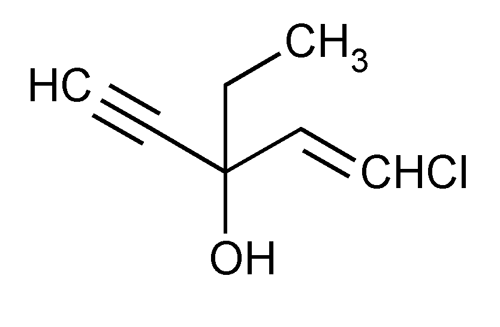
Ethchlorvynol
1-Penten-4-yn-3-ol, 1-chloro-3-ethyl-.
1-Chloro-3-ethyl-1-penten-4-yn-3-ol
» Ethchlorvynol contains not less than 98.0 percent and not more than 100.0 percent of E-ethchlorvynol (C7H9ClO), calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight, light-resistant glass or polyethylene containers, using polyethylene-lined closures.
Identification—
B:
Dissolve about 1 g in 20 mL of methanol. To 1 mL of the solution, add about 4 drops of 6 N ammonium hydroxide, mix, then add silver nitrate TS, a few drops at a time: a yellowish white precipitate is formed, and it at first redissolves, but becomes insoluble when an excess of silver nitrate TS has been added.
C:
To 10 mL of the solution prepared in Identification test B add 5 mL of freshly prepared m-phenylenediamine hydrochloride-oxalic acid solution (prepared by dissolving 1 g of m-phenylenediamine hydrochloride and 1 g of oxalic acid in 35 mL of water and filtering, if necessary): a reddish orange color is produced in about 3 minutes.
Refractive index  831
831 :
between 1.476 and 1.480.
:
between 1.476 and 1.480.
Acidity—
Dissolve 5.0 mL of Ethchlorvynol in 50 mL of a mixture of equal volumes of water and methanol that has been neutralized to the phenolphthalein endpoint with 0.1 N sodium hydroxide. Add 1 mL of phenolphthalein TS, and titrate with 0.10 N sodium hydroxide to a pink endpoint: not more than 1.7 mL of 0.10 N sodium hydroxide is required for neutralization.
Water, Method I  921
921 :
not more than 0.2%.
:
not more than 0.2%.
Chromatographic purity—
Calculate the percentage of each peak, other than the E-ethchlorvynol peak, observed in the chromatogram of the Ethchlorvynol obtained as directed in the Assay taken by the same formula:
100ri / rt
in which ri is the response of each secondary peak and rt is the sum of all of the peaks observed in the chromatogram: not more than 0.2% of toluene, not more than 1.5% of Z-ethchlorvynol, and not more than 0.3% of any other impurity is found, and the total of all observed impurities is not more than 2.0%.
Assay—
Resolution solution—
Add 2.5 µL of toluene to 0.5 mL of USP Ethchlorvynol RS, and mix.
Chromatographic system
(see Chromatography  621
621 )—The gas chromatograph is equipped with a thermal conductivity detector and a 1.8-m × 4-mm glass column (pretreated with 10% dimethyldichlorosilane in toluene) packed with 10% phase G16 on 60- to 80-mesh support S1AB. The column is maintained at about 160
)—The gas chromatograph is equipped with a thermal conductivity detector and a 1.8-m × 4-mm glass column (pretreated with 10% dimethyldichlorosilane in toluene) packed with 10% phase G16 on 60- to 80-mesh support S1AB. The column is maintained at about 160 , and the injector and the detector are maintained at about 200
, and the injector and the detector are maintained at about 200 . The carrier gas is dry helium, flowing at a rate of about 30 mL per minute. Chromatograph the Resolution solution, and record the peak responses as directed for Procedure: the relative retention times for toluene,
. The carrier gas is dry helium, flowing at a rate of about 30 mL per minute. Chromatograph the Resolution solution, and record the peak responses as directed for Procedure: the relative retention times for toluene,  -chlorovinylethyl ketone (if present), Z-ethchlorvynol, and E-ethchlorvynol are about 0.1, 0.2, 0.8, and 1.0, respectively; the resolution, R, between the Z- and E-ethchlorvynol peaks is not less than 1.0; and the relative standard deviation for replicate injections is not more than 2.0%.
-chlorovinylethyl ketone (if present), Z-ethchlorvynol, and E-ethchlorvynol are about 0.1, 0.2, 0.8, and 1.0, respectively; the resolution, R, between the Z- and E-ethchlorvynol peaks is not less than 1.0; and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
[note—Use peak areas where peak responses are indicated.] Inject about 3 µL of Ethchlorvynol into the chromatograph, record the chromatogram, and measure the responses for all of the peaks. Calculate the percentage of E-ethchlorvynol (C7H9ClO) in the specimen of Ethchlorvynol taken by the formula:
100re / rt
in which re is the response of the E-ethchlorvynol peak obtained in the chromatogram of the Ethchlorvynol and rt is the sum of the responses of all of the peaks observed in the chromatogram.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Ravi Ravichandran, Ph.D.
Senior Scientist 1-301-816-8330 |
(MDPP05) Monograph Development-Psychiatrics and Psychoactives |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 2321
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.