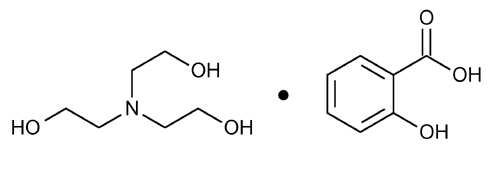
Trolamine Salicylate
» Trolamine Salicylate is a compounded mixture of Trolamine and Salicylic Acid in propylene glycol. It contains not less than 95.0 percent and not more than 105.0 percent of the labeled amount of C13H21NO6.
Packaging and storage—
Preserve in tight containers in a cool place.
USP Reference standards  11
11 —
—
USP Salicylic Acid RS.
USP Salicylic Acid RS.
Identification, Ultraviolet Absorption  197U
197U —
—
Solution:
1 mg per mL, in 0.1-cm cells. The test solution and the Standard solution contain the equivalent of 1 mg of salicylic acid per mL.
Medium:
methanol.
Absorptivities do not differ by more than 1.0%.
Specific gravity  841
841 :
between 1.190 and 1.220.
:
between 1.190 and 1.220.
Refractive index  831
831 :
between 1.505 and 1.535 at 20
:
between 1.505 and 1.535 at 20 .
.
pH  791
791 :
between 6.5 and 7.5, in a 50 mg per mL solution in water.
:
between 6.5 and 7.5, in a 50 mg per mL solution in water.
Limit of free salicylic acid—
Adsorbent:
0.25-mm layer of chromatographic silica gel mixture.
Test solution—
Transfer about 48 mg of Trolamine Salicylate, accurately weighed, to a 50-mL volumetric flask, dilute with xylene to volume, and mix.
Standard solution 1—
Dissolve an accurately weighed quantity of USP Salicylic Acid RS in xylene, and dilute quantitatively, and stepwise if necessary, with xylene to obtain a solution having a known concentration of about 0.2 mg per mL.
Standard solution 2—
Dilute an accurately measured quantity of Standard solution 1 in xylene to obtain a solution having a known concentration of about 0.1 mg per mL.
Application volume:
5 µL of each solution.
Developing solvent system:
a mixture of toluene, acetone, and glacial acetic acid (17:8:0.2).
Procedure—
Proceed as directed for Thin-Layer Chromatography under Chromatography  621
621 . Develop in a chamber previously equilibrated with Developing solvent system. Examine the plate under long-wavelength UV light. Any secondary spot obtained from the Test solution is not greater in size or intensity than the spot obtained from Standard solution 1: not more than 0.02% of free salicylic acid is found.
. Develop in a chamber previously equilibrated with Developing solvent system. Examine the plate under long-wavelength UV light. Any secondary spot obtained from the Test solution is not greater in size or intensity than the spot obtained from Standard solution 1: not more than 0.02% of free salicylic acid is found.
Chromatographic purity—
Mobile phase, Standard preparation, and Chromatographic system—
Proceed as directed in the Assay.
Test preparation—
Use the Assay preparation.
Procedure—
Inject a volume (about 10 µL) of the Test preparation into the chromatograph, record the chromatogram, and measure all of the peak responses. Calculate the percentage of each impurity in the portion of Trolamine Salicylate taken by the formula:
100(ri / rs)
in which ri is the peak response for each impurity, and rs is the sum of the responses of all the peaks: not more than 1.0% of any individual impurity is found, and not more than 2.0% of total impurities is found.
Assay—
Mobile phase—
Prepare a filtered and degassed mixture of water and acetonitrile (7:3). Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
Standard preparation—
Dissolve an accurately weighed quantity of USP Salicylic Acid RS in methanol, and dilute quantitatively, and stepwise if necessary, with methanol to obtain a solution having a known concentration of about 48 µg per mL.
Assay preparation—
Transfer a portion of Trolamine Salicylate, equivalent to about 300 mg of salicylic acid, accurately weighed, to a 250-mL volumetric flask, and dilute with methanol to volume. Transfer 2 mL of this solution to a 50-mL volumetric flask, dilute with methanol to volume, and mix.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 308-nm detector and a 4.0-mm × 12.5-cm column that contains packing L1. The flow rate is about 1 mL per minute. The column temperature is maintained at 30
)—
The liquid chromatograph is equipped with a 308-nm detector and a 4.0-mm × 12.5-cm column that contains packing L1. The flow rate is about 1 mL per minute. The column temperature is maintained at 30 . Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the column efficiency is not less than 8000 theoretical plates; the tailing factor is not more than 1.5; and the relative standard deviation for replicate injections is not more than 2.0%.
. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the column efficiency is not less than 8000 theoretical plates; the tailing factor is not more than 1.5; and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 10 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the percentage of C13H21NO6 in the portion of Trolamine Salicylate taken by the formula:
100(rU / rS)
in which rU and rS are the peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Clydewyn M. Anthony, Ph.D.
Scientist 1-301-816-8139 |
(MDCCA05) Monograph Development-Cough Cold and Analgesics |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3820
Pharmacopeial Forum: Volume No. 30(4) Page 1312
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.