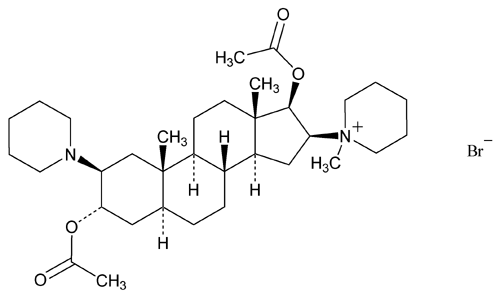
Vecuronium Bromide
» Vecuronium Bromide contains not less than 98.0 percent and not more than 102.0 percent of C34H57BrN2O4, calculated on the dried basis.
Packaging and storage—
Preserve in tight containers, and store at room temperature.
USP Reference standards  11
11 —
—
USP Endotoxin RS.
USP Pancuronium Bromide RS .
.
USP Vecuronium Bromide RS .
.
USP Vecuronium Bromide Related Compound A RS .
.
USP Vecuronium Bromide Related Compound B RS .
.
USP Vecuronium Bromide Related Compound C RS .
.
USP Vecuronium Bromide Related Compound F RS .
.
USP Endotoxin RS.
USP Pancuronium Bromide RS
 .
.
USP Vecuronium Bromide RS
 .
.
USP Vecuronium Bromide Related Compound A RS
 .
.
USP Vecuronium Bromide Related Compound B RS
 .
.
USP Vecuronium Bromide Related Compound C RS
 .
.
USP Vecuronium Bromide Related Compound F RS
 .
.
Identification—
B:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of the Standard preparation, as obtained in the Assay.
Specific rotation  781S
781S :
between
:
between  16
16 and
and  20
20 , at 20
, at 20 .
.
Test solution:
10 mg per mL, in dehydrated alcohol.
Bacterial endotoxins  85
85 :
not more than 10 USP Endotoxin Units per mg of vecuronium bromide.
:
not more than 10 USP Endotoxin Units per mg of vecuronium bromide.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for two hours: it loses not more than 2.5% of its weight.
for two hours: it loses not more than 2.5% of its weight.
Related compounds—
[note—This note applies to all of the solution preparations. The addition, with sonication, of a small amount of acetonitrile (a maximum of 0.5 mL per 25 mg) to the weighed quantity of the related compounds, as well as to vecuronium bromide, may be used to aid in dissolution. Shaking and sonication may also be used after the addition of the required amount of 0.0025 N hydrochloric acid.]
Cation suppressor regeneration solution:
0.02 M tetrabutylammonium hydroxide.
Mobile phase—
Mix 1500 mL of filtered water, 250 mL of filtered methanol, and 1 mL of hydrochloric acid in a 2000-mL volumetric flask. Leave at room temperature for a few minutes, add 45 mL of filtered tetrahydrofuran, and dilute with water to volume. Mix and degas.[note—Avoid evaporation of tetrahydrofuran during degassing.]
System suitability solution—
Dissolve suitable quantities of USP Vecuronium Bromide RS, USP Pancuronium Bromide RS, USP Vecuronium Bromide Related Compound A RS, USP Vecuronium Bromide Related Compound B RS, USP Vecuronium Bromide Related Compound C RS, and USP Vecuronium Bromide Related Compound F RS in 0.0025 N hydrochloric acid, and dilute quantitatively, and stepwise if necessary, with 0.0025 N hydrochloric acid to obtain a solution containing about 0.005 mg of each compound per mL.
Standard solution—
Dissolve an accurately weighed quantity of USP Vecuronium Bromide RS in 0.0025 N hydrochloric acid to obtain a solution having a known concentration of about 0.005 mg per mL.
Test solution—
Transfer about 25 mg of Vecuronium Bromide, accurately weighed, to a 25-mL volumetric flask. Add 0.5 mL of acetonitrile, sonicate to aid dissolution, rapidly add 0.0025 N hydrochloric acid to volume, and mix promptly.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a conductivity detector, a 4-mm cation suppressor, and a 4.6-mm × 25-cm column that contains 5-µm packing L1. The flow rate is about 1.5 mL per minute. The flow rate for the cation suppressor is about 2 mL per minute. Chromatograph the System suitability solution, and record the peak responses as directed for Procedure: the relative retention times are given in Table 1; and the ratio of the height of the peak for vecuronium bromide related compound F to the height of the trough between the peak for vecuronium bromide related compound F and the peak for pancuronium bromide is not less than 2.0. Chromatograph the Standard solution, and record the peak responses as directed for Procedure: the relative standard deviation for three replicate injections of vecuronium bromide is not more than 10.0%. [note—The system may need equilibration for 4 hours or longer. It is recommended that the column be rinsed with a mixture of acetonitrile and water (1:1) and stored in the same mixture when not in use.]
