ULTRAVIOLET, VISIBLE, INFRARED, ATOMIC ABSORPTION, FLUORESCENCE, TURBIDIMETRY, NEPHELOMETRY, AND RAMAN MEASUREMENT
Absorption spectrophotometry is the measurement of an interaction between electromagnetic radiation and the molecules, or atoms, of a chemical substance. Techniques frequently employed in pharmaceutical analysis include UV, visible, IR, and atomic absorption spectroscopy. Spectrophotometric measurement in the visible region was formerly referred to as colorimetry; however, it is more precise to use the term “colorimetry” only when considering human perception of color.
Fluorescence spectrophotometry is the measurement of the emission of light from a chemical substance while it is being exposed to UV, visible, or other electromagnetic radiation. In general, the light emitted by a fluorescent solution is of maximum intensity at a wavelength longer than that of the exciting radiation, usually by some 20 to 30 nm.
Light-Scattering involves measurement of the light scattered because of submicroscopic optical density inhomogeneities of solutions and is useful in the determination of weight-average molecular weights of polydisperse systems in the molecular weight range from 1000 to several hundred million. Two such techniques utilized in pharmaceutical analysis are turbidimetry and nephelometry.
Raman spectroscopy (inelastic light-scattering) is a light-scattering process in which the specimen under examination is irradiated with intense monochromatic light (usually laser light) and the light scattered from the specimen is analyzed for frequency shifts.
The wavelength range available for these measurements extends from the short wavelengths of the UV through the IR. For convenience of reference, this spectral range is roughly divided into the UV (190 to 380 nm), the visible (380 to 780 nm), the near-IR (780 to 3000 nm), and the IR (2.5 to 40 µm or 4000 to 250 cm 1).
1).
COMPARATIVE UTILITY OF SPECTRAL RANGES
For many pharmaceutical substances, measurements can be made in the UV and visible regions of the spectrum with greater accuracy and sensitivity than in the near-IR and IR. When solutions are observed in 1-cm cells, concentrations of about 10 µg of the specimen per mL often will produce absorbances of 0.2 to 0.8 in the UV or the visible region. In the IR and near-IR, concentrations of 1 to 10 mg per mL and up to 100 mg per mL, respectively, may be needed to produce sufficient absorption; for these spectral ranges, cell lengths of from 0.01 mm to upwards of 3 mm are commonly used.
The UV and visible spectra of substances generally do not have a high degree of specificity. Nevertheless, they are highly suitable for quantitative assays, and for many substances they are useful as additional means of identification.
There has been increasing interest in the use of near-IR spectroscopy in pharmaceutical analysis, especially for rapid identification of large numbers of samples, and also for water determination.
The near-IR region is especially suitable for the determination of –OH and –NH groups, such as water in alcohol, –OH in the presence of amines, alcohols in hydrocarbons, and primary and secondary amines in the presence of tertiary amines.
The IR spectrum is unique for any given chemical compound with the exception of optical isomers, which have identical spectra. However, polymorphism may occasionally be responsible for a difference in the IR spectrum of a given compound in the solid state. Frequently, small differences in structure result in significant differences in the spectra. Because of the large number of maxima in an IR absorption spectrum, it is sometimes possible to quantitatively measure the individual components of a mixture of known qualitative composition without prior separation.
The Raman spectrum and the IR spectrum provide similar data, although the intensities of the spectra are governed by different molecular properties. Raman and IR spectroscopy exhibit different relative sensitivities for different functional groups, e.g., Raman spectroscopy is particularly sensitive to C–S and C–C multiple bonds, and some aromatic compounds are more easily identified by means of their Raman spectra. Water has a highly intense IR absorption spectrum, but a particularly weak Raman spectrum. Therefore, water has only limited IR “windows” that can be used to examine aqueous solutes, while its Raman spectrum is almost completely transparent and useful for solute identification. The two major limitations of Raman spectroscopy are that the minimum detectable concentration of specimen is typically 10 1 M to 10
1 M to 10 2 M and that the impurities in many substances fluoresce and interfere with the detection of the Raman scattered signal.
2 M and that the impurities in many substances fluoresce and interfere with the detection of the Raman scattered signal.
Optical reflectance measurements provide spectral information similar to that obtained by transmission measurements. Since reflectance measurements probe only the surface composition of the specimen, difficulties associated with the optical thickness and the light-scattering properties of the substance are eliminated. Thus, reflectance measurements are frequently more simple to perform on intensely absorbing materials. A particularly common technique used for IR reflectance measurements is termed attenuated total reflectance (ATR), also known as multiple internal reflectance (MIR). In the ATR technique, the beam of the IR spectrometer is passed through an appropriate IR window material (e.g., KRS-5, a TlBr-TlI eutectic mixture), which is cut at such an angle that the IR beam enters the first (front) surface of the window, but is totally reflected when it impinges on the second (back) surface (i.e., the angle of incidence of the radiation upon the second surface of the window exceeds the critical angle for that material). By appropriate window construction, it is possible to have many internal reflections of the IR beam before it is transmitted out of the window. If a specimen is placed in close contact with the window along the sides that totally reflect the IR beam, the intensity of reflected radiation is reduced at each wavelength (frequency) that the specimen absorbs. Thus, the ATR technique provides a reflectance spectrum that has been increased in intensity, when compared to a simple reflectance measurement, by the number of times that the IR beam is reflected within the window. The ATR technique provides excellent sensitivity, but it yields poor reproducibility, and is not a reliable quantitative technique unless an internal standard is intimately mixed with each test specimen.
