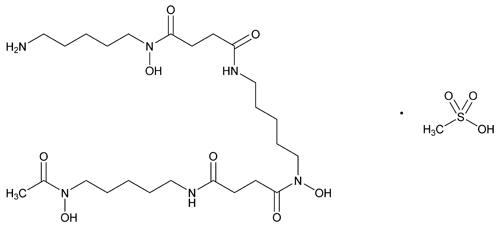
Deferoxamine Mesylate
C25H48N6O8·CH4O3S 656.79
Butanediamide, N¢-[5-[[4-[[5-(acetylhydroxyamino)pentyl]amino]-1,4-dioxobutyl]hydroxyamino]pentyl]-N-(5-aminopentyl)-N-hydroxy-, monomethanesulfonate;
N-[5-[3-[(5-Aminopentyl)hydroxycarbamoyl]propionamido]pentyl]-3-[[5-(N-hydroxyacetamido)pentyl]carbamoyl]propionohydroxamic acid monomethanesulfonate (salt)

 [138-14-7].
[138-14-7].
(de fer ox' a meen mes' i late).
C25H48N6O8·CH4O3S 656.79
Butanediamide, N¢-[5-[[4-[[5-(acetylhydroxyamino)pentyl]amino]-1,4-dioxobutyl]hydroxyamino]pentyl]-N-(5-aminopentyl)-N-hydroxy-, monomethanesulfonate;
N-[5-[3-[(5-Aminopentyl)hydroxycarbamoyl]propionamido]pentyl]-3-[[5-(N-hydroxyacetamido)pentyl]carbamoyl]propionohydroxamic acid monomethanesulfonate (salt)
DEFINITION
Change to read:
Deferoxamine Mesylate contains NLT  93.0%
93.0% (RB 1-May-2011) and NMT 102.0% of C25H48N6O8·CH4O3S, calculated on the anhydrous basis.
(RB 1-May-2011) and NMT 102.0% of C25H48N6O8·CH4O3S, calculated on the anhydrous basis.
IDENTIFICATION
• B.
The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
ASSAY
Change to read:
• Procedure
Solution A:
1.32 g/L of dibasic ammonium phosphate in water. Adjust with phosphoric acid to a pH of 3.0.
Solution B:
Acetonitrile and Solution A (1:1)
Mobile phase:
See Table 1.
Table 1
| Time (min) |
Solution A (%) |
Solution B (%) |
|---|---|---|
| 0 | 88 | 12 |
| 20 | 80 | 20 |
| 35 | 57.5 | 42.5 |
| 35.1 | 88 | 12 |
| 40 | 88 | 12 |
Diluent:
Acetonitrile and water (6:94)
Standard solution:
1.0 mg/mL of USP Deferoxamine Mesylate RS in Diluent
Sample solution:
1.0 mg/mL of Deferoxamine Mesylate in Diluent
Chromatographic system
Mode:
LC
Detector:
UV 220 nm
Column:
4.6-mm × 7.5-cm; 3.5-µm packing L1
Column temperature:
32
Autosampler temperature:
5
Flow rate:
1.5 mL/min
Injection size:
20 µL
System suitability
Sample:
Standard solution
Suitability requirements
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of deferoxamine mesylate (C25H48N6O8·CH4O3S) in the portion of Deferoxamine Mesylate taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response of deferoxamine from the Sample solution |
| rS | = | = peak response of deferoxamine from the Standard solution |
| CS | = | = concentration of the Standard solution (mg/mL) |
| CU | = | = concentration of the Sample solution (mg/mL) |
Acceptance criteria:
 93.0%
93.0% (RB 1-May-2011)–102.0% on the anhydrous basis
(RB 1-May-2011)–102.0% on the anhydrous basis
IMPURITIES
• Residue on Ignition  281
281 :
NMT 0.1%, 2.0 g being used
:
NMT 0.1%, 2.0 g being used
• Chloride and Sulfate, Chloride  221
221 :
A 1.2-g portion shows no more chloride than corresponds to 0.20 mL of 0.020 N hydrochloric acid (NMT 0.012%).
:
A 1.2-g portion shows no more chloride than corresponds to 0.20 mL of 0.020 N hydrochloric acid (NMT 0.012%).
• Chloride and Sulfate, Sulfate  221
221 :
A 0.5-g portion shows no more sulfate than corresponds to 0.20 mL of 0.020 N sulfuric acid (NMT 0.04%).
:
A 0.5-g portion shows no more sulfate than corresponds to 0.20 mL of 0.020 N sulfuric acid (NMT 0.04%).
• Heavy Metals, Method II  231
231 :
NMT 10 ppm
:
NMT 10 ppm
Change to read:
• Organic Impurities
Solution A, Solution B, Diluent, Mobile phase, Sample solution, and Chromatographic system:
Proceed as directed in the Assay.
Standard stock solution:
Use the Standard solution, prepared as directed in the Assay. [Note—USP Deferoxamine Mesylate RS contains impurity A as a minor component. ]
Standard solution:
0.01 mg/mL of USP Deferoxamine Mesylate RS in Diluent from the Standard stock solution
System suitability
Samples:
Standard stock solution and Standard solution
Suitability requirements
Resolution:
NLT 2.0 between the impurity A and deferoxamine peaks, Standard stock solution
Relative standard deviation:
NMT 5.0% for the deferoxamine peak, Standard solution
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of each impurity in the portion of Deferoxamine Mesylate taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response of each impurity from the Sample solution |
| rS | = | = peak response of deferoxamine from the Standard solution |
| CS | = | = concentration of the Standard solution (mg/mL) |
| CU | = | = concentration of the Sample solution (mg/mL) |
Acceptance criteria:
See Table 2.
[Note—The reporting level for impurities is 0.04%. ]
Table 2
| Name | Relative Retention Time |
Acceptance Criteria, NMT (%) |
|---|---|---|
| Impurity A* | 0.85 | |
| Deferoxamine | 1.0 | — |
| Any unspecified impurity | — | 1.0 |
| Total impurities |
— | 5.0 |
| — | ||
|
*
Des-methylene impurity (desferrioxamine A1).
|
||
SPECIFIC TESTS
• pH  791
791 :
4.0–6.0, in a solution (1 in 100)
:
4.0–6.0, in a solution (1 in 100)
• Water Determination, Method I  921
921 :
NMT 2.0%
:
NMT 2.0%
• Sterility Tests  71
71 :
Where the label states that Deferoxamine Mesylate is sterile, it meets the requirements.
:
Where the label states that Deferoxamine Mesylate is sterile, it meets the requirements.
• Bacterial Endotoxins Test  85
85 :
Where the label states that Deferoxamine Mesylate is sterile or must be subjected to further processing during the preparation of injectable dosage forms, it contains NMT 0.33 USP Endotoxin Unit/mg of deferoxamine mesylate.
:
Where the label states that Deferoxamine Mesylate is sterile or must be subjected to further processing during the preparation of injectable dosage forms, it contains NMT 0.33 USP Endotoxin Unit/mg of deferoxamine mesylate.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers. Store in a cold place.
• Labeling:
Where it is intended for use in preparing injectable dosage forms, the label states that it is sterile or must be subjected to further processing during the preparation of injectable dosage forms.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Elena Gonikberg, Ph.D.
Principal Scientific Liaison 1-301-816-8251 |
(SM32010) Monographs - Small Molecules 3 |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2813
Pharmacopeial Forum: Volume No. 35(4) Page 847