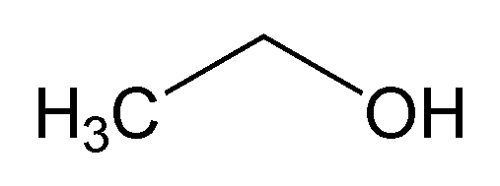
Alcohol
(al' ka hol).
DEFINITION
Alcohol contains NLT 92.3% and NMT 93.8%, by weight, corresponding to NLT 94.9% and NMT 96.0%, by volume, at 15.56 , of C2H5OH.
, of C2H5OH.
IDENTIFICATION
• A.
It meets the requirements of the test for Specific Gravity  841
841 .
.
• B. Infrared Absorption  197F
197F or
or  197S
197S :
Neat
:
Neat
IMPURITIES
Inorganic Impurities
• Limit of Nonvolatile Residue:
Evaporate 100 mL in a tared dish on a water bath, and dry at 100 –105
–105 for 1 h: the weight of the residue is NMT 2.5 mg.
for 1 h: the weight of the residue is NMT 2.5 mg.
Organic Impurities
• Procedure
Sample solution A:
Alcohol (substance under test)
Sample solution B:
300 ppm of 4-methylpentan-2-ol in Sample solution A
Standard solution A:
200 ppm of methanol in Sample solution A
Standard solution B:
10 ppm of methanol and 10 ppm of acetaldehyde in Sample solution A
Standard solution C:
30 ppm of acetal in Sample solution A
Standard solution D:
2 ppm of benzene in Sample solution A
Chromatographic system
Mode:
GC
Detector:
Flame ionization
Column:
0.32-mm × 30-m fused silica capillary column bonded with a 1.8-µm layer of phase G43
Split ratio:
20:1
Temperature
Detector:
280
Injector:
200
Column:
See the temperature program table below.
| Initial Temperature ( |
Temperature Ramp ( |
Final Temperature ( |
Hold Time at Final Temperature (min) |
|---|---|---|---|
| 40 | 0 | 40 | 12 |
| 40 | 10 | 240 | 10 |
Linear velocity:
35 cm/s
Carrier gas:
Helium
Injection size:
1.0 µL
System suitability
Sample:
Standard solution B
Suitability requirements
Resolution:
NLT 1.5 between the first major peak (acetaldehyde) and the second major peak (methanol)
Analysis
Samples:
Sample solution A, Sample solution B, Standard solution A, Standard solution B, Standard solution C, and Standard solution D
Methanol calculation
Result = (rU/rS)
| rU | = | = peak area of methanol from Sample solution A |
| rS | = | = peak area of methanol from Standard solution A |
Acetaldehyde and Acetal calculation
Result = [(AE/AT  AE) × CS] + [(DE/(DT
AE) × CS] + [(DE/(DT  DE) × CU]
DE) × CU]
| AE | = | = area of the acetaldehyde peak from Sample solution A |
| AT | = | = area of the acetaldehyde peak from Standard solution B |
| CS | = | = concentration of acetaldehyde added in Standard solution B, 10 ppm |
| DE | = | = area of the acetal peak from Sample solution A |
| DT | = | = area of the acetal peak from Standard solution C |
| CU | = | = concentration of acetal added in Standard solution C, 30 ppm |
Benzene calculation
Result = [BE/(BT  BE)] × CS
BE)] × CS
| BE | = | = area of the benzene peak from Sample solution A |
| BT | = | = area of the benzene peak from Standard solution D |
| CS | = | = concentration of benzene added in Standard solution D, 2 ppm |
[Note—If necessary, the identity of benzene can be confirmed using another suitable chromatographic system (stationary phase with a different polarity). ]
Other impurities calculation
Result = (rU/rM) × CM
| rU | = | = peak area of each impurity in Sample solution B |
| rM | = | = peak area of 4-methylpentan-2-ol in Sample solution B |
| CM | = | = concentration of 4-methylpentan-2-ol in Sample solution B |
Acceptance criteria:
See Impurity Table 1.
