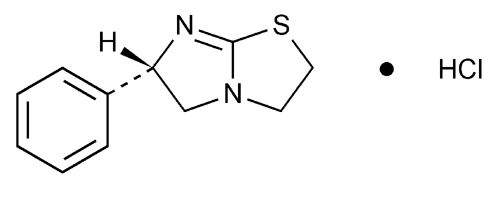
Levamisole Hydrochloride
Imidazo[2,1-b]thiazole, 2,3,5,6-tetrahydro-6-phenyl-, monohydrochloride, (S)-.
(
» Levamisole Hydrochloride contains not less than 98.5 percent and not more than 101.0 percent of C11H12N2S·HCl, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers, protected from light.
Completeness of solution  641
641 —
A test solution of 500 mg of Levamisole Hydrochloride dissolved in 10 mL of water meets the requirements.
—
A test solution of 500 mg of Levamisole Hydrochloride dissolved in 10 mL of water meets the requirements.
Color of solution—
The test solution prepared for the test for Completeness of solution is colorless or not more intensely colored than a color matching fluid prepared by mixing 2.5 mL of Matching Fluid F (see Color and Achromicity  631
631 ) with 97.5 mL of 0.12 N hydrochloric acid.
) with 97.5 mL of 0.12 N hydrochloric acid.
Identification—
A:
The IR absorption spectrum of a potassium bromide dispersion of it, previously dried, exhibits maxima only at the same wavelengths as that of a similar preparation of USP Levamisole Hydrochloride RS.
B:
The color, size, and RF value of the principal spot in the chromatogram of Test solution B obtained in the test for Chromatographic purity, when examined under short-wavelength UV light, correspond to the respective characteristics of the principal spot in the chromatogram of Reference solution A obtained in the test for Chromatographic purity.
C:
A solution of it responds to the tests for Chloride  191
191 .
.
Light absorption—
Its absorbance (see Spectrophotometry and Light-scattering  851
851 ) at 310 nm, determined in a 0.2 N methanolic hydrochloric acid solution containing 1 mg per mL using a 1-cm cell, is not more than 0.20.
) at 310 nm, determined in a 0.2 N methanolic hydrochloric acid solution containing 1 mg per mL using a 1-cm cell, is not more than 0.20.
pH  791
791 :
between 3.0 and 4.5, in a solution (1 in 20).
:
between 3.0 and 4.5, in a solution (1 in 20).
Loss on drying  731
731 :
Dry it at 105
:
Dry it at 105 for 4 hours: it loses not more than 0.5% of its weight.
for 4 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method I  231
231 :
0.001%.
:
0.001%.
Chromatographic purity—
Prepare a solution of it in methanol containing 50 mg per mL (Test solution A). Dilute 1.0 mL of Test solution A to 10 mL with methanol, and mix (Test solution B). Prepare a solution of USP Levamisole Hydrochloride RS in methanol having a concentration of 5 mg per mL (Reference solution A). Dilute 1.0 mL of Test solution B to 20 mL with methanol, and mix (Reference solution B). Apply separate 10-µL portions of the four solutions on the starting line to a suitable thin-layer chromatographic plate (see Chromatography  621
621 ), coated with a 0.25-mm layer of chromatographic silica gel mixture. Allow the spots to dry, and develop the chromatogram in a solvent system consisting of a mixture of toluene, acetone, and ammonium hydroxide (60:40:1) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, and dry it at 105
), coated with a 0.25-mm layer of chromatographic silica gel mixture. Allow the spots to dry, and develop the chromatogram in a solvent system consisting of a mixture of toluene, acetone, and ammonium hydroxide (60:40:1) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, and dry it at 105 for 15 minutes. Locate the spots on the plate by examination under short-wavelength UV light: any spot obtained from Test solution A, other than the one corresponding to levamisole, does not exceed, in size or intensity, the principal spot obtained from Reference solution B, corresponding to not more than 0.5% of any individual impurity. Expose the plate to iodine vapor in a closed chamber for 15 minutes, and locate the spots on the plate: any spot obtained from Test solution A, other than the one corresponding to levamisole, does not exceed, in size or intensity, the principal spot obtained from Reference solution B, corresponding to not more than 0.5% of any individual impurity, and the total of all impurities found does not exceed 1.0%.
for 15 minutes. Locate the spots on the plate by examination under short-wavelength UV light: any spot obtained from Test solution A, other than the one corresponding to levamisole, does not exceed, in size or intensity, the principal spot obtained from Reference solution B, corresponding to not more than 0.5% of any individual impurity. Expose the plate to iodine vapor in a closed chamber for 15 minutes, and locate the spots on the plate: any spot obtained from Test solution A, other than the one corresponding to levamisole, does not exceed, in size or intensity, the principal spot obtained from Reference solution B, corresponding to not more than 0.5% of any individual impurity, and the total of all impurities found does not exceed 1.0%.
Assay—
Dissolve about 200 mg of Levamisole Hydrochloride, accurately weighed, in 30 mL of alcohol. Add 5.0 mL of 0.01 N hydrochloric acid, and titrate with 0.1 N sodium hydroxide VS, determining the two inflection points potentiometrically. Determine the volume, in mL, of 0.1 N sodium hydroxide consumed between the two inflection points. Each mL of 0.1 N sodium hydroxide consumed is equivalent to 24.08 mg of C11H12N2S·HCl.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
| Monograph | Thomas A. Sigambris, M.S.
Scientist 1-301-998-6789 |
(VET05) Veterinary Drugs 05 |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 2764