Calcium Silicate
» Calcium Silicate crystalline or amorphous, is a compound of calcium oxide and silicon dioxide. It contains not less than 4.0 percent of calcium oxide and not less than 35.0 percent of silicon dioxide.
Packaging and storage—
Preserve in well-closed containers.
Labeling—
The labeling states the claimed percentage or range of percentages for the content of calcium oxide and for the content of silicon dioxide.
Identification—
A:
Mix 0.5 g with 10 mL of 3 N hydrochloric acid, filter, and neutralize the filtrate to litmus paper with 6 N ammonium hydroxide: the neutralized filtrate so obtained responds to the tests for Calcium  191
191 .
.
B:
Prepare a bead by fusing a few crystals of sodium ammonium phosphate on a platinum loop in the flame of a gas burner. Place the hot, transparent bead in contact with the specimen of Calcium Silicate, and again fuse. Silica floats about in the bead, producing, upon cooling, an opaque bead having a web-like structure.
pH  791
791 :
between 8.4 and 11.2, determined in a well-mixed aqueous suspension (1 in 20).
:
between 8.4 and 11.2, determined in a well-mixed aqueous suspension (1 in 20).
Loss on ignition  733
733 —
Transfer about 1 g, accurately weighed, to a suitable tared crucible, dry at 105
—
Transfer about 1 g, accurately weighed, to a suitable tared crucible, dry at 105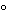 for 2 hours, and ignite at 900
for 2 hours, and ignite at 900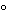 to constant weight: it loses not more than 20.0% of its weight.
to constant weight: it loses not more than 20.0% of its weight.
Heavy metals  231
231 —
Boil 4.0 g with a mixture of 50 mL of water and 10 mL of hydrochloric acid for 20 minutes, adding water to maintain the volume during the boiling. Add ammonium hydroxide until the mixture is only slightly acid to litmus paper. Filter with the aid of suction, and wash with 15 mL to 20 mL of water, combining the washing with the original filtrate. Add 2 drops of phenolphthalein TS, then add a slight excess of 6 N ammonium hydroxide. Discharge the pink color with dilute hydrochloric acid (1 in 100). Dilute with water to 100 mL, and use 25 mL of the solution for the test: the limit is 20 µg per g.
—
Boil 4.0 g with a mixture of 50 mL of water and 10 mL of hydrochloric acid for 20 minutes, adding water to maintain the volume during the boiling. Add ammonium hydroxide until the mixture is only slightly acid to litmus paper. Filter with the aid of suction, and wash with 15 mL to 20 mL of water, combining the washing with the original filtrate. Add 2 drops of phenolphthalein TS, then add a slight excess of 6 N ammonium hydroxide. Discharge the pink color with dilute hydrochloric acid (1 in 100). Dilute with water to 100 mL, and use 25 mL of the solution for the test: the limit is 20 µg per g.
Limit of fluoride—
note—Store all solutions in polytef containers.
Buffer solution—
Transfer 147 g of sodium citrate to a 500-mL volumetric flask, dissolve in and dilute with water to volume.
Ionic strength adjustment buffer—
Transfer 42 mL of hydrochloric acid, 121 g of tris(hydroxymethyl)aminomethane, and 115 g of sodium tartrate to a 500-mL volumetric flask containing 250 mL of water. Stir to dissolve, and dilute with water to volume.
Electrode system—
Use a fluoride-specific, ion-indicating electrode and a suitable reference electrode connected to a pH meter capable of measuring potentials with a reproducibility of ±0.2 mV (see pH  791
791 ).
).
Standard stock solution—
Dissolve an accurately weighed quantity of USP Sodium Fluoride RS in water to obtain a solution containing 221 µg per mL. Each mL of this stock solution contains 100 µg of fluoride ion.
Test solution—
Transfer about 2.0 g of Calcium Silicate, accurately weighed, to a 100-mL polytef beaker containing a magnetic stir bar. Add 20 mL of water and 2.0 mL of hydrochloric acid. Cover with a watch glass, and heat to a vigorous boil for 1 minute, stirring continuously. Remove from heat, and cool. Transfer the cooled suspension to a 100-mL polytef beaker. Add 25 mL of Buffer solution, and adjust with ammonium hydroxide or hydrochloric acid to a pH between 5 and 6. Add 50 mL of Ionic strength adjustment buffer and water to make 100 mL of solution.