Impurity Table 1
| Name | Acceptance Criteria |
|---|---|
| Methanol | NMT 0.5, corresponding to 200 ppm |
| Acetaldehyde and Acetal | NMT 10 ppm, expressed as acetaldehyde |
| Benzene | NMT 2 ppm |
| Sum of all other impuritiesa | NMT 300 ppm |
|
a
Disregard any peaks of less than 9 ppm.
|
|
SPECIFIC TESTS
• Specific gravity  841
841 :
0.812–0.816 at 15.56
:
0.812–0.816 at 15.56 , indicating 92.3%–93.8%, by weight, or 94.9%–96.0%, by volume, of C2H5OH
, indicating 92.3%–93.8%, by weight, or 94.9%–96.0%, by volume, of C2H5OH
• Ultraviolet Absorption
Analytical wavelength:
235–340 nm
Cell:
5 cm
Reference:
Water
Acceptance criteria
Absorbance:
NMT 0.40 at 240 nm; NMT 0.30, between 250 nm and 260 nm; NMT 0.10, between 270 nm and 340 nm
Curve:
The absorption curve is smooth
• Clarity of Solution
[Note—The Sample solution is to be compared to Reference suspension A and to water in diffused daylight 5 min after preparation of Reference suspension A. ]
Hydrazine solution:
10 mg/mL of hydrazine sulfate in water. [Note—Allow to stand for 4–6 h. ]
Methenamine solution:
Transfer 2.5 g of methenamine to a 100-mL glass-stoppered flask, add 25.0 mL of water, insert the glass stopper, and mix to dissolve.
Primary opalescent suspension:
Transfer 25.0 mL of Hydrazine solution to the Methenamine solution in the 100-mL glass-stoppered flask. Mix, and allow to stand for 24 h. [Note—This suspension is stable for 2 months, provided it is stored in a glass container free from surface defects. The suspension must not adhere to the glass and must be well mixed before use. ]
Opalescence standard:
Transfer 15.0 mL of the Primary opalescent suspension to a 1000-mL volumetric flask, and dilute with water to volume. [Note—This suspension should not be used beyond 24 h after preparation. ]
Reference suspension A:
Opalescence standard and water (1 in 20)
Reference suspension B:
Opalescence standard and water (1 in 10)
Sample solution A:
Substance to be examined
Sample solution B:
Dilute 1.0 mL of Sample solution A with water to 20 mL, and allow to stand for 5 min before testing.
Analysis:
Transfer a sufficient portion of Sample solution A and Sample solution B to separate test tubes of colorless, transparent, neutral glass with a flat base and an internal diameter of 15–25 mm to obtain a depth of 40 mm. Similarly transfer portions of Reference suspension A, Reference suspension B, and water to separate matching test tubes. Compare Sample solution A, Sample solution B, Reference suspension A, Reference suspension B, and water in diffused daylight, viewing vertically against a black background. (See Spectrophotometry and Light-Scattering  851
851 , Visual Comparison.)
, Visual Comparison.)
[Note—The diffusion of light must be such that Reference suspension A can readily be distinguished from water, and that Reference suspension B can readily be distinguished from Reference suspension A. ]
Acceptance criteria:
Sample solution A and Sample solution B show the same clarity as that of water or their opalescence is not more pronounced than that of Reference suspension A.
• Acidity or Alkalinity
Phenolphthalein solution:
Dissolve 0.1 g of phenolphthalein in 80 mL of alcohol, and dilute with water to 100 mL.
Analysis:
To 20 mL of alcohol add 20 mL of freshly boiled and cooled water and 0.1 mL of Phenolphthalein solution. The solution is colorless. Add 1.0 mL of 0.01 N sodium hydroxide.
Acceptance criteria:
The solution is pink (30 ppm, expressed as acetic acid).
• Color of Solution
Standard stock solution:
Combine 3.0 mL of ferric chloride CS, 3.0 mL of cobaltous chloride CS, 2.4 mL of cupric sulfate CS, and 1.6 mL of dilute hydrochloric acid (10 g/L).
Standard solution:
Transfer 1.0 mL of Standard stock solution to a 100-mL volumetric flask, and dilute with dilute hydrochloric acid (10 g/L). [Note—Prepare the Standard solution immediately before use. ]
Sample solution:
Substance to be examined
Analysis:
Transfer a sufficient portion of the Sample solution to a test tube of colorless, transparent, neutral glass with a flat base and an internal diameter of 15–25 mm to obtain a depth of 40 mm. Similarly transfer portions of the Standard solution and water to separate, matching test tubes. Compare the Sample solution, Standard solution, and water in diffused daylight, viewing vertically against a white background. (See Spectrophotometry and Light-Scattering  851
851 , Visual Comparison.)
, Visual Comparison.)
Acceptance criteria:
The Sample solution has the appearance of water or is not more intensely colored than the Standard solution.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers, protected from light.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2088
Pharmacopeial Forum: Volume No. 30(5) Page 1843