Fluorescence spectrophotometry is often more sensitive than absorption spectrophotometry. In absorption measurements, the specimen transmittance is compared to that of a blank; and at low concentrations, both solutions give high signals. Conversely, in fluorescence spectrophotometry, the solvent blank has low rather than high output, so that the background radiation that may interfere with determinations at low concentrations is much less. Whereas few compounds can be determined conveniently at concentrations below 10 5 M by light absorption, it is not unusual to employ concentrations of 10
5 M by light absorption, it is not unusual to employ concentrations of 10 7 M to 10
7 M to 10 8 M in fluorescence spectrophotometry.
8 M in fluorescence spectrophotometry.
THEORY AND TERMS
The power of a radiant beam decreases in relation to the distance that it travels through an absorbing medium. It also decreases in relation to the concentration of absorbing molecules or ions encountered in that medium. These two factors determine the proportion of the total incident energy that emerge. The decrease in power of monochromatic radiation passing through a homogeneous absorbing medium is stated quantitatively by Beer's law, log10(1/T) = A = abc, in which the terms are as defined below.
Absorbance [Symbol: A]—
The logarithm, to the base 10, of the reciprocal of the transmittance (T). [Note—Descriptive terms used formerly include optical density, absorbancy, and extinction. ]
Absorptivity [Symbol: a]—
The quotient of the absorbance (A) divided by the product of the concentration of the substance (c), expressed in g per L, and the absorption path length (b) in cm. [Note—It is not to be confused with absorbancy index; specific extinction; or extinction coefficient. ]
Molar Absorptivity [Symbol: 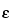 ]—
The quotient of the absorbance (A) divided by the product of the concentration, expressed in moles per L, of the substance and the absorption path length in cm. It is also the product of the absorptivity (a) and the molecular weight of the substance. [Note—Terms formerly used include molar absorbancy index; molar extinction coefficient; and molar absorption coefficient. ]
]—
The quotient of the absorbance (A) divided by the product of the concentration, expressed in moles per L, of the substance and the absorption path length in cm. It is also the product of the absorptivity (a) and the molecular weight of the substance. [Note—Terms formerly used include molar absorbancy index; molar extinction coefficient; and molar absorption coefficient. ]
For most systems used in absorption spectrophotometry, the absorptivity of a substance is a constant independent of the intensity of the incident radiation, the internal cell length, and the concentration, with the result that concentration may be determined photometrically.
Beer's law gives no indication of the effect of temperature, wavelength, or the type of solvent. For most analytical work the effects of normal variation in temperature are negligible.
Deviations from Beer's law may be caused by either chemical or instrumental variables. Apparent failure of Beer's law may result from a concentration change in solute molecules because of association between solute molecules or between solute and solvent molecules, or dissociation or ionization. Other deviations might be caused by instrumental effects such as polychromatic radiation, slit-width effects, or stray light.
Even at a fixed temperature in a given solvent, the absorptivity may not be truly constant. However, in the case of specimens having only one absorbing component, it is not necessary that the absorbing system conform to Beer's law for use in quantitative analysis. The concentration of an unknown may be found by comparison with an experimentally determined standard curve.
Although, in the strictest sense, Beer's law does not hold in atomic absorption spectrophotometry because of the lack of quantitative properties of the cell length and the concentration, the absorption processes taking place in the flame under conditions of reproducible aspiration do follow the Beer relationship in principle. Specifically, the negative log of the transmittance, or the absorbance, is directly proportional to the absorption coefficient, and, consequently, is proportional to the number of absorbing atoms. On this basis, calibration curves may be constructed to permit evaluation of unknown absorption values in terms of concentration of the element in solution.
Absorption Spectrum—
A graphic representation of absorbance, or any function of absorbance, plotted against wavelength or function of wavelength.
Transmittance [Symbol: T]—
The quotient of the radiant power transmitted by a specimen divided by the radiant power incident upon the specimen. [Note—Terms formerly used include transmittancy and transmission. ]
Fluorescence Intensity [Symbol: I]—
An empirical expression of fluorescence activity, commonly given in terms of arbitrary units proportional to detector response. The fluorescence emission spectrum is a graphical presentation of the spectral distribution of radiation emitted by an activated substance, showing intensity of emitted radiation as ordinate, and wavelength as abscissa. The fluorescence excitation spectrum is a graphical presentation of the activation spectrum, showing intensity of radiation emitted by an activated substance as ordinate, and wavelength of the incident (activating) radiation as abscissa. As in absorption spectrophotometry, the important regions of the electromagnetic spectrum encompassed by the fluorescence of organic compounds are the UV, visible, and near-IR, i.e., the region from 250 to 800 nm. After a molecule has absorbed radiation, the energy can be lost as heat or released in the form of radiation of the same or longer wavelength as the absorbed radiation. Both absorption and emission of radiation are due to the transitions of electrons between different energy levels, or orbitals, of the molecule. There is a time delay between the absorption and emission of light; this interval, the duration of the excited state, has been measured to be about 10 9 second to 10
9 second to 10 8 second for most organic fluorescent solutions. The short lifetime of fluorescence distinguishes this type of luminescence from phosphorescence, which is a long-lived afterglow having a lifetime of 10
8 second for most organic fluorescent solutions. The short lifetime of fluorescence distinguishes this type of luminescence from phosphorescence, which is a long-lived afterglow having a lifetime of 10 3 second up to several minutes.