Standard response line—
Obtain a standard response line with four standard solutions containing 0, 0.10, 0.20, and 0.40 µg per mL of fluoride as follows. Add 23 mL of water, 2 mL of hydrochloric acid, and 25 mL of Buffer solution to a 100-mL polytef beaker. Adjust with ammonium hydroxide to a pH between 5 and 6, and add Ionic strength adjustment buffer to obtain 100 mL of solution. Insert the electrode into the solution, stir for at least 15 minutes, and record the potential for the standard solution containing 0 µg of fluoride per mL. When the electrode has stabilized, add 100 µL of the Standard stock solution to the beaker, and stir. Allow the electrode to stabilize for 5 minutes, and measure the potential for the standard solution containing 0.10 µg of fluoride per mL. Similarly add another 100 µL and 200 µL of the Standard stock solution and record the potential for the standard solutions containing 0.20 µg per mL of fluoride and 0.40 µg per mL of fluoride, respectively. After each addition, continue to stir for 5 minutes before recording the reading.
Procedure—
Insert the calibrated electrode into the Test solution, stir for 5 minutes, and record the measurement. From the measured potential of the Test solution and the Standard response line, determine the concentration, C, in µg per mL, of fluoride ion in the Test solution. Calculate the quantity, in µg per g, of fluoride in Calcium Silicate by the formula:
100C/W
in which W is the weight, in g, of Calcium Silicate taken. The limit is 50 µg per g.
Limit of lead—
Palladium matrix modifier—
Palladium nitrate (1% PD).1
Magnesium matrix modifier—
Magnesium nitrate (2% Mg).2
Lead standard solution—
A solution containing Pb(NO3)2 in 0.5 M nitric acid corresponding to 1000 mg of lead/mL.3
Nitric acid, 65 percent—
[7697-37-2]—Use a suitable grade with a content of not less than 65.0%.4
Nitric acid diluent—
Transfer 42 mL of nitric acid, 65 percent to a 1000-mL volumetric flask. Dilute with water to volume, and mix.
Test solution—
Accurately weigh 2.0 g of Calcium Silicate into a 150-mL beaker. Mix with 50 mL of Nitric acid diluent. Cover with a watch glass, and boil for 20 minutes. Allow to cool. With the aid of a vacuum, pass through a glass filter5 and wash the filter several times with water. Transfer the filtrate into a 100-mL volumetric flask. Dilute with water to volume, and mix.
Matrix modifier solution—
Transfer 1.0 mL of palladium matrix modifier and 100 uL of magnesium matrix modifier to a 20-mL volumetric flask. Dilute with water to volume, and mix.
Standard lead solution—
Transfer 100 uL of lead standard solution to a 100-mL volumetric flask. Dilute with water to volume, and mix. This solution contains the equivalent of 1.0 µg of lead per mL.
Calibration solutions—
To a series of 100-mL volumetric flasks, pipette 0, 1, and 5 mL of the Standard lead solution, and dilute with 50 mL of Nitric acid diluent. Dilute with water to volume, and mix. These solutions contain 0, 0.01, and 0.05 µg of lead per mL, respectively.
System suitability solution—
Transfer 5 mL of Standard lead solution and 50 mL of Nitric acid diluent into a 100-mL volumetric flask. Dilute with water to volume, and mix. This solution contains the equivalent of 0.05 µg of lead per mL.
Procedure—
Concomitantly determine the absorbances of the Calibration solutions, Test solution, and System suitability solution at the lead emission line at 283.3 nm with a suitable graphite furnace atomic absorption spectrophotometer equipped with a lead hollow-cathode lamp using argon as the carrier gas and an adequate means of Zeeman background correction. Under typical conditions, the Test solution and Calibration solutions volumes are 20 µL, the volume of the Matrix modifier solution is 10 µL, the injection temperature is 20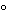 , and the oven conditions are as follows (see Table 1). [note—These conditions may be optimized for each instrument.]
