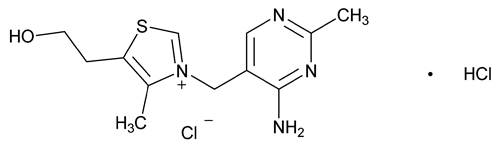
Thiamine Hydrochloride
C12H17ClN4OS·HCl337.27
Thiazolium, 3-[(4-amino-2-methyl-5-pyrimidinyl)methyl]-5-(2-hydroxyethyl)-4-methyl-, chloride, monohydrochloride;
Thiamine monohydrochloride

 [67-03-8].
[67-03-8].
(thye' a min hye'' droe klor' ide).
C12H17ClN4OS·HCl337.27
Thiazolium, 3-[(4-amino-2-methyl-5-pyrimidinyl)methyl]-5-(2-hydroxyethyl)-4-methyl-, chloride, monohydrochloride;
Thiamine monohydrochloride
DEFINITION
Thiamine Hydrochloride contains NLT 98.0% and NMT 102.0% of thiamine hydrochloride (C12H17ClN4OS·HCl), calculated on the anhydrous basis.
IDENTIFICATION
• A. Infrared Absorption  197K
197K
Analysis: Dry specimens at 105 for 2 h.
for 2 h.
Acceptance criteria: Meets the requirements
• B. Identification Tests—General, Chloride  191
191 : A 20-mg/mL solution meets the requirements.
: A 20-mg/mL solution meets the requirements.
ASSAY
• Procedure
Solution A: 0.005 M sodium 1-octanesulfonate in dilute glacial acetic acid (1 in 100)
Solution B: Methanol and acetonitrile (3:2)
Mobile phase: Solution B and Solution A (40:60)
Internal standard solution: 2% (v/v) of methylbenzoate in methanol
Standard solution: Prepare a 1-mg/mL solution of USP Thiamine Hydrochloride RS in Mobile phase. Transfer 20.0 mL of this solution and 5.0 mL of Internal standard solution to a 50-mL volumetric flask, and dilute with Mobile phase to volume. The Standard solution contains 400 µg/mL of thiamine hydrochloride.
Sample solution: Prepare a 2-mg/mL of Thiamine Hydrochloride in Mobile phase. Transfer 10.0 mL of this solution and 5.0 mL of Internal standard solution to a 50-mL volumetric flask, and dilute with Mobile phase to volume.
Chromatographic system
Mode: LC
Detector: UV 254 nm
Column: 4-mm × 30-cm; packing L1
Flow rate: 1 mL/min
[Note—The flow rate may be adjusted as needed to obtain a retention time of about 12 min for thiamine hydrochloride. ]
Injection size: 10 µL
System suitability
Sample: Standard solution
Suitability requirements
Resolution: NLT 4.0 between the thiamine and methylbenzoate peaks
Tailing factor: NMT 2.0 for the thiamine peak
Column efficiency: NLT 1500 theoretical plates for the thiamine peak
Relative standard deviation: NMT 2.0% for the ratios of thiamine peak areas to the internal standard peak area
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of thiamine hydrochloride (C12H17ClN4OS·HCl) in the portion of Thiamine Hydrochloride taken:
Result = (RU/RS) × (CS/CU) × 100
| RU | = | = internal standard ratio (peak area of thiamine/peak area of the internal standard) from the Sample solution |
| RS | = | = internal standard ratio (peak area of thiamine/peak area of the internal standard) from the Standard solution |
| CS | = | = concentration of USP Thiamine Hydrochloride RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Thiamine Hydrochloride in the Sample solution (mg/mL) |
Acceptance criteria: 98.0%–102.0% on the anhydrous basis
IMPURITIES
• Residue on Ignition  281
281 : NMT 0.2%
: NMT 0.2%
• Limit of Nitrate
Sample solution: 20 mg/mL of Thiamine Hydrochloride
Analysis: To 2 mL of the Sample solution add 2 mL of sulfuric acid. Cool, and superimpose 2 mL of ferrous sulfate TS.
Acceptance criteria: No brown ring is produced at the junction of the two layers.
• Related Compounds
Solution A, Solution B, and Mobile phase: Proceed as directed in the Assay.
Sample solution: 1.0 mg/mL of Thiamine Hydrochloride in Mobile phase
Chromatographic system
Mode: LC
Detector: UV 254 nm
Column: 4.0-mm × 15-cm; packing L1
Flow rate: 0.75 mL/min
Injection size: 10 µL
Analysis
Sample: Sample solution
Allow the Sample solution to elute for NLT three times the retention time of the main peak.
Calculate the percentage of total secondary peaks in the portion of Thiamine Hydrochloride taken:
Result = (rU/rT) × 100
| rU | = | = sum of the areas of all the peaks, except that of the thiamine peak |
| rT | = | = sum of the areas of all the peaks |
Acceptance criteria: NMT 1.0%
SPECIFIC TESTS
• Water Determination, Method I  921
921 : NMT 5.0%
: NMT 5.0%
• Absorbance of Solution
Sample solution: 100 mg/mL in water. Filter through a fine-porosity, sintered-glass funnel.
Blank: Water
Instrumental conditions
Mode: UV-Vis
Analytical wavelength: 400 nm
Cell: 1 cm
Analysis
Samples: Sample solution and Blank
Determine the absorbance of the Sample solution against that of the Blank.
Acceptance criteria: NMT 0.025
ADDITIONAL REQUIREMENTS
• Packaging and Storage: Preserve in tight, light-resistant containers.
Auxiliary Information— Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Huy T. Dinh, M.S. Scientific Liaison 1-301-816-8594 | (DS2010) Monographs - Dietary Supplements |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4835
Chromatographic Column—
Chromatographic columns text is not derived from, and not part of, USP 35 or NF 30.