3 second up to several minutes.
Turbidance [Symbol: S]—
The light-scattering effect of suspended particles. The amount of suspended matter may be measured by observation of either the transmitted light (turbidimetry) or the scattered light (nephelometry).
Turbidity [Symbol: 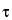 ]—
In light-scattering measurements, the turbidity is the measure of the decrease in incident beam intensity per unit length of a given suspension.
]—
In light-scattering measurements, the turbidity is the measure of the decrease in incident beam intensity per unit length of a given suspension.
Raman Scattering Activity—
The molecular property (in units of cm4 per g) governing the intensity of an observed Raman band for a randomly oriented specimen. The scattering activity is determined from the derivative of the molecular polarizability with respect to the molecular motion giving rise to the Raman shifted band. In general, the Raman band intensity is linearly proportional to the concentration of the analyte.
USE OF REFERENCE STANDARDS
With few exceptions, the Pharmacopeial spectrophotometric tests and assays call for comparison against a USP Reference Standard. This is to ensure measurement under conditions identical for the test specimen and the reference substance. These conditions include wavelength setting, slit-width adjustment, cell placement and correction, and transmittance levels. It should be noted that cells exhibiting identical transmittance at a given wavelength may differ considerably in transmittance at other wavelengths. Appropriate cell corrections should be established and used where required.
The expressions, “similar preparation” and “similar solution,” as used in tests and assays involving spectrophotometry, indicate that the reference specimen, generally a USP Reference Standard, is to be prepared and observed in a manner identical for all practical purposes to that used for the test specimen. Usually in making up the solution of the specified Reference Standard, a solution of about (i.e., within 10%) the desired concentration is prepared and the absorptivity is calculated on the basis of the exact amount weighed out; if a previously dried specimen of the Reference Standard has not been used, the absorptivity is calculated on the anhydrous basis.
The expressions, “concomitantly determine” and “concomitantly measured,” as used in tests and assays involving spectrophotometry, indicate that the absorbances of both the solution containing the test specimen and the solution containing the reference specimen, relative to the specified test blank, are to be measured in immediate succession.
APPARATUS
Many types of spectrophotometers are available. Fundamentally, most types, except those used for IR spectrophotometry, provide for passing essentially monochromatic radiant energy through a specimen in suitable form, and measuring the intensity of the fraction that is transmitted. Fourier transform IR spectrophotometers use an interferometric technique whereby polychromatic radiation passes through the analyte and onto a detector on an intensity and time basis. UV, visible, and dispersive IR spectrophotometers comprise an energy source, a dispersing device (e.g., a prism or grating), slits for selecting the wavelength band, a cell or holder for the test specimen, a detector of radiant energy, and associated amplifiers and measuring devices. In diode array spectrophotometers, the energy from the source is passed through the test specimen and then dispersed via a grating onto several hundred light-sensitive diodes, each of which in turn develops a signal proportional to the number of photons at its small wavelength interval; these signals then may be computed at rapid chosen intervals to represent a complete spectrum. Fourier transform IR systems utilize an interferometer instead of a dispersing device and a digital computer to process the spectral data. Some instruments are manually operated, whereas others are equipped for automatic and continuous recording. Instruments that are interfaced to a digital computer have the capabilities also of co-adding and storing spectra, performing spectral comparisons, and performing difference spectroscopy (accomplished with the use of a digital absorbance subtraction method).
Instruments are available for use in the visible; in the visible and UV; in the visible, UV, and near-IR; and in the IR regions of the spectrum. Choice of the type of spectrophotometric analysis and of the instrument to be used depends upon factors such as the composition and amount of available test specimen, the degree of accuracy, sensitivity, and selectivity desired, and the manner in which the specimen is handled.
