- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Pentobarbital Sodium |
 |
(Ph. Eur. monograph 0419)
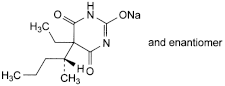
C11H17N2NaO3 248.3 57-33-0
Barbiturate.
Ph Eur
Pentobarbital sodium contains not less than 99.0 per cent and not more than the equivalent of 101.5 per cent of the sodium derivative of 5-ethyl-5-[(1RS)-1-methylbutyl]pyrimidine-2,4,6(1H,3H,5H)-trione, calculated with reference to the dried substance.
A white or almost white, crystalline powder, hygroscopic, very soluble in water.
A. Dissolve 1 g in 10 mL of water R and add 5 mL of dilute acetic acid R. A white, crystalline precipitate is formed. Filter, wash the precipitate with water R and dry at 100 °C to 105 °C. Determine the melting point (2.2.14) of the precipitate. Mix equal parts of the precipitate and pentobarbital CRS and determine the melting point of the mixture. The difference between the melting points (which are about 131 °C) is not greater than 2 °C.
B. Examine by thin-layer chromatography (2.2.27), using a TLC silica gel GF254 plate R.
Test solution Dissolve 25 mg of the precipitate obtained in identification test A in alcohol R and dilute to 25 mL with the same solvent.
Reference solution Dissolve 25 mg of pentobarbital CRS in alcohol R and dilute to 25 mL with the same solvent.
Apply to the plate 10 µL of each solution. Develop over a path of 18 cm using the lower layer from a mixture of 5 volumes of concentrated ammonia R, 15 volumes of alcohol R and 80 volumes of chloroform R. Examine immediately in ultraviolet light at 254 nm. The principal spot in the chromatogram obtained with the test solution is similar in position and size to the principal spot in the chromatogram obtained with the reference solution.
C. To about 10 mg add about 10 mg of vanillin R and 2 mL of sulfuric acid R. Mix and heat on a water-bath for 2 min. A reddish-brown colour develops. Cool and add cautiously 5 mL of ethanol R. The colour becomes violet and then blue.
D. Ignite 1 g. The residue gives reaction (a) of sodium (2.3.1).
Dissolve 1.0 g in carbon dioxide-free water R and dilute to 10 mL with the same solvent. The pH measured immediately after preparation of the solution is 9.6 to 11.0.
Examine by thin-layer chromatography (2.2.27), using a TLC silica gel GF254 plate R.
Test solution Dissolve 0.2 g of the substance to be examined in alcohol R and dilute to 10 mL with the same solvent.
Reference solution Dilute 0.5 mL of the test solution to 100 mL with alcohol R.
Apply to the plate 10 µL of each solution. Develop over a path of 15 cm using the lower layer from a mixture of 5 volumes of concentrated ammonia R, 15 volumes of alcohol R and 80 volumes of chloroform R. Examine immediately in ultraviolet light at 254 nm. Any spot in the chromatogram obtained with the test solution, apart from the principal spot, is not more intense than the spot in the chromatogram obtained with the reference solution (0.5 per cent). Spray with diphenylcarbazone mercuric reagent R. Allow the plate to dry in air and spray with freshly prepared alcoholic potassium hydroxide solution R diluted 1 in 5 with aldehyde-free alcohol R. Heat at 100 °C to 105 °C for 5 min and examine immediately in daylight. Any spot in the chromatogram obtained with the test solution, apart from the principal spot, is not more intense than the spot in the chromatogram obtained with the reference solution (0.5 per cent).
Not more than 3.5 per cent. Dissolve 2.00 g in 75 mL of dimethylformamide R, heating gently if necessary. Titrate with 0.1 M sodium methoxide until the colour changes from olive-green to blue, using 0.25 mL of a 10 g/L solution of thymol blue R in dimethylformamide R as indicator. Carry out a blank titration.
1 mL of 0.1 M sodium methoxide is equivalent to 22.63 mg of pentobarbital.
Dissolve 0.3 g in 5 mL of a 50 g/L solution of anhydrous sodium carbonate R. Add a solution of 0.3 g of nitrobenzyl chloride R in 10 mL of alcohol R and heat under a reflux condenser for 30 min. Cool to 25 °C, if necessary scratching the wall of the container with a glass rod to induce crystallisation. Filter and wash the precipitate with five quantities, each of 5 mL, of water R. In a small flask, heat the precipitate with 25 mL of alcohol R under a reflux condenser until dissolved (about 10 min). Cool to 25 °C, if necessary scratching the wall of the flask with a glass rod to induce crystallisation, and filter. The precipitate, washed with two quantities, each of 5 mL, of water R and dried at 100 °C to 105 °C for 30 min, melts (2.2.14) at 136 °C to 148 °C.
Dissolve 1.0 g in water R and dilute to 10.0 mL with the same solvent. To 9 mL of the solution, add 3 mL of dilute acetic acid R and 3 mL of buffer solution pH 3.5 R and filter. Dilute the filtrate to 18 mL with water R. 12 mL of the solution complies with limit test A for heavy metals (20 ppm). In preparing the test solution, replace the buffer solution with water R. Prepare the standard using lead standard solution (1 ppm Pb) R.
Not more than 3.0 per cent, determined on 1.00 g by drying in an oven at 105 °C.
Dissolve 0.200 g in 15 mL of a 127.5 g/L solution of silver nitrate R in pyridine R. Titrate with 0.1 M ethanolic sodium hydroxide until a pure blue colour is obtained, using 0.5 mL of thymolphthalein solution R as indicator. Carry out a blank titration.
1 mL of 0.1 M ethanolic sodium hydroxide is equivalent to 24.83 mg of C11H17N2NaO3.
Store in an airtight container.
Ph Eur