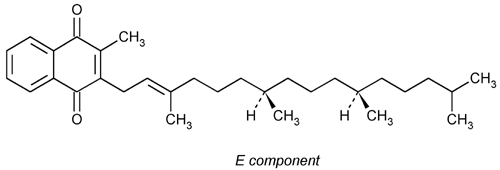
Phytonadione
1,4-Naphthalenedione, 2-methyl-3-(3,7,11,15-tetramethyl-2-hexadecenyl)-, [R-[R*,R*-(E)]]-.
Phylloquinone
» Phytonadione is a mixture of E and Z isomers containing not less than 97.0 percent and not more than 103.0 percent of C31H46O2. It contains not more than 21.0 percent of the Z isomer.
Packaging and storage—
Preserve in tight, light-resistant containers.
Identification—
Solution:
10 µg per mL.
Medium: n
-hexane.
Absorptivities at 248 nm do not differ by more than 3.0%.
Refractive index  831
831 :
between 1.523 and 1.526.
:
between 1.523 and 1.526.
Reaction—
A 1 in 20 solution of it in dehydrated alcohol is neutral to litmus.
Limit of menadione—
Mix about 20 mg with 0.5 mL of a mixture of equal volumes of 6 N ammonium hydroxide and alcohol, then add 1 drop of ethyl cyanoacetate, and shake gently: no purple or blue color is produced.
Z isomer content—
[note—Protect solutions containing Phytonadione from exposure to light.]
Mobile phase, Internal standard solution, Assay preparation, Chromatographic system, and Procedure—
Proceed as directed in the Assay, except to calculate the percentage of Zisomer taken by the formula:
100rZ / (rZ + rE)
in which rZ is the peak area of the (Z)-phytonadione isomer peak and rE is the peak area of the (E)-phytonadione isomer peak obtained from the Assay preparation.
Assay—
[note—Protect solutions containing Phytonadione from exposure to light.]
Mobile phase—
Prepare a filtered and degassed solution of n-hexane and n-amyl alcohol (2000:1.5).
Internal standard solution—
Dissolve cholesteryl benzoate in Mobile phase to obtain a solution having a concentration of 2.5 mg per mL.
Standard preparation—
Transfer about 60 mg of USP Phytonadione RS, accurately weighed, to a 50-mL volumetric flask, add 20 mL of Mobile phase, mix, dilute with Mobile phase to volume, and again mix. Pipet 4 mL of the resulting solution into a 50-mL volumetric flask, dilute with Mobile phase to volume, and mix. Pipet 10 mL of this solution and 7 mL of Internal standard solution into a 25-mL volumetric flask, dilute with Mobile phase to volume, and mix.
Assay preparation—
Prepare as directed under Standard preparation, using Phytonadione instead of the Reference Standard.
Chromatographic system
(see Chromatography  621
621 )—The liquid chromatographic is equipped with a 254-nm detector and a 4.6-mm × 25-cm column that contains packing L3. The flow rate is about 1 mL per minute. Chromatograph replicate injections of the Standard preparation, and record the peak responses as directed for Procedure: the relative standard deviation is not more than 2.0%, and the resolution, R, between (Z)-phytonadione and (E)-phytonadione is not less than 1.5.
)—The liquid chromatographic is equipped with a 254-nm detector and a 4.6-mm × 25-cm column that contains packing L3. The flow rate is about 1 mL per minute. Chromatograph replicate injections of the Standard preparation, and record the peak responses as directed for Procedure: the relative standard deviation is not more than 2.0%, and the resolution, R, between (Z)-phytonadione and (E)-phytonadione is not less than 1.5.
Procedure—
Separately inject equal volumes (about 50 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. The relative retention times are about 0.7 for the internal standard, 0.9 for (Z)-phytonadione, and 1.0 for (E)-phytonadione. Calculate the quantity, in mg, of C31H46O2 in the portion of Phytonadione taken by the formula:
1.56C(RU / RS)
in which C is the concentration, in µg per mL, of USP Phytonadione RS in the Standard preparation, and RU and RS are the relative peak response ratios for the Assay preparation and the Standard preparation, respectively. Calculate RU and RS by the formula:
(response for the (Z)-phytonadione peak + response for the (E)-phytonadione peak) / response for the internal standard peak.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Curtis Phinney
1-301-816-8540 |
(DSN05) Dietary Supplements - Non-Botanicals |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3302
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.