The apparatus used in atomic absorption spectrophotometry has several unique features. For each element to be determined, a specific source that emits the spectral line to be absorbed should be selected. The source is usually a hollow-cathode lamp, the cathode of which is designed to emit the desired radiation when excited. Since the radiation to be absorbed by the test specimen element is usually of the same wavelength as that of its emission line, the element in the hollow-cathode lamp is the same as the element to be determined. The apparatus is equipped with an aspirator for introducing the test specimen into a flame, which is usually provided by air–acetylene, air–hydrogen, or, for refractory cases, nitrous oxide–acetylene. The flame, in effect, is a heated specimen chamber. A detector is used to read the signal from the chamber. Interfering radiation produced by the flame during combustion may be negated by the use of a chopped source lamp signal of a definite frequency. The detector should be tuned to this alternating current frequency so that the direct current signal arising from the flame is ignored. The detecting system, therefore, reads only the change in signal from the hollow-cathode source, which is directly proportional to the number of atoms to be determined in the test specimen. For Pharmacopeial purposes, apparatus that provides the readings directly in absorbance units is usually required. However, instruments providing readings in percent transmission, percent absorption, or concentration may be used if the calculation formulas provided in the individual monographs are revised as necessary to yield the required quantitative results. Percent absorption or percent transmittance may be converted to absorbance, A, by the following two equations:
A = 2  log10 (100
log10 (100  % absorption)
% absorption)
or:
A = 2  log10 (% transmittance)
log10 (% transmittance)
Depending upon the type of apparatus used, the readout device may be a meter, digital counter, recorder, or printer. Both single-beam and double-beam instruments are commercially available, and either type is suitable.
Measurement of fluorescence intensity can be made with a simple filter fluorometer. Such an instrument consists of a radiation source, a primary filter, a specimen chamber, a secondary filter, and a fluorescence detection system. In most such fluorometers, the detector is placed on an axis at 90 from that of the exciting beam. This right-angle geometry permits the exciting radiation to pass through the test specimen and not contaminate the output signal received by the fluorescence detector. However, the detector unavoidably receives some of the exciting radiation as a result of the inherent scattering properties of the solutions themselves, or if dust or other solids are present. Filters are used to eliminate this residual scatter. The primary filter selects short-wavelength radiation capable of exciting the test specimen, while the secondary filter is normally a sharp cut-off filter that allows the longer-wavelength fluorescence to be transmitted but blocks the scattered excitation.
from that of the exciting beam. This right-angle geometry permits the exciting radiation to pass through the test specimen and not contaminate the output signal received by the fluorescence detector. However, the detector unavoidably receives some of the exciting radiation as a result of the inherent scattering properties of the solutions themselves, or if dust or other solids are present. Filters are used to eliminate this residual scatter. The primary filter selects short-wavelength radiation capable of exciting the test specimen, while the secondary filter is normally a sharp cut-off filter that allows the longer-wavelength fluorescence to be transmitted but blocks the scattered excitation.
Most fluorometers use photomultiplier tubes as detectors, many types of which are available, each having special characteristics with respect to spectral region of maximum sensitivity, gain, and electrical noise. The photocurrent is amplified and read out on a meter or recorder.
A spectrofluorometer differs from a filter fluorometer in that filters are replaced by monochromators, of either the prism or the grating type. For analytical purposes, the spectrofluorometer is superior to the filter fluorometer in wavelength selectivity, flexibility, and convenience, in the same way in which a spectrophotometer is superior to a filter photometer.
Many radiation sources are available. Mercury lamps are relatively stable and emit energy mainly at discrete wavelengths. Tungsten lamps provide an energy continuum in the visible region. The high-pressure xenon arc lamp is often used in spectrofluorometers because it is a high-intensity source that emits an energy continuum extending from the UV into the IR.
In spectrofluorometers, the monochromators are equipped with slits. A narrow slit provides high resolution and spectral purity, while a large slit sacrifices these for high sensitivity. Choice of slit size is determined by the separation between exciting and emitting wavelengths as well as the degree of sensitivity needed.
Specimen cells used in fluorescence measurements may be round tubes or rectangular cells similar to those used in absorption spectrophotometry, except that they are polished on all four vertical sides. A convenient test specimen size is 2 to 3 mL, but some instruments can be fitted with small cells holding 100 to 300 µL, or with a capillary holder requiring an even smaller amount of specimen.
Light-scattering instruments are available and consist in general of a mercury lamp, with filters for the strong green or blue lines, a shutter, a set of neutral filters with known transmittance, and a sensitive photomultiplier to be mounted on an arm that can be rotated around the solution cell and set at any angle from  135
135 to 0
to 0 to +135
to +135 by a dial outside the light-tight housing. Solution cells are of various shapes, such as square for measuring 90
by a dial outside the light-tight housing. Solution cells are of various shapes, such as square for measuring 90 scattering; semioctagonal for 45
scattering; semioctagonal for 45 , 90
, 90 , and 135
, and 135 scattering; and cylindrical for scattering at all angles. Since the determination of molecular weight requires a precise measure of the difference in refractive index between the solution and solvent, [(n
scattering; and cylindrical for scattering at all angles. Since the determination of molecular weight requires a precise measure of the difference in refractive index between the solution and solvent, [(n  n0)/c], a second instrument, a differential refractometer, is needed to measure this small difference.
n0)/c], a second instrument, a differential refractometer, is needed to measure this small difference.
Raman spectrometers include the following major components: a source of intense monochromatic radiation (invariably a laser); optics to collect the light scattered by the test specimen; a (double) monochromator to disperse the scattered light and reject the intense incident frequency; and a suitable light-detection and amplification system. Raman measurement is simple in that most specimens are examined directly in melting-point capillaries. Because the laser source can be focused sharply, only a few microliters of the specimen is required.
