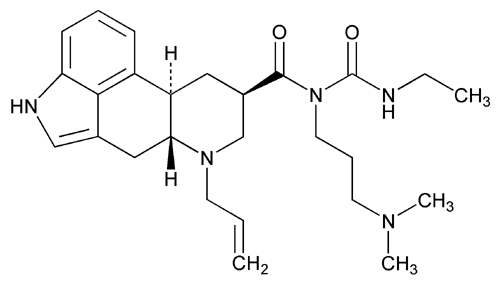
Add the following:
Ergoline-8
1-[(6-Allylergolin-8
» Cabergoline contains not less than 98.0 percent and not more than 102.0 percent of C26H37N5O2, calculated on the anhydrous basis.
Packaging and storage—
Store in tight containers, protected from light.
Identification—
B:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the Standard preparation, as obtained in the Assay.
Specific rotation  781S
781S :
between –77
:
between –77 and –83
and –83 .
.
Test solution—
Dissolve Cabergoline in alcohol to obtain a solution having a known concentration of about 1 mg per mL, calculated on the anhydrous basis.
Water, Method I  921
921 :
not more than 0.5%.
:
not more than 0.5%.
Related compounds—
[note—Prepare solutions immediately before use, and protect from light.]
Mobile phase—
Prepare as directed in the Assay.
Resolution solution—
To 10 mL of 0.1 M sodium hydroxide add 50 mg of Cabergoline. Stir for about 15 minutes. To 1 mL of the suspension add 1 mL of 0.1 M hydrochloric acid, and dilute with Mobile phase to 10 mL. Sonicate until dissolution is complete. The main degradation product obtained is cabergoline related compound A.
Test solution—
Use the Assay preparation, prepared as directed in the Assay.
Chromatographic system
(see Chromatography  621
621 )—Prepare as directed in the Assay. Chromatograph the Resolution solution, and record the peak responses as directed for Procedure. Identify peaks due to cabergoline related compound A and cabergoline using the relative retention times given in Table 1; the resolution, R, between cabergoline and cabergoline related compound A is not less than 3.0.
)—Prepare as directed in the Assay. Chromatograph the Resolution solution, and record the peak responses as directed for Procedure. Identify peaks due to cabergoline related compound A and cabergoline using the relative retention times given in Table 1; the resolution, R, between cabergoline and cabergoline related compound A is not less than 3.0.
Table 1
| Name | Relative Retention Time | Limit (%) |
| Cabergoline related compound D1 | 0.3 | NMT 0.1 |
| Cabergoline related compound B2 | 0.6 | NMT 0.1 |
| Cabergoline related compound A3 | 0.8 | NMT 0.3 |
| Cabergoline | 1.0 | — |
| Cabergoline related compound C4 | 2.9 | NMT 0.3 |
| Any unspecified impurity | — | NMT 0.10 |
| Total | — | NMT 0.8 |
|
1
(6aR,9R,10aR)-N-[3-(Dimethylamino)propyl]-7-(prop-2-enyl)-4,6,6a,7,8,9,10,10a-octahydroindolo[4,3-fg]quinoline-9-carboxamide.
2
(6aR,9R,10aR)-N9-[3-(Dimethylamino)propyl]-N4-ethyl-7-(prop-2-enyl)-6a,7,8,9,10,10a-hexahydroindolo[4,3-fg]quinoline-4,9(6H)-dicarboxamide.
3
(6aR,9R,10aR)-7-(Prop-2-enyl)-4,6,6a,7,8,9,10,10a-octahydroindolo[4,3-fg]quinoline-9-carboxylic acid.
4
(6aR,9R,10aR)-N9-[3-(Dimethylamino)propyl]-N4-ethyl-N9-(ethylcarbamoyl)-7-(prop-2-enyl)-6a,7,8,9,10,10a-hexahydroindolo[4,3-fg]quinoline-4,9(6H)-dicarboxamide.
|
||
Procedure—
Inject about 100 µL of the Test solution into the chromatograph, and record the chromatogram. Calculate the percentage of each impurity in the portion of Cabergoline taken by the formula:
100(ri / rs)
in which ri is the peak area of each impurity peak found in the Test solution; and rs is the sum of the peak areas of all the impurities and the main peak due to cabergoline in the Test solution. The relative retention times and the limits of cabergoline related compounds A, B, C, and D and their corresponding limits are given in Table 1.
Assay—
[note—Prepare solutions immediately before use, and protect from light.]
Buffer—
Transfer 6.8 g of monobasic potassium phosphate to a 1-L volumetric flask. Dissolve the contents in 900 mL of water. Adjust with phosphoric acid to a pH of 2.0. Dilute with water to volume, add 0.2 mL of triethylamine, and mix well.
Mobile phase—
Prepare a mixture of Buffer and acetonitrile (84:16), and degas. Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
Standard preparation—
Dissolve an accurately weighed quantity of USP Cabergoline RS in Mobile phase to obtain a solution having a known concentration of about 0.25 mg per mL. [note—Sonication may be used to aid in the dissolution.]
Assay preparation—
Dissolve an accurately weighed quantity of Cabergoline in Mobile phase to obtain a solution having a nominal concentration of about 0.25 mg per mL. [note—Sonication may be used to aid in the dissolution.]
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 280-nm detector and a 4.0-mm × 25-cm column that contains 10-µm packing L1. The flow rate is about 1.3 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the column efficiency is not less than 1000 theoretical plates; and the relative standard deviation for five replicate injections is not more than 2.0% for the cabergoline peak.
)—
The liquid chromatograph is equipped with a 280-nm detector and a 4.0-mm × 25-cm column that contains 10-µm packing L1. The flow rate is about 1.3 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the column efficiency is not less than 1000 theoretical plates; and the relative standard deviation for five replicate injections is not more than 2.0% for the cabergoline peak.
Procedure—
Inject equal volumes (about 100 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the peak responses. Calculate the percentage of C26H37N5O2 in the portion of Cabergoline taken by the formula:
 USP32
USP32
100(CS / CU)(rU / rS)
in which CS is the concentration, in mg per mL, of cabergoline in the Standard preparation; CU is the nominal concentration, in mg per mL, of Cabergoline in the Assay preparation; and rU and rS are the peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Ravi Ravichandran, Ph.D.
Senior Scientist 1-301-816-8330 |
(MDPP05) Monograph Development-Psychiatrics and Psychoactives |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 1740
Pharmacopeial Forum: Volume No. 34(1) Page 75
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.