfu/g.
• Bacterial Endotoxins Test  85
85 :
It contains NMT 0.3 USP Endotoxin Units/mg.
:
It contains NMT 0.3 USP Endotoxin Units/mg.
• Water Determination, Method Ic  921
921 :
5.0%–7.0%
:
5.0%–7.0%
• Optical Rotation, Specific Rotation  781S
781S :
:
 39
39 to
to  41
41 (t=20
(t=20 ), calculated on the anhydrous and solvent-free basis.
), calculated on the anhydrous and solvent-free basis.
Sample solution:
10 mg/mL in methanol
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in well closed, light-resistant containers, and store at room temperature.
• USP Reference Standards  11
11
USP Docetaxel Identification RS  Contains docetaxel and small amount of 2-debenzoxyl 2-pentenoyl docetaxel, 6-oxodocetaxel, 4-epidocetaxel, and 4-epi-6-oxodocetaxel.
Contains docetaxel and small amount of 2-debenzoxyl 2-pentenoyl docetaxel, 6-oxodocetaxel, 4-epidocetaxel, and 4-epi-6-oxodocetaxel.
 Contains docetaxel and small amount of 2-debenzoxyl 2-pentenoyl docetaxel, 6-oxodocetaxel, 4-epidocetaxel, and 4-epi-6-oxodocetaxel.
Contains docetaxel and small amount of 2-debenzoxyl 2-pentenoyl docetaxel, 6-oxodocetaxel, 4-epidocetaxel, and 4-epi-6-oxodocetaxel.
USP Endotoxin RS
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Feiwen Mao, M.S.
Senior Scientific Liaison 1-301-816-8320 |
(SM32010) Monographs - Small Molecules 3 |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2953
Pharmacopeial Forum: Volume No. 35(5) Page 1133