PROCEDURE
Absorption Spectrophotometry
Detailed instructions for operating spectrophotometers are supplied by the manufacturers. To achieve significant and valid results, the operator of a spectrophotometer should be aware of its limitations and of potential sources of error and variation. The instruction manual should be followed closely on such matters as care, cleaning, and calibration of the instrument, and techniques of handling absorption cells, as well as instructions for operation. The following points require special emphasis.
Check the instrument for accuracy of calibration. Where a continuous source of radiant energy is used, attention should be paid to both the wavelength and photometric scales; where a spectral line source is used, only the photometric scale need be checked. A number of sources of radiant energy have spectral lines of suitable intensity, adequately spaced throughout the spectral range selected. The best single source of UV and visible calibration spectra is the quartz-mercury arc, of which the lines at 253.7, 302.25, 313.16, 334.15, 365.48, 404.66, and 435.83 nm may be used. The glass-mercury arc is equally useful above 300 nm. The 486.13-nm and 656.28-nm lines of a hydrogen discharge lamp may be used also. The wavelength scale may be calibrated also by means of suitable glass filters, which have useful absorption bands through the visible and UV regions. Standard glasses containing didymium (a mixture of praseodymium and neodymium) have been used widely, although glasses containing holmium were found to be superior. Standard holmium oxide solution has superseded the use of holmium glass.1 The wavelength scales of near-IR and IR spectrophotometers are readily checked by the use of absorption bands provided by polystyrene films, carbon dioxide, water vapor, or ammonia gas.
For checking the photometric scale, a number of standard inorganic glass filters as well as standard solutions of known transmittances such as potassium dichromate are available.2
Quantitative absorbance measurements usually are made on solutions of the substance in liquid-holding cells. Since both the solvent and the cell window absorb light, compensation must be made for their contribution to the measured absorbance. Matched cells are available commercially for UV and visible spectrophotometry for which no cell correction is necessary. In IR spectrophotometry, however, corrections for cell differences usually must be made. In such cases, pairs of cells are filled with the selected solvent and the difference in their absorbances at the chosen wavelength is determined. The cell exhibiting the greater absorbance is used for the solution of the test specimen and the measured absorbance is corrected by subtraction of the cell difference.
With the use of a computerized Fourier transform IR system, this correction need not be made, since the same cell can be used for both the solvent blank and the test solution. However, it must be ascertained that the transmission properties of the cell are constant.
Comparisons of a test specimen with a Reference Standard are best made at a peak of spectral absorption for the compound concerned. Assays prescribing spectrophotometry give the commonly accepted wavelength for peak spectral absorption of the substance in question. It is known that different spectrophotometers may show minor variation in the apparent wavelength of this peak. Good practice demands that comparisons be made at the wavelength at which peak absorption occurs. Should this differ by more than ±1 nm from the wavelength specified in the individual monograph, recalibration of the instrument may be indicated.
test preparation
For determinations utilizing UV or visible spectrophotometry, the specimen generally is dissolved in a solvent. Unless otherwise directed in the monograph, determinations are made at room temperature using a path length of 1 cm. Many solvents are suitable for these ranges, including water, alcohols, chloroform, lower hydrocarbons, ethers, and dilute solutions of strong acids and alkalies. Precautions should be taken to utilize solvents free from contaminants absorbing in the spectral region being used. It is usually advisable to use water-free methanol or alcohol, or alcohol denatured by the addition of methanol but not containing benzene or other interfering impurities, as the solvent. Solvents of special spectrophotometric quality, guaranteed to be free from contaminants, are available commercially from several sources. Some other analytical reagent-grade organic solvents may contain traces of impurities that absorb strongly in the UV region. New lots of these solvents should be checked for their transparency, and care should be taken to use the same lot of solvent for preparation of the test solution and the standard solution and for the blank.
