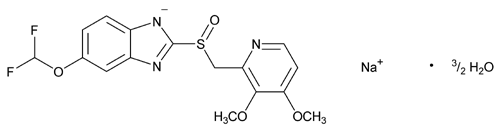
Add the following:
1H-Benzimidazole, 5-(difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridyl)methyl]sulfinyl]-, sodium salt, hydrate (2:3).
5-(Difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridyl)methyl]sulfinyl]benzimidazole, sodium salt, sesquihydrate
» Pantoprazole Sodium contains not less than 98.0 percent and not more than 102.0 percent of C16H14F2N3NaO4S, calculated on the anhydrous basis.
Packaging and storage—
Preserve in well-closed, light-resistant containers. Store at room temperature.
Labeling—
If a test for Related compounds other than Test 1 is used, then the labeling states the test with which the article complies.
USP Reference standards  11
11 —
—
USP Pantoprazole Sodium RS.
USP Pantoprazole Related Compound A RS.
USP Pantoprazole Related Compound B RS.
USP Pantoprazole Related Compound C RS.
USP Pantoprazole Related Compound D and F Mixture RS.
USP Pantoprazole Related Compound E RS.
USP Pantoprazole Sodium RS.
USP Pantoprazole Related Compound A RS.
USP Pantoprazole Related Compound B RS.
USP Pantoprazole Related Compound C RS.
USP Pantoprazole Related Compound D and F Mixture RS.
USP Pantoprazole Related Compound E RS.
Identification—
B:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of the Standard preparation, as obtained in the Assay.
C:
It meets the requirements of the pyroantimonate precipitate test for Sodium  191
191 .
.
Water, Method I  921
921 :
between 5.0% and 8.0%.
:
between 5.0% and 8.0%.
Heavy metals, Method II  231
231 :
not more than 0.002%.
:
not more than 0.002%.
Related compounds—
[Note—On the basis of the synthetic route, perform either Test 1 or Test 2. Test 2 is recommended when impurities C, D, E, and F are potential related compounds.]
test 1—
[Note—Protect all solutions from light, and use amber autosampler vials and low-actinic glassware.]
Diluent, Mobile phase, System suitability preparation, and Chromatographic system—
Proceed as directed in the Assay.
Standard solution—
Transfer about 20 mg of USP Pantoprazole Sodium RS, accurately weighed, to a 50-mL volumetric flask, dissolve in 5 to 10 mL of a mixture of acetonitrile and water (1:1), and dilute with Diluent to volume. Further dilute with Diluent quantitatively, and stepwise if necessary, to obtain a solution having a known concentration of about 0.0004 mg per mL.
Test solution—
Transfer about 20 mg of Pantoprazole Sodium, accurately weighed, to a 50-mL volumetric flask, dissolve in 5 to 10 mL of a mixture of acetonitrile and water (1:1), dilute with Diluent to volume, and mix.
Chromatographic system
(see Chromatography  621
621 )—Prepare as directed in the Assay. Chromatograph the System suitability preparation, and record the peak responses as directed for Procedure. Identify the components on the basis of their relative retention times (Table 1): the resolution, R, between the pantoprazole related compound A and pantoprazole peaks is not less than 10.0.
)—Prepare as directed in the Assay. Chromatograph the System suitability preparation, and record the peak responses as directed for Procedure. Identify the components on the basis of their relative retention times (Table 1): the resolution, R, between the pantoprazole related compound A and pantoprazole peaks is not less than 10.0.
Table 1
| Name | Relative Retention Time |
Limit (%) |
| Pantoprazole related compound A1 | 0.52 | 0.20 |
| Pantoprazole sodium | 1.0 | N/A |
| Pantoprazole related compound B2 | 1.7 | 0.15 |
| Any other individual impurity | — | 0.10 |
| Total impurities | — | 0.5 |
|
1
5-(Difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridyl)methyl]sulfonyl]-1H-benzimidazole.
2
5-(Difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridyl)methyl]thio]-1H-benzimidazole.
|
||
Procedure—
Separately inject equal volumes (about 20 µL) of the Standard solution and the Test solution into the chromatograph, record the chromatograms, and measure the peak responses. Calculate the percentage of each impurity in the portion of Pantoprazole Sodium taken by the formula:
100(CS / CT)(ri / rS)
in which CS and CT are the concentrations, in mg per mL, of pantoprazole sodium in the Standard solution and the Test solution, respectively; ri is the peak response of each impurity obtained from the Test solution; and rS is the pantoprazole peak response obtained from the Standard solution. The reporting level for impurities is 0.05%.
test 2—
Diluent—
Prepare a mixture of acetonitrile and 0.001 N sodium hydroxide solution (50:50).