, and the oven conditions are as follows (see Table 1). [note—These conditions may be optimized for each instrument.]
Run the calibration curve; the correlation coefficient is not less than 0.99; and the recovery for the System suitability solution is between 85% and 115%. Calculate the percentage of lead concentration in the Test solution: not more than 0.001%, is found.
Table 1
| Step | Temperature |
| Drying 1 | 110 |
| Drying 2 | 130 |
| Pyrrolysis | 950 |
| Read | 1800 |
| Clean out | 2450 |
Assay for silicon dioxide—
Transfer the appropriate amount of Calcium Silicate (see Table 2) accurately weighed, to a beaker, add 5 mL of water and 10 mL of perchloric acid, and heat until dense white fumes of perchloric acid are evolved. Cover the beaker with a watch glass, and continue to heat for 2 hours. Allow to cool, add 30 mL of water, filter, and wash the precipitate with 200 mL of hot water. [note—Retain the combined filtrate and washings for use in the Assay for calcium oxide.] Transfer the filter paper and its contents to a platinum crucible, heat slowly to dryness, then heat sufficiently to char the filter paper, and ignite at about 900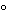 to 1000
to 1000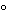 to constant weight. Moisten the residue with 5 drops of perchloric acid, add 15 mL of hydrofluoric acid, heat cautiously on a hot plate until all of the acid is driven off, and ignite at a temperature not lower than 1000
to constant weight. Moisten the residue with 5 drops of perchloric acid, add 15 mL of hydrofluoric acid, heat cautiously on a hot plate until all of the acid is driven off, and ignite at a temperature not lower than 1000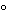 to constant weight. Cool in a desiccator, and weigh: the loss in weight represents the weight of SiO2. The percentage of silicon dioxide in the Calcium Silicate is between 90.0% and 110.0% of the content stated in the labeling, or within the range of percentages stated in the labeling.
to constant weight. Cool in a desiccator, and weigh: the loss in weight represents the weight of SiO2. The percentage of silicon dioxide in the Calcium Silicate is between 90.0% and 110.0% of the content stated in the labeling, or within the range of percentages stated in the labeling.
Table 2
| Sample Weight | Calcium Oxide Content |
| about 400 mg | greater than 25% |
| about 600 mg | 11%–25% |
| about 1000 mg | 4%–10% |
Assay for calcium oxide—
Neutralize the combined filtrate and washings retained from the Assay for silicon dioxide to litmus with 1 N sodium hydroxide. Add, while stirring, about 10 mL of 0.05 M edetate disodium VS from a 50-mL buret. Add 15 mL of 1 N sodium hydroxide and 300 mg of hydroxy naphthol blue, and continue the titration to a blue endpoint. Each mL of 0.05 M edetate disodium is equivalent to 2.804 mg of CaO. The percentage of CaO in the Calcium Silicate is between 90.0% and 110.0% of the content stated in the labeling, or within the range of percentages stated in the labeling.
Ratio of silicon dioxide to calcium oxide—
Divide the percentage of silicon dioxide obtained in the Assay for silicon dioxide by the percentage of calcium oxide obtained in the Assay for calcium oxide: the quotient obtained is between 0.5 and 20.
Sum of CaO, SiO2, and Loss on ignition—
The sum of the percentages obtained in the three tests is not less than 90.0%.
1
A suitable grade is available as catalog number RCMMPD10KN-50 from VWR; www.vwr.com
2
Available as catalog number RCMMMG20KN-50, from VWR, www.vwr.com.
3
A suitable grade is available as catalog number 1.19776 from EMD Chemicals, www.emdchemicals.com.
4
A suitable grade is available as catalog number 441-2 from EMD Chemicals, www.emdchemicals.com.
5
Glass filter: Whatman GF/B glass microfiber filters, 1.0 µm, Whatman no. 1821-090, is suitable. Available as catalog number 28497-492, from VWR, www.vwr.com.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Robert H. Lafaver, B.A.
Scientist 1-301-816-8335 |
(EM105) Excipient Monographs 1 |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 1180
Pharmacopeial Forum: Volume No. 33(3) Page 484
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.