No solvent in appreciable thickness is completely transparent throughout the near-IR and IR spectrum. Carbon tetrachloride (up to 5 mm in thickness) is practically transparent to 6 µm (1666 cm 1). Carbon disulfide (1 mm in thickness) is suitable as a solvent to 40 µm (250 cm
1). Carbon disulfide (1 mm in thickness) is suitable as a solvent to 40 µm (250 cm 1) with the exception of the 4.2-µm to 5.0-µm (2381-cm
1) with the exception of the 4.2-µm to 5.0-µm (2381-cm 1 to 2000-cm
1 to 2000-cm 1) and the 5.5-µm to 7.5-µm (1819-cm
1) and the 5.5-µm to 7.5-µm (1819-cm 1 to 1333-cm
1 to 1333-cm 1) regions, where it has strong absorption. Other solvents have relatively narrow regions of transparency. For IR spectrophotometry, an additional qualification for a suitable solvent is that it must not affect the material, usually sodium chloride, of which the cell is made. The test specimen may also be prepared by dispersing the finely ground solid specimen in mineral oil or by mixing it intimately with previously dried alkali halide salt (usually potassium bromide). Mixtures with alkali halide salts may be examined directly or as transparent disks or pellets obtained by pressing the mixture in a die. Typical drying conditions for potassium bromide are 105
1) regions, where it has strong absorption. Other solvents have relatively narrow regions of transparency. For IR spectrophotometry, an additional qualification for a suitable solvent is that it must not affect the material, usually sodium chloride, of which the cell is made. The test specimen may also be prepared by dispersing the finely ground solid specimen in mineral oil or by mixing it intimately with previously dried alkali halide salt (usually potassium bromide). Mixtures with alkali halide salts may be examined directly or as transparent disks or pellets obtained by pressing the mixture in a die. Typical drying conditions for potassium bromide are 105 in vacuum for 12 hours, although grades are commercially available that require no drying. Infrared microscopy or a mineral oil dispersion is preferable where disproportionation between the alkali halide and the test specimen is encountered. For suitable materials the test specimen may be prepared neat as a thin sample for IR microscopy or suspended neat as a thin film for mineral oil dispersion. For Raman spectrometry, most common solvents are suitable, and ordinary (nonfluorescing) glass specimen cells can be used. The IR region of the electromagnetic spectrum extends from 0.8 to 400 µm. From 800 to 2500 nm (0.8 to 2.5 µm) is generally considered to be the near-IR (NIR) region; from 2.5 to 25 µm (4000 to 400 cm
in vacuum for 12 hours, although grades are commercially available that require no drying. Infrared microscopy or a mineral oil dispersion is preferable where disproportionation between the alkali halide and the test specimen is encountered. For suitable materials the test specimen may be prepared neat as a thin sample for IR microscopy or suspended neat as a thin film for mineral oil dispersion. For Raman spectrometry, most common solvents are suitable, and ordinary (nonfluorescing) glass specimen cells can be used. The IR region of the electromagnetic spectrum extends from 0.8 to 400 µm. From 800 to 2500 nm (0.8 to 2.5 µm) is generally considered to be the near-IR (NIR) region; from 2.5 to 25 µm (4000 to 400 cm 1) is generally considered to be the mid-range (mid-IR) region; and from 25 to 400 µm is generally considered to be the far-IR (FIR) region. Unless otherwise specified in the individual monograph, the region from 3800 to 650 cm
1) is generally considered to be the mid-range (mid-IR) region; and from 25 to 400 µm is generally considered to be the far-IR (FIR) region. Unless otherwise specified in the individual monograph, the region from 3800 to 650 cm 1 (2.6 to 15 µm) should be used to ascertain compliance with monograph specifications for IR absorption.
1 (2.6 to 15 µm) should be used to ascertain compliance with monograph specifications for IR absorption.
Where values for IR line spectra are given in an individual monograph, the letters s, m, and w signify strong, medium, and weak absorption, respectively; sh signifies a shoulder, bd signifies a band, and v means very. The values may vary as much as 0.1 µm or 10 cm 1, depending upon the particular instrument used. Polymorphism gives rise to variations in the IR spectra of many compounds in the solid state. Therefore, when conducting IR absorption tests, if a difference appears in the IR spectra of the analyte and the standard, dissolve equal portions of the test substance and the standard in equal volumes of a suitable solvent, evaporate the solutions to dryness in similar containers under identical conditions, and repeat the test on the residues.
1, depending upon the particular instrument used. Polymorphism gives rise to variations in the IR spectra of many compounds in the solid state. Therefore, when conducting IR absorption tests, if a difference appears in the IR spectra of the analyte and the standard, dissolve equal portions of the test substance and the standard in equal volumes of a suitable solvent, evaporate the solutions to dryness in similar containers under identical conditions, and repeat the test on the residues.
In NIR spectroscopy much of the current interest centers around the ease of analysis. Samples can be analyzed in powder form or by means of reflectance techniques, with little or no preparation. Compliance with in-house specifications can be determined by computerized comparison of spectra with spectra previously obtained from reference materials. Many pharmaceutical materials exhibit low absorptivity in this spectral region, which allows incident near-IR radiation to penetrate samples more deeply than UV, visible, or IR radiation. NIR spectrophotometry may be used to observe matrix modifications and, with proper calibration, may be used in quantitative analysis.
In atomic absorption spectrophotometry, the nature of the solvent and the concentration of solids must be given special consideration. An ideal solvent is one that interferes to a minimal extent in the absorption or emission processes and one that produces neutral atoms in the flame. If there is a significant difference between the surface tension or viscosity of the test solution and standard solution, the solutions are aspirated or atomized at a different rate, causing significant differences in the signals generated. The acid concentration of the solutions also affects the absorption processes. Thus, the solvents used in preparing the test specimen and the standard should be the same or as much alike in these respects as possible, and should yield solutions that are easily aspirated via the specimen tube of the burner-aspirator. Since undissolved solids present in the solutions may give rise to matrix or bulk interferences, the total undissolved solids content in all solutions should be kept below 2% wherever possible.
calculations
The application of absorption spectrophotometry in an assay or a test generally requires the use of a Reference Standard. Where such a measurement is specified in an assay, a formula is provided in order to permit calculation of the desired result. A numerical constant is frequently included in the formula. The following derivation is provided to introduce a logical approach to the deduction of the constants appearing in formulas in the assays in many monographs.
