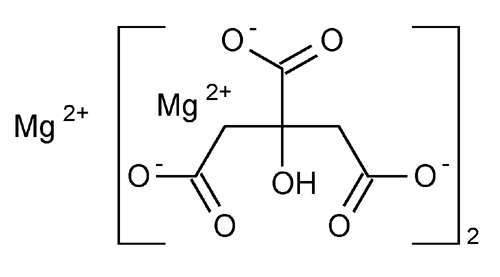
Magnesium Citrate
1,2,3-Propanetricarboxylic acid, hydroxy-, magnesium salt (2:3).
Magnesium citrate (3:2)
» Magnesium Citrate contains not less than 14.5 percent and not more than 16.4 percent of magnesium (Mg), calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Labeling—
Magnesium Citrate that loses not more than 2.0% of its weight in the test for Loss on drying may be labeled as Anhydrous Magnesium Citrate.
Identification—
A:
A solution (10 mg per mL) responds to the tests for Magnesium  191
191 .
.
B:
A solution (80 mg per mL) responds to the tests for Citrate  191
191 .
.
pH  791
791 :
between 5.0 and 9.0, in a suspension (50 mg per mL).
:
between 5.0 and 9.0, in a suspension (50 mg per mL).
Loss on drying  731
731 —
Dry 1 g in a mechanical convection oven at 135
—
Dry 1 g in a mechanical convection oven at 135 for 16 hours, then to constant weight: it loses not more than 29% of its weight, except that where it is labeled as anhydrous, it loses not more than 2.0% of its weight.
for 16 hours, then to constant weight: it loses not more than 29% of its weight, except that where it is labeled as anhydrous, it loses not more than 2.0% of its weight.
Chloride  221
221 —
A 300-mg portion shows no more chloride than corresponds to 0.20 mL of 0.020 N hydrochloric acid (0.05%).
—
A 300-mg portion shows no more chloride than corresponds to 0.20 mL of 0.020 N hydrochloric acid (0.05%).
Sulfate  221
221 —
A 100-mg portion shows no more sulfate than corresponds to 0.20 mL of 0.020 N sulfuric acid (0.2%).
—
A 100-mg portion shows no more sulfate than corresponds to 0.20 mL of 0.020 N sulfuric acid (0.2%).
Arsenic, Method I  211
211 :
3 µg per g.
:
3 µg per g.
Heavy metals, Method I  231
231 —
Dissolve 0.4 g in 25 mL of water, and proceed as directed for Test Preparation, except to use glacial acetic acid to adjust the pH: the limit is 50 µg per g.
—
Dissolve 0.4 g in 25 mL of water, and proceed as directed for Test Preparation, except to use glacial acetic acid to adjust the pH: the limit is 50 µg per g.
Iron  241
241 —
Boil 50 mg with 5 mL of 2 N nitric acid for 1 minute. Cool, dilute with water to 45 mL, add 2 mL of hydrochloric acid, and mix: the limit is 200 µg per g.
—
Boil 50 mg with 5 mL of 2 N nitric acid for 1 minute. Cool, dilute with water to 45 mL, add 2 mL of hydrochloric acid, and mix: the limit is 200 µg per g.
Limit of calcium—
Dilute hydrochloric acid
, Lanthanum solution, Standard preparations, and Blank solution—Prepare as directed in the test for Calcium under Magnesium Carbonate.
Test preparation—
Transfer 250 mg of Magnesium Citrate to a beaker, add 30 mL of Dilute hydrochloric acid, and stir until dissolved. Transfer the solution so obtained to a 200-mL volumetric flask containing 4 mL of Lanthanum solution, dilute with water to volume, and mix.
Procedure—
Proceed as directed in the test for Calcium under Magnesium Carbonate: the limit is 1.0%, calculated on the dried basis.
Assay—
Weigh accurately about 400 mg of Magnesium Citrate, dissolve in 50 mL of water, add 20 mL of ammonia–ammonium chloride buffer TS and 0.1 mL of eriochrome black TS, and titrate with 0.05 M edetate disodium VS to a blue endpoint. Perform a blank determination, and make any necessary correction. From the volume of 0.05 M edetate disodium consumed, deduct the volume of 0.05 M edetate disodium corresponding to the amount of calcium in the portion of Magnesium Citrate taken, based on the amount of calcium found in the test for Limit of calcium. Each mg of calcium (Ca) is equivalent to 0.25 mL of 0.05 M edetate disodium. The difference is the volume of 0.05 M edetate disodium consumed by the magnesium. Each mL of 0.05 M edetate disodium is equivalent to 1.215 mg of magnesium (Mg).
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
| Monograph | Elena Gonikberg, Ph.D.
Senior Scientist 1-301-816-8251 |
(MDGRE05) Monograph Development-Gastrointestinal Renal and Endocrine |
USP32–NF27 Page 2831