)—
The liquid chromatograph is equipped with a conductivity detector, a 4-mm cation suppressor, and a 4.6-mm × 25-cm column that contains 5-µm packing L1. The flow rate is about 1.5 mL per minute. The flow rate for the cation suppressor is about 2 mL per minute. Chromatograph the System suitability solution, and record the peak responses as directed for Procedure: the relative retention times are given in Table 1; and the ratio of the height of the peak for vecuronium bromide related compound F to the height of the trough between the peak for vecuronium bromide related compound F and the peak for pancuronium bromide is not less than 2.0. Chromatograph the Standard solution, and record the peak responses as directed for Procedure: the relative standard deviation for three replicate injections of vecuronium bromide is not more than 10.0%. [note—The system may need equilibration for 4 hours or longer. It is recommended that the column be rinsed with a mixture of acetonitrile and water (1:1) and stored in the same mixture when not in use.]
Procedure—
Separately inject equal volumes (about 25 µL) of the Standard solution and the Test solution into the chromatograph, record the chromatograms, and measure the peak responses. Calculate the percentage of each vecuronium bromide related compound in the portion of Vecuronium Bromide taken by the formula:
100(1/F)(CS / CT)(rU / rS)
in which F is the relative response factor of the specific related compound obtained from Table 1; CS is the concentration, in mg per mL, of USP Vecuronium Bromide RS in the Standard solution; CT is the concentration, in mg per mL, of vecuronium bromide in the Test solution; rU is the peak area of each related compound in the Test solution; and rS is the peak area for vecuronium bromide in the Standard solution: the limits of impurities are specified in Table 1.
Table 1
| Compound Name | Relative Retention Time |
Relative Response Factor (RRF) |
Limit (%) |
| Pancuronium bromide | about 0.5 | 1.1 | 0.5 |
| Vecuronium bromide related compound F1 | about 0.6 | 1.3 | 0.5 |
| Vecuronium bromide related compound C2 | about 0.9 | 1.4 | 0.5 |
| Vecuronium bromide | 1.0 | — | — |
| Vecuronium bromide related compound A3 | about 1.8 | 0.4 | 0.3 |
| Vecuronium bromide related compound B4 | about 2.2 | 1.0 | 0.5 |
| Any individual unspecified impurity | — | 1.0 | 0.1 |
| Total | — | — | 1.0 |
|
1
3-Deacetyl vecuronium bromide, (Piperidinium, 1-[(2
2
3,17-Bis deacetyl vecuronium bromide; (Piperidinium, 1-[(2
3
Dipiperidino diol diacetate; (3
4
17-Deacetyl vecuronium bromide; (Piperidinium, 1-[(2
|
|||
Assay—
Solution A—
Transfer 8.0 g of sodium perchlorate to a 1000-mL volumetric flask, dissolve in 6.0 mL of water, dilute with acetonitrile to volume, mix, filter, and degas.
Solution B—
Transfer 3.2 g of ammonium chloride to a 2000-mL volumetric flask, dissolve in 16 mL of ammonium hydroxide, dilute with methanol to volume, mix, filter, and degas. [note—Avoid excessive degassing to prevent the loss of ammonium hydroxide.].
Mobile phase—
Prepare a mixture of Solution A and Solution B (3:2). Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
Diluent—
Pipet 1.0 mL of 1 N hydrochloric acid into a 1000-mL volumetric flask, dilute with acetonitrile to volume, and mix.
Standard preparation—
Dissolve an accurately weighed quantity of USP Vecuronium Bromide RS in Diluent to obtain a solution having a known concentration of about 0.5 mg per mL.
Assay preparation—
Transfer about 50 mg of Vecuronium Bromide, accurately weighed, to a 100-mL volumetric flask, dissolve in and dilute with Diluent, and mix.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 215-nm detector and a 4.6-mm × 25-cm column that contains 5-µm packing L3. The flow rate is about 0.5 mL per minute. The column temperature is maintained at 40
)—
The liquid chromatograph is equipped with a 215-nm detector and a 4.6-mm × 25-cm column that contains 5-µm packing L3. The flow rate is about 0.5 mL per minute. The column temperature is maintained at 40 . Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the column efficiency is not less than 5000 theoretical plates; and the relative standard deviation for replicate injections is not more than 2.0%.
. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the column efficiency is not less than 5000 theoretical plates; and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 20 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the quantity, in mg, of C34H57BrN2O4 in the portion of Vecuronium Bromide taken by the formula:
100C(rU / rS)
in which C is the concentration, in mg per mL, of USP Vecuronium Bromide RS in the Standard preparation; and rU and rS are the peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Ravi Ravichandran, Ph.D.
Senior Scientist 1-301-816-8330 |
(MDPP05) Monograph Development-Psychiatrics and Psychoactives |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
|
| Radhakrishna S Tirumalai, Ph.D.
Senior Scientist 1-301-816-8339 |
(MSA05) Microbiology and Sterility Assurance |
USP32–NF27 Page 3850
Pharmacopeial Forum: Volume No. 33(4) Page 682
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.