The Beer's law relationship is valid for the solutions of both the Reference Standard (S) and the test specimen (U):
AS = abCS

 (1)
(1)
AU = abCU

 (2)
(2)
in which AS is the absorbance of the Standard solution of concentration CS; and AU is the absorbance of the test specimen solution of concentration CU. If CS and CU are expressed in the same units and the absorbances of both solutions are measured in matching cells having the same dimensions, the absorptivity, a, and the cell thickness, b, are the same; consequently, the two equations may be combined and rewritten to solve for CU:
CU = CS(AU/AS)

 (3)
(3)
Quantities of solid test specimens to be taken for analysis are generally specified in mg. Instructions for dilution are given in the assay and, since dilute solutions are used for absorbance measurements, concentrations are usually expressed for convenience in units of µg per mL. Taking a quantity, in mg, of a test specimen of a drug substance or solid dosage form for analysis, it therefore follows that a volume (VU), in L, of solution of concentration CU may be prepared from the amount of test specimen that contains a quantity WU, in mg, of the drug substance [Note—CU is numerically the same whether expressed as µg per mL or mg per L ] , such that:
WU = VUCU

 (4)
(4)
The form in which the formula appears in the assay in a monograph for a solid article may be derived by substituting CU of equation (3) into equation (4). In summary, the use of equation (4), with due consideration for any unit conversions necessary to achieve equality in equation (5), permits the calculation of the constant factor (VU) occurring in the final formula:
WU = VUCS(AU/AS)

 (5)
(5)
The same derivation is applicable to formulas that appear in monographs for liquid articles that are assayed by absorption spectrophotometry. For liquid dosage forms, results of calculations are generally expressed in terms of the quantity, in mg, of drug substance in each mL of the article. Thus it is necessary to include in the denominator an additional term, the volume (V), in mL, of the test preparation taken.
Assays in the visible region usually call for comparing concomitantly the absorbance produced by the Assay preparation with that produced by a Standard preparation containing approximately an equal quantity of a USP Reference Standard. In some situations, it is permissible to omit the use of a Reference Standard. This is true where spectrophotometric assays are made with routine frequency, and where a suitable standard curve is available, prepared with the respective USP Reference Standard, and where the substance assayed conforms to Beer's law within the range of about 75% to 125% of the final concentration used in the assay. Under these circumstances, the absorbance found in the assay may be interpolated on the standard curve, and the assay result calculated therefrom.
Such standard curves should be confirmed frequently, and always when a new spectrophotometer or new lots of reagents are put into use.
In spectrophotometric assays that direct the preparation and use of a standard curve, it is permissible and preferable, when the assay is employed infrequently, not to use the standard curve but to make the comparison directly against a quantity of the Reference Standard approximately equal to that taken of the specimen, and similarly treated.
Fluorescence Spectrophotometry
The measurement of fluorescence is a useful analytical technique. Fluorescence is light emitted from a substance in an excited state that has been reached by the absorption of radiant energy. A substance is said to be fluorescent if it can be made to fluoresce. Many compounds can be assayed by procedures utilizing either their inherent fluorescence or the fluorescence of suitable derivatives.
Test specimens prepared for fluorescence spectrophotometry are usually one-tenth to one-hundredth as concentrated as those used in absorption spectrophotometry, for the following reason. In analytical applications, it is preferable that the fluorescence signal be linearly related to the concentration; but if a test specimen is too concentrated, a significant part of the incoming light is absorbed by the specimen near the cell surface, and the light reaching the center is reduced. That is, the specimen itself acts as an “inner filter.” However, fluorescence spectrophotometry is inherently a highly sensitive technique, and concentrations of 10 5 M to 10
5 M to 10 7 M frequently are used. It is necessary in any analytical procedure to make a working curve of fluorescence intensity versus concentration in order to establish a linear relationship. All readings should be corrected for a solvent blank.
7 M frequently are used. It is necessary in any analytical procedure to make a working curve of fluorescence intensity versus concentration in order to establish a linear relationship. All readings should be corrected for a solvent blank.
Fluorescence measurements are sensitive to the presence of dust and other solid particles in the test specimen. Such impurities may reduce the intensity of the exciting beam or give misleading high readings because of multiple reflections in the specimen cell. It is, therefore, wise to eliminate solid particles by centrifugation; filtration also may be used, but some filter papers contain fluorescent impurities.
Temperature regulation is often important in fluorescence spectrophotometry. For some substances, fluorescence efficiency may be reduced by as much as 1% to 2% per degree of temperature rise. In such cases, if maximum precision is desired, temperature-controlled specimen cells are useful. For routine analysis, it may be sufficient to make measurements rapidly enough so that the specimen does not heat up appreciably from exposure to the intense light source. Many fluorescent compounds are light-sensitive. Exposed in a fluorometer, they may be photo-degraded into more or less fluorescent products. Such effects may be detected by observing the detector response in relationship to time, and may be reduced by attenuating the light source with filters or screens.
