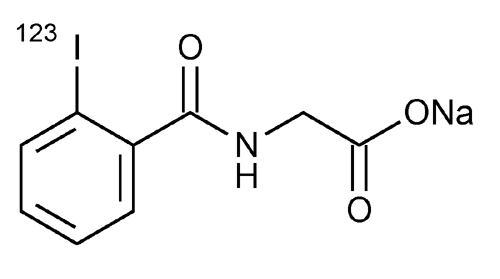
Iodohippurate Sodium I 123 Injection
Glycine, N-[2-(iodo-123I)benzoyl]-, monosodium salt.
Sodium o-iodo-123I-hippurate
» Iodohippurate Sodium I 123 Injection is a sterile, aqueous solution containing o-iodohippurate sodium in which a portion of the molecules contain radioactive iodine (123I) in the molecular structure. It may contain a preservative or stabilizer.
Iodohippurate Sodium I 123 Injection contains not less than 90.0 percent and not more than 110.0 percent of the labeled amount of I 123 as iodohippurate sodium expressed in megabecquerels (microcuries or millicuries) per mL at the time indicated in the labeling. It contains not less than 90.0 percent and not more than 110.0 percent of the labeled amount of o-iodohippuric acid. Other chemical forms of radioactivity do not exceed 3.0 percent of total radioactivity.
Packaging and storage—
Preserve in single-dose or in multiple-dose containers that are adequately shielded.
Labeling—
Label it to include the following, in addition to the information specified for Labeling under Injections  1
1 : the time and date of calibration; the amount of I 123 as iodohippurate sodium expressed as total megabecquerels (microcuries or millicuries) per mL at the time of calibration; the name and quantity of any added preservative or stabilizer; the expiration time; and the statement “Caution—Radioactive Material.” The labeling indicates that in making dosage calculations, correction is to be made for radioactive decay, and also indicates that the radioactive half-life of I 123 is 13.2 hours.
: the time and date of calibration; the amount of I 123 as iodohippurate sodium expressed as total megabecquerels (microcuries or millicuries) per mL at the time of calibration; the name and quantity of any added preservative or stabilizer; the expiration time; and the statement “Caution—Radioactive Material.” The labeling indicates that in making dosage calculations, correction is to be made for radioactive decay, and also indicates that the radioactive half-life of I 123 is 13.2 hours.
Radionuclidic identification (see Radioactivity  821
821 )—
Its gamma-ray spectrum is identical to that of a specimen of I 123 of known purity that exhibits a major photopeak having an energy of 159 keV.
)—
Its gamma-ray spectrum is identical to that of a specimen of I 123 of known purity that exhibits a major photopeak having an energy of 159 keV.
Bacterial endotoxins  85
85 —
The limit of endotoxin content is not more than 175/V USP Endotoxin Unit per mL of the Injection, when compared with the USP Endotoxin RS, in which V is the maximum recommended total dose, in mL, at the expiration time.
—
The limit of endotoxin content is not more than 175/V USP Endotoxin Unit per mL of the Injection, when compared with the USP Endotoxin RS, in which V is the maximum recommended total dose, in mL, at the expiration time.
pH  791
791 :
between 7.0 and 8.5.
:
between 7.0 and 8.5.
Radionuclidic purity—
Using a suitable counting assembly (see Selection of a Counting Assembly under Radioactivity  821
821 ), determine the radionuclidic purity of the Injection: not less than 85% of the total radioactivity is present as I 123.
), determine the radionuclidic purity of the Injection: not less than 85% of the total radioactivity is present as I 123.
Radiochemical purity—
Mobile phase, Standard preparation, and Chromatographic system—
Proceed as directed in the Assay for o-iodohippuric acid, except to provide that the liquid chromatograph is also equipped with a radioactivity detector (see Radioactivity  821
821 ).
).
Procedure—
Inject about 50 µL equivalent to 1.8 to 3.7 MBq (50 to 100 µCi) of Injection into the chromatograph, record the chromatogram, and measure the area of all radioactivity peaks. The radioactivity under the o-iodohippuric acid I 123 peak is not less than 97.0% of the total area of all peaks observed, and its retention time is within ±10% of the value obtained for USP o-Iodohippuric Acid RS.
Biological distribution—
Inject intravenously between 0.75 and 22 MBq (20 and 600 µCi) of the Injection, in a volume of 0.10 mL to 0.15 mL, into the caudal vein of each of three 125-g to 225-g anesthetized rats. Clamp the opening of the urethra with a hemostat. Sacrifice the animals 1 hour after administration, and carefully remove the intact bladder and contents and thyroid by dissection. Place each organ and remaining carcass (excluding the tail) in separate, suitable counting containers, and determine the radioactivity, in counts per minute, in each container with an appropriate detector using the same counting geometry. Determine the percentage of radioactivity in each organ: not less than 75% of the administered dose is found in the bladder, and not more than 3% of the administered dose is found in the thyroid in two of the rats.
Other requirements—
It meets the requirements under Injections  1
1 , except that the Injection may be distributed or dispensed prior to the completion of the test for Sterility, the latter test being started on the day of final manufacture, and except that it is not subject to the recommendation on Volume in Container.
, except that the Injection may be distributed or dispensed prior to the completion of the test for Sterility, the latter test being started on the day of final manufacture, and except that it is not subject to the recommendation on Volume in Container.
Assay for o-iodohippuric acid—
Mobile phase—
Prepare a filtered and degassed mixture of water, methanol, and glacial acetic acid (75:25:1). Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
Standard preparation—
Using an accurately weighed quantity of USP o-Iodohippuric Acid RS, prepare a solution in water having a known concentration of about 2 mg per mL.
Assay preparation—
Use Iodohippurate Sodium I 123 Injection.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 265-nm detector and an 8-mm × 10-cm column that contains packing L11. The flow rate is about 5 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed under Procedure: the column efficiency determined from the analyte peak is not less than 500 theoretical plates, the tailing factor for the analyte peak is not more than 1.5, and the relative standard deviation for replicate injections is not more than 1.5%.
)—
The liquid chromatograph is equipped with a 265-nm detector and an 8-mm × 10-cm column that contains packing L11. The flow rate is about 5 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed under Procedure: the column efficiency determined from the analyte peak is not less than 500 theoretical plates, the tailing factor for the analyte peak is not more than 1.5, and the relative standard deviation for replicate injections is not more than 1.5%.
Procedure—
Separately inject equal volumes (about 50 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatogram, and measure the responses for the major peaks. Calculate the quantity, in mg, of o-iodohippuric acid in each mL of Injection taken by the formula:
C(rU / rS)
in which C is the concentration, in mg per mL, of USP o-Iodohippuric Acid RS in the Standard preparation; and rU and rS are the o-iodohippuric acid peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Assay for radioactivity—
Using a suitable counting assembly (see Selection of a Counting Assembly under Radioactivity  821
821 ), determine the radioactivity in MBq (µCi or mCi) per mL of Injection by use of a calibrated system as directed under Radioactivity
), determine the radioactivity in MBq (µCi or mCi) per mL of Injection by use of a calibrated system as directed under Radioactivity  821
821 .
.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Ian DeVeau, Ph.D.
Director, Veterinary Drugs and Radiopharmaceuticals 1-301-816-8178 |
(RMI05) Radiopharmaceuticals and Medical Imaging Agents 05 |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
|
| Radhakrishna S Tirumalai, Ph.D.
Senior Scientist 1-301-816-8339 |
(MSA05) Microbiology and Sterility Assurance |
USP32–NF27 Page 2653
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.