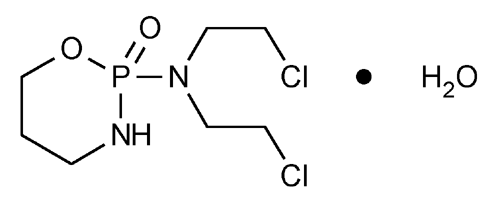
Cyclophosphamide
2H-1,3,2-Oxazaphosphorin-2-amine, N,N-bis(2-chloroethyl)tetrahydro-, 2-oxide, monohydrate, (±).
(±)-2-[Bis(2-chloroethyl)amino]tetrahydro-2H-1,3,2-oxazaphosphorine 2-oxide monohydrate
Anhydrous 261.09
» Cyclophosphamide contains not less than 97.0 percent and not more than 103.0 percent of C7H15Cl2N2O2P, calculated on the anhydrous basis.
Caution—Great care should be taken in handling Cyclophosphamide, as it is a potent cytotoxic agent.
Packaging and storage—
Preserve in tight containers, at a temperature between 2 and 30
and 30 .
.
Identification—
B:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that of the Standard preparation, both relative to the internal standard, as obtained in the Assay.
pH  791
791 :
between 3.9 and 7.1, in a solution (1 in 100), determined 30 minutes after its preparation.
:
between 3.9 and 7.1, in a solution (1 in 100), determined 30 minutes after its preparation.
Water, Method I  921
921 :
between 5.7% and 6.8%.
:
between 5.7% and 6.8%.
Heavy metals  231
231 —
Dissolve 1.0 g in 25 mL of water, and filter if necessary: the limit is 0.002%.
—
Dissolve 1.0 g in 25 mL of water, and filter if necessary: the limit is 0.002%.
Assay—
Mobile phase—
Prepare a suitable, degassed solution of water and acetonitrile (70:30).
Internal standard solution—
Dissolve about 185 mg of ethylparaben in 250 mL of alcohol in a 1000-mL volumetric flask, dilute with water to volume, and mix.
Standard preparation—
Transfer an accurately weighed quantity of USP Cyclophosphamide RS, equivalent to about 25 mg of anhydrous cyclophosphamide, to a 50-mL volumetric flask, add about 25 mL of water, and shake to dissolve the USP Reference Standard. Add 5.0 mL of Internal standard solution, dilute with water to volume, and mix to obtain a Standard preparation having a known concentration of about 0.5 mg of anhydrous cyclophosphamide per mL.
Assay preparation—
Transfer an accurately weighed quantity of Cyclophosphamide, equivalent to about 200 mg of anhydrous cyclophosphamide, to a 200-mL volumetric flask, add about 50 mL of water, shake for about 5 minutes, dilute with water to volume, and mix. Pipet 25 mL of this solution and 5 mL of Internal standard solution into a 50-mL volumetric flask, dilute with water to volume, and mix.
Chromatographic system
(see Chromatography  621
621 )—The liquid chromatograph is equipped with a 195-nm detector and a 3.9-mm × 30-cm column that contains packing L1. The flow rate is about 1.5 mL per minute. Chromatograph six replicate injections of the Standard preparation, and record the peak responses as directed for Procedure: the relative standard deviation is not more than 2%, and the resolution factor between cyclophosphamide and ethylparaben is not less than 2.
)—The liquid chromatograph is equipped with a 195-nm detector and a 3.9-mm × 30-cm column that contains packing L1. The flow rate is about 1.5 mL per minute. Chromatograph six replicate injections of the Standard preparation, and record the peak responses as directed for Procedure: the relative standard deviation is not more than 2%, and the resolution factor between cyclophosphamide and ethylparaben is not less than 2.
Procedure—
Separately inject equal volumes (about 25 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. The relative retention times are about 0.7 for cyclophosphamide and 1.0 for ethylparaben. Calculate the quantity, in mg, of C7H15Cl2N2O2P in the Cyclophosphamide taken by the formula:
400C(RU / RS)
in which C is the concentration, in mg per mL, of anhydrous cyclophosphamide in the Standard preparation, as determined from the concentration of USP Cyclophosphamide RS corrected for moisture content by a titrimetric water determination; and RU and RS are the ratios of the peak responses of cyclophosphamide to those of ethylparaben in the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Feiwen Mao, M.S.
Scientist 1-301-816-8320 |
(MDOOD05) Monograph Development-Ophthalmics Oncologics and Dermatologicals |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 2040
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.