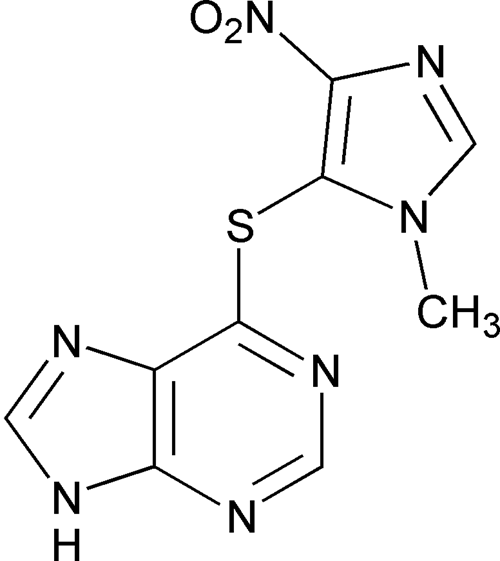
Azathioprine
1H-Purine, 6-[(1-methyl-4-nitro-1H-imidazol-5-yl)thio]-.
6-[(1-Methyl-4-nitroimidazol-5-yl)thio]purine
» Azathioprine contains not less than 98.0 percent and not more than 101.5 percent of C9H7N7O2S, calculated on the dried basis.
Packaging and storage—
Preserve in tight, light-resistant containers.
Identification—
B:
The principal spot in the test preparation chromatogram in the test for Limit of mercaptopurine shows the same RF value as that obtained with the solution of USP Azathioprine RS.
Acidity or alkalinity—
Shake 2.0 g with 100 mL of water for 15 minutes, and filter: 20.0 mL of the filtrate requires for neutralization not more than 0.10 mL of 0.020 N hydrochloric acid or not more than 0.10 mL of 0.020 N sodium hydroxide, methyl red TS being used as the indicator.
Loss on drying  731
731 —
Dry it in vacuum at 105
—
Dry it in vacuum at 105 for 5 hours: it loses not more than 1.0% of its weight.
for 5 hours: it loses not more than 1.0% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Limit of mercaptopurine—
Prepare three solutions in 6 N ammonium hydroxide containing, respectively, 20 mg of Azathioprine per mL, 20 mg of USP Azathioprine RS per mL, and 200 µg of USP Mercaptopurine RS, on the anhydrous basis, per mL. Apply 5-µL volumes of the solutions at points about 2 cm from the bottom edge of a thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of microcrystalline cellulose. Allow the spots to dry, and develop the chromatogram in a suitable chamber, using butyl alcohol, previously saturated with 6 N ammonium hydroxide, as the solvent, until the solvent front has moved about 15 cm from the point of application. Remove the plate, air-dry, and locate the spots by viewing under short- and long-wavelength UV light: any spot in the chromatogram from Azathioprine, other than the principal spot, is not more intense than the spot in the chromatogram obtained with USP Mercaptopurine RS (1.0%).
) coated with a 0.25-mm layer of microcrystalline cellulose. Allow the spots to dry, and develop the chromatogram in a suitable chamber, using butyl alcohol, previously saturated with 6 N ammonium hydroxide, as the solvent, until the solvent front has moved about 15 cm from the point of application. Remove the plate, air-dry, and locate the spots by viewing under short- and long-wavelength UV light: any spot in the chromatogram from Azathioprine, other than the principal spot, is not more intense than the spot in the chromatogram obtained with USP Mercaptopurine RS (1.0%).
Assay—
Dissolve about 300 mg of Azathioprine, accurately weighed, in 80 mL of dimethylformamide. Add 5 drops of a 1 in 100 solution of thymol blue in dimethylformamide, and titrate with 0.1 N tetrabutylammonium hydroxide VS, using a magnetic stirrer, and taking precautions to prevent absorption of atmospheric carbon dioxide. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N tetrabutylammonium hydroxide is equivalent to 27.73 mg of C9H7N7O2S.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Feiwen Mao, M.S.
Scientist 1-301-816-8320 |
(MDOOD05) Monograph Development-Ophthalmics Oncologics and Dermatologicals |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 1610
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.