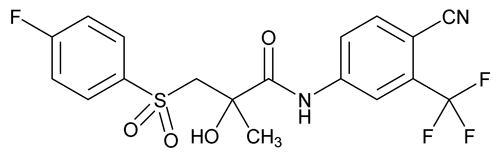
Bicalutamide
C18H14F4N2O4S 430.37
Propanamide, N-[4-cyano-3-(trifluoromethyl)phenyl]-3-[(4-fluorophenyl)sulfonyl]-2-hydroxy-2-methyl-, (±)-;
(±)-4¢-Cyano- ,
, ,
, -trifluoro-3-[(p-fluorophenyl)sulfonyl]-2-methyl-m-lactotoluidide
-trifluoro-3-[(p-fluorophenyl)sulfonyl]-2-methyl-m-lactotoluidide


 [90357-06-5].
[90357-06-5].
(bye'' ka loo' ta mide).
C18H14F4N2O4S 430.37
Propanamide, N-[4-cyano-3-(trifluoromethyl)phenyl]-3-[(4-fluorophenyl)sulfonyl]-2-hydroxy-2-methyl-, (±)-;
(±)-4¢-Cyano-
DEFINITION
Bicalutamide contains NLT 98.0% and NMT 102.0% of C18H14F4N2O4S, calculated on the anhydrous and solvent-free basis.
IDENTIFICATION
• B.
The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
ASSAY
• Procedure
Solution A:
0.01% (v/v) of trifluoroacetic acid in water
Solution B:
0.01% (v/v) of trifluoroacetic acid in acetonitrile
Mobile phase:
Solution A and Solution B (52:48)
Diluent:
Solution A and Solution B (1:2)
System suitability solution:
5 µg/mL of USP Bicalutamide Related Compound A RS and 50 µg/mL of USP Bicalutamide RS in Diluent
Standard solution:
0.05 mg/mL of USP Bicalutamide RS in Diluent
Sample solution:
0.05 mg/mL of Bicalutamide in Diluent
Chromatographic system
Mode:
LC
Detector:
UV 270 nm
Column:
4.0-mm × 10-cm; 3-µm packing L1
Flow rate:
1 mL/min
Injection size:
10 µL
System suitability
Samples:
System suitability solution and Standard solution
[Note—The relative retention times for bicalutamide related compound A isomer A and bicalutamide related compound A isomer B are 0.75 and 0.78, respectively. ]
Suitability requirements
Resolution:
NLT 2.0 between bicalutamide related compound A isomer B and bicalutamide, System suitability solution
Relative standard deviation:
NMT 2%, Standard solution
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C18H14F4N2O4S in the portion of Bicalutamide taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Bicalutamide RS in the Standard solution (mg/mL) |
| CU | = | = concentration of the Sample solution (mg/mL) |
Acceptance criteria:
98.0%–102.0% on the anhydrous and solvent-free basis
IMPURITIES
Organic Impurities
• Procedure
Solution A, Solution B, Diluent, System suitability solution, and Chromatographic system:
Proceed as directed in the Assay.
Mobile phase:
See the gradient table below.
| Time (min) |
Solution A (%) |
Solution B (%) |
|---|---|---|
| 0 | 67 | 33 |
| 16.5 | 67 | 33 |
| 26.5 | 40 | 60 |
| 32.5 | 5 | 95 |
| 32.6 | 67 | 33 |
| 35 | 67 | 33 |
Standard solution:
1 µg/mL of USP Bicalutamide RS in Diluent
Sample solution:
1 mg/mL of Bicalutamide in Diluent
System suitability
Sample:
System suitability solution
Suitability requirements
Resolution 1:
NLT 0.8 between bicalutamide related compound A isomer A and bicalutamide related compound A isomer B
Resolution 2:
NLT 8.5 between bicalutamide related compound A isomer B and bicalutamide
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of each impurity in the portion of Bicalutamide taken:
Result = (rU/rS) × (CS/CU) × (1/F) × 100
| rU | = | = peak area of each impurity from the Sample solution |
| rS | = | = peak area of bicalutamide from the Standard solution |
| CS | = | = concentration of bicalutamide in the Standard solution (mg/mL) |
| CU | = | = concentration of Bicalutamide in the Sample solution (mg/mL) |
| F | = | = relative response factor (see Impurity Table 1) |
Acceptance criteria
Individual impurities:
See Impurity Table 1.
Total impurities:
NMT 0.5%
Impurity Table 1
| Name | Relative Retention Time |
Relative Response Factor |
Acceptance Criteria, NMT (%) |
|---|---|---|---|
| Bicalutamide aminobenzonitrilea | 0.30 | 1.4 | 0.1 |
| Bicalutamide related compound A isomer Ab | 0.64 | 1.0 | 0.1 |
| Bicalutamide related compound A isomer Bb | 0.67 | 1.0 | 0.1 |
| Desfluoro bicalutamidec | 0.83 | 1.1 | 0.2 |
| 2-Fluoro bicalutamided | 0.94 | 1.0 | 0.2 |
| Bicalutamide | 1.00 | — | — |
| Deoxybicalutamidee | 1.33 | 1.0 | 0.2 |
| Bicalutamide sulfidef | 1.56 | 1.0 | 0.1 |
| Any unspecified impurity | — | 1.0 | 0.1 |
|
a
4-Amino-2-(trifluoromethyl)benzonitrile.
b
N-[4-Cyano-3-(trifluoromethyl)phenyl]-3-[(4-fluorophenyl)sulfinyl]-2-hydroxy-2-methylpropanamide.
c
N-[4-Cyano-3-(trifluoromethyl)phenyl]-2-hydroxy-2-methyl-3-
(phenylsulfonyl)propanamide.
d
N-[4-Cyano-3-(trifluoromethyl)phenyl]-3-(2-fluorophenylsulfonyl)-2-hydroxy-2-methylpropanamide.
e
N-[4-Cyano-3-(trifluoromethyl)phenyl]-3-(4-fluorophenylsulfonyl)-2-methylpropanamide.
f
N-[4-Cyano-3-(trifluoromethyl)phenyl]-3-(4-fluorophenylthio)-2-hydroxy-2-methylpropanamide.
|
|||
SPECIFIC TESTS
• Water Determination, Method I  921
921 :
NMT 0.2%
:
NMT 0.2%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers, and store at room temperature.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Feiwen Mao, M.S.
Senior Scientific Liaison 1-301-816-8320 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2354
Pharmacopeial Forum: Volume No. 36(5) Page 1165