Standard solution—
Dissolve an accurately weighed quantity of USP Pantoprazole Sodium RS in Diluent, and dilute quantitatively to obtain a solution having a known concentration of about 0.03 mg per mL.
Test solution—
Prepare a solution of Pantoprazole Sodium in Diluent having a known concentration of about 0.46 mg per mL.
System suitability solution—
Dissolve suitable amounts of USP Pantoprazole Sodium RS, USP Pantoprazole Related Compound A RS, USP Pantoprazole Related Compound B RS, USP Pantoprazole Related Compound C RS, USP Pantoprazole Related Compound D and F Mixture RS, and USP Pantoprazole Related Compound E RS in Diluent to obtain a solution containing about 0.46 mg of pantoprazole sodium per mL and about 1.3 µg each of related compounds A, B, C, and E per mL, and about 1.3 µg of the D and F mixture per mL.
Solution A—
Prepare a solution of dibasic potassium phosphate (1.74 g/L) adjusted with a solution of phosphoric acid (330 g/L) to a pH of 7.00 ± 0.05.
Solution B—
Use acetonitrile.
Mobile phase—
Use variable mixtures of Solution A and Solution B as directed below for Chromatographic system. Make adjustments as necessary (see System Suitability under Chromatography  621
621 ).
).
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with either a programmable variable wavelength detector or two separate detectors capable of monitoring at 290 nm and at 305 nm, and a 4-mm × 12.5-cm column that contains 5-µm packing L1. The column temperature is maintained at 40
)—
The liquid chromatograph is equipped with either a programmable variable wavelength detector or two separate detectors capable of monitoring at 290 nm and at 305 nm, and a 4-mm × 12.5-cm column that contains 5-µm packing L1. The column temperature is maintained at 40 . The flow rate is about 1.0 mL per minute. The chromatograph is programmed as follows.
. The flow rate is about 1.0 mL per minute. The chromatograph is programmed as follows.
Chromatograph the System suitability solution, and record the peak responses at 290 nm as directed for Procedure. Identify the components based on relative retention times (Table 2): the resolution, R, between pantoprazole related compound E and pantoprazole related compounds D and F is not less than 1.5. Chromatograph the Standard solution at 290 nm, and record the peak responses as directed for Procedure: the tailing factor is not more than 2; and the relative standard deviation for replicate injections is not more than 5.0%.
| Time (minutes) |
Solution A
(%) |
Solution B
(%) |
|
| 0–40 | 80®20 | 20®80 | linear gradient |
| 40–45 | 20®80 | 80®20 | linear gradient |
| 45–55 | 80 | 20 | re-equilibration |
Procedure—
Separately inject equal volumes (about 20 µL) of the Standard solution and the Test solution into the chromatograph, record the chromatograms at 290 nm and 305 nm, and measure the responses for the major peaks. [Note—Pantoprazole related compound C is monitored using a wavelength of 305 nm, and all other compounds are monitored at 290 nm.] Calculate the percentage of each impurity in the portion of Pantoprazole Sodium taken by the formula:
100 (1 / F)(CS / CU)(ri / rS)
in which CS is the concentration, in mg per mL, of pantoprazole sodium in the Standard solution; CU is the concentration, in mg per mL of Pantoprazole Sodium in the Test solution; F is the response factor of an individual pantoprazole related compound relative to the response of pantoprazole sodium (Table 2); ri is the peak response of each impurity obtained from the Test solution; and rS is the pantoprazole peak response obtained from the Standard solution. The reporting level for impurities is 0.05%.
Table 2
| Impurity Name | Relative Retention Time |
Relative Response Factor (F) |
Limit (%) |
| Related compound A | 0.9 | 1.0 | 0.20 |
| Related compound B | 1.5 | 1.0 | 0.15 |
| Related compound C1 | 0.6 | 3.3 | 0.102 |
| Related compounds D3 and F5 | 1.2 | 1.0 | 0.204 |
| Related compound E6 | 1.3 | 1.0 | 0.10 |
| Any other individual impurity |
— | 0.10 | |
| Total impurities | — | 0.5 | |
|
1
5-(Difluoromethoxy)-1H-benzimidazole-2-thiol.
2
At 305 nm.