Change of solvent may markedly affect the intensity and spectral distribution of fluorescence. It is inadvisable, therefore, to alter the solvent specified in established methods without careful preliminary investigation. Many compounds are fluorescent in organic solvents but virtually nonfluorescent in water; thus, a number of solvents should be tried before it is decided whether or not a compound is fluorescent. In many organic solvents, the intensity of fluorescence is increased by elimination of dissolved oxygen, which has a strong quenching effect. Oxygen may be removed by bubbling an inert gas such as nitrogen or helium through the test specimen.
A semiquantitative measure of the strength of fluorescence is given by the ratio of the fluorescence intensity of a test specimen and that of a standard obtained with the same instrumental settings. Frequently, a solution of stated concentration of quinine in 0.1 N sulfuric acid or fluorescein in 0.1 N sodium hydroxide is used as a reference standard.
Light-Scattering
Turbidity can be measured with a standard photoelectric filter photometer or spectrophotometer, preferably with illumination in the blue portion of the spectrum. Nephelometric measurements require an instrument with a photocell placed so as to receive scattered rather than transmitted light; this geometry applies also to fluorometers, so that, in general, fluorometers can be used as nephelometers, by proper selection of filters. A ratio turbidimeter combines the technology of 90 nephelometry and turbidimetry: it contains photocells that receive and measure scattered light at a 90
nephelometry and turbidimetry: it contains photocells that receive and measure scattered light at a 90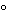 angle from the sample as well as receiving and measuring the forward scatter in front of the sample; it also measures light transmitted directly through the sample. Linearity is attained by calculating the ratio of the 90
angle from the sample as well as receiving and measuring the forward scatter in front of the sample; it also measures light transmitted directly through the sample. Linearity is attained by calculating the ratio of the 90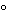 angle scattered light measurement to the sum of the forward scattered light measurement and the transmitted light measurement. The benefit of using a ratio turbidimetry system is that the measurement of stray light becomes negligible.
angle scattered light measurement to the sum of the forward scattered light measurement and the transmitted light measurement. The benefit of using a ratio turbidimetry system is that the measurement of stray light becomes negligible.
In practice, it is advisable to ensure that settling of the particles being measured is negligible. This is usually accomplished by including a protective colloid in the liquid suspending medium. It is important that results be interpreted by comparison of readings with those representing known concentrations of suspended matter, produced under precisely the same conditions.
Turbidimetry or nephelometry may be useful for the measurement of precipitates formed by the interaction of highly dilute solutions of reagents, or other particulate matter, such as suspensions of bacterial cells. In order that consistent results may be achieved, all variables must be carefully controlled. Where such control is possible, extremely dilute suspensions may be measured.
The specimen solute is dissolved in the solvent at several different accurately known concentrations, the choice of concentrations being dependent on the molecular weight of the solute and ranging from 1% for Mw = 10,000 to 0.01% for Mw = 1,000,000. Each solution must be very carefully cleaned before measurement by repeated filtration through fine filters. A dust particle in the solution vitiates the intensity of the scattered light measured. A criterion for a clear solution is that the dissymmetry, 45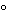 /135
/135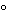 scattered intensity ratio, has attained a minimum.
scattered intensity ratio, has attained a minimum.
The turbidity and refractive index of the solutions are measured. From the general 90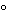 light-scattering equation, a plot of HC/
light-scattering equation, a plot of HC/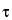 versus C is made and extrapolated to infinite dilution, and the weight-average molecular weight, M, is calculated from the intercept, 1/M.
versus C is made and extrapolated to infinite dilution, and the weight-average molecular weight, M, is calculated from the intercept, 1/M.
Visual Comparison
Where a color or a turbidity comparison is directed, color-comparison tubes that are matched as closely as possible in internal diameter and in all other respects should be used. For color comparison, the tubes should be viewed downward, against a white background, with the aid of a light source directed from beneath the bottoms of the tubes, while for turbidity comparison the tubes should be viewed horizontally, against a dark background, with the aid of a light source directed from the sides of the tubes.
In conducting limit tests that involve a comparison of colors in two like containers (e.g., matched color-comparison tubes), a suitable instrument, rather than the unaided eye, may be used.
1
National Institute of Standards and Technology (NIST), Gaithersburg, MD 20899: “Spectral Transmittance Characteristics of Holmium Oxide in Perchloric Acid,” J. Res. Natl. Bur. Stds. 90, No. 2, 115 (1985). The performance of an uncertified filter should be checked against a certified standard.
2
For further detail regarding checks on photometric scale of a spectrophotometer, reference may be made to the following NIST publications: J. Res. Nalt. Bur. Stds. 76A, 469 (1972) [re: SRM 93l, “Liquid Absorbance Standards for Ultraviolet and Visible Spectrophotometry” as well as potassium chromate and potassium dichromate]; NIST Spec. Publ. 260–116 (1994) [re: SRM 930 and SRM 1930, “Glass Filters for Spectrophotometry.”
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| General Chapter | Horacio N. Pappa, Ph.D.
Principal Scientific Liaison 1-301-816-8319 |
(GCCA2010) General Chapters - Chemical Analysis |
USP35–NF30 Page 409
Pharmacopeial Forum: Volume No. 35(1) Page 157