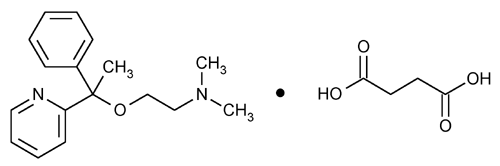
Doxylamine Succinate
Ethanamine, N,N-dimethyl-2-[1-phenyl-1-(2-pyridinyl)ethoxy]-, butanedioate (1:1).
2-[
» Doxylamine Succinate contains not less than 98.0 percent and not more than 101.0 percent of C17H22N2O·C4H6O4, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed, light-resistant containers.
Identification—
A:
Ultraviolet absorption  197U
197U —
—
Solution:
20 µg per mL.
Medium:
0.1 N hydrochloric acid.
Absorptivities at 262 nm, calculated on the dried basis, do not differ by more 3.0%.
B:
It meets the requirements under Identification—Organic Nitrogenous Bases  181
181 .
.
C:
Dissolve about 500 mg in 5 mL of water, and add a slight excess of 6 N ammonium hydroxide. Extract the liberated doxylamine with several portions of ether, discard the ether extracts, and evaporate the aqueous solution on a steam bath to dryness. Add 2 mL of 3 N hydrochloric acid, and again evaporate on the steam bath to dryness. Cool, add about 10 mL of ether, allow to stand for a few minutes, and decant the clear supernatant. Evaporate the ether solution to dryness, and dry the residue at 105 for 30 minutes: the succinic acid so obtained melts between 184
for 30 minutes: the succinic acid so obtained melts between 184 and 188
and 188 , the procedure for Class I being used (see Melting Range or Temperature
, the procedure for Class I being used (see Melting Range or Temperature  741
741 ).
).
Melting range, Class I  741
741 :
between 103
:
between 103 and 108
and 108 , but the range between beginning and end of melting does not exceed 3
, but the range between beginning and end of melting does not exceed 3 .
.
Loss on drying  731
731 —
Dry it in vacuum over phosphorus pentoxide for 5 hours: it loses not more than 0.5% of its weight.
—
Dry it in vacuum over phosphorus pentoxide for 5 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Volatile related compounds—
Dissolve 650 mg in 20 mL of 0.1 N hydrochloric acid in a separator. Render the solution alkaline with 2.5 N sodium hydroxide, and immediately extract with four 25-mL portions of ether, filtering each extract through an ether-saturated pledget of cotton. Evaporate the combined ether extracts on a water bath with the aid of a current of air to dryness at a temperature not exceeding 50 , and dissolve the residue in 5 mL of alcohol. Inject about 1 µL of this solution into a suitable gas chromatograph (see Chromatography
, and dissolve the residue in 5 mL of alcohol. Inject about 1 µL of this solution into a suitable gas chromatograph (see Chromatography  621
621 ) equipped with a flame-ionization detector. Under typical conditions, the instrument contains a 2-m × 4-mm glass column containing 5% packing G16 and 5% packing G12 on 60- to 80-mesh S1A. The column is maintained at about 212
) equipped with a flame-ionization detector. Under typical conditions, the instrument contains a 2-m × 4-mm glass column containing 5% packing G16 and 5% packing G12 on 60- to 80-mesh S1A. The column is maintained at about 212 , the injection port and detector block are maintained at about 250
, the injection port and detector block are maintained at about 250 , and dry helium is used as the carrier gas. The total relative area of all extraneous peaks (except that of the solvent peak) does not exceed 2.0%, and the relative area of any individual extraneous peak does not exceed 1.0%.
, and dry helium is used as the carrier gas. The total relative area of all extraneous peaks (except that of the solvent peak) does not exceed 2.0%, and the relative area of any individual extraneous peak does not exceed 1.0%.
Assay—
Dissolve about 500 mg of Doxylamine Succinate, accurately weighed, in 80 mL of glacial acetic acid. Add crystal violet TS, and titrate with 0.1 N perchloric acid VS to an emerald-green endpoint. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 19.42 mg of C17H22N2O·C4H6O4.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Daniel K. Bempong, Ph.D.
Senior Scientist 1-301-816-8143 |
(MDPS05) Monograph Development-Pulmonary and Steroids |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 2216
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.