3
5-(Difluoromethoxy)-2-[(RS)-[(3,4-dimethoxypyridin-2-yl)methyl]sulfinyl]-1-methyl-1H-benzimidazole.
4
Impurities D and F are not fully resolved and should be integrated together.
5
6-(Difluoromethoxy)-2-[(RS)-[(3,4-dimethoxypyridin-2-yl)methyl]sulfinyl]-1-methyl-1H-benzimidazole.
6
Mixture of the stereoisomers of 6,6¢-bis(difluoromethoxy)-2,2¢-bis[[(3,4-dimethoxypyridin-2-yl)methyl]sulfinyl]-1H,1¢H-5,5¢-bibenzimidazolyl.
|
|||
Assay—
[Note—Protect all solutions from light, and use amber autosampler vials and low-actinic glassware.]
Ammonium phosphate buffer—
Dissolve 1.32 g of dibasic ammonium phosphate in 1000 mL of water. Adjust with phosphoric acid to a pH of 7.5.
Acetonitrile–methanol mixture—
Prepare a mixture of acetonitrile and methanol (7:3).
Solution A—
Use a filtered and degassed mixture of Ammonium phosphate buffer and Acetonitrile–methanol mixture (85:15).
Solution B—
Use Acetonitrile–methanol mixture.
Diluent—
Transfer 25 mL of ammonium hydroxide to a suitable container, and dilute with water to 500 mL.
Mobile phase—
Use variable mixtures of Solution A and Solution B as directed for Chromatographic system. Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
System suitability preparation—
Dissolve suitable amounts of USP Pantoprazole Sodium RS, USP Pantoprazole Related Compound A RS, and USP Pantoprazole Related Compound B RS in a mixture of acetonitrile and water (1:1) to obtain a solution having about 0.5 mg of each component per mL. Transfer 1 mL of this solution to a 100-mL volumetric flask, and dilute with Diluent to volume.
Standard preparation—
Transfer about 20 mg of USP Pantoprazole Sodium RS, accurately weighed, to a 50-mL volumetric flask, dissolve in 5 to 10 mL of a mixture of acetonitrile and water (1:1), and dilute with Diluent to volume. Further dilute with Diluent quantitatively, and stepwise if necessary, to obtain a solution having a known concentration of about 0.06 mg per mL.
Assay preparation—
Transfer about 20 mg of Pantoprazole Sodium, accurately weighed, to a 50-mL volumetric flask, dissolve in 5 to 10 mL of a mixture of acetonitrile and water (1:1), and dilute with Diluent to volume. Further dilute with Diluent quantitatively, and stepwise if necessary, to obtain a solution having a known concentration of about 0.06 mg per mL.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 285-nm detector and 3.9-mm × 15-cm column that contains 4-µm packing L1. The flow rate is about 1 mL per minute. The column temperature is maintained at 30
)—
The liquid chromatograph is equipped with a 285-nm detector and 3.9-mm × 15-cm column that contains 4-µm packing L1. The flow rate is about 1 mL per minute. The column temperature is maintained at 30 , and the autosampler temperature is maintained at 4
, and the autosampler temperature is maintained at 4 . The chromatograph is programmed as follows:
. The chromatograph is programmed as follows:
Chromatograph the System suitability preparation, and record the peak responses as directed for Procedure. Identify the components based on their relative retention times (Table 1): the resolution, R, between the pantoprazole related compound A and pantoprazole peaks is not less than 10.0. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the relative standard deviation for replicate injections is not more than 2.0%.
| Time (minutes) |
Solution A
(%) |
Solution B
(%) |
|
| 0–10 | 86 | 14 | isocratic |
| 10–35 | 86®42 | 14®58 | linear gradient |
| 35–36 | 42®86 | 58®14 | linear gradient |
| 36–46 | 86 | 14 | re-equilibration |
Procedure—
Separately inject equal volumes (about 20 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the percentage of C16H14F2N3NaO4S in the portion of Pantoprazole Sodium taken by the formula:
 3
3
100(CS / CU)(rU / r S)
in which CS and CU are the concentrations, in mg per mL, of pantoprazole sodium in the Standard preparation and the Assay preparation, respectively; and rU and rS are the peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Elena Gonikberg, Ph.D.
Senior Scientist 1-301-816-8251 |
(MDGRE05) Monograph Development-Gastrointestinal Renal and Endocrine |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3200
Pharmacopeial Forum: Volume No. 33(6) Page 1194
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.