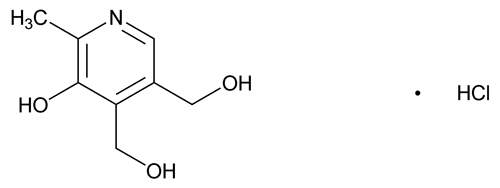
Pyridoxine Hydrochloride
C8H11NO3·HCl205.64
3,4-Pyridinedimethanol, 5-hydroxy-6-methyl-, hydrochloride;
Pyridoxol hydrochloride

 [58-56-0].
[58-56-0].
(pir'' i dox' een hye'' droe klor' ide).
C8H11NO3·HCl205.64
3,4-Pyridinedimethanol, 5-hydroxy-6-methyl-, hydrochloride;
Pyridoxol hydrochloride
DEFINITION
Pyridoxine Hydrochloride contains NLT 98.0% and NMT 102.0% of pyridoxine hydrochloride (C8H11NO3·HCl), calculated on the dried basis.
IDENTIFICATION
• B. Identification Tests—General, Chloride  191
191 : Meets the requirements
: Meets the requirements
ASSAY
• Procedure
Mobile phase: Mix 10 mL of glacial acetic acid, 0.6 g of sodium 1-hexanesulfonate, and 700 mL of water in a 1000-mL volumetric flask. Adjust with glacial acetic acid or 1 N sodium hydroxide to a pH of 3.0. Add 235 mL of methanol, and dilute with water to volume.
Internal standard solution: 5 mg/mL of p-hydroxybenzoic acid in Mobile phase
Standard solution: Prepare a 0.5-mg/mL solution of USP Pyridoxine Hydrochloride RS in Mobile phase. Transfer 10.0 mL of this solution and 1.0 mL of Internal standard solution to a 100-mL volumetric flask, and dilute with Mobile phase to volume.
Sample solution: Prepare a 0.5-mg/mL solution of Pyridoxine Hydrochloride in Mobile phase. Transfer 10.0 mL of this solution and 1.0 mL of Internal standard solution to a 100-mL volumetric flask, and dilute with Mobile phase to volume.
Chromatographic system
Mode: LC
Detector: UV 280 nm
Column: 4.6-mm × 25-cm; packing L1
Flow rate: 1.5 mL/min
Injection size: 20 µL
System suitability
Sample: Standard solution
[Note—The relative retention times for pyridoxine and p-hydroxybenzoic acid are about 0.7 and 1.0, respectively. ]
Suitability requirements
Resolution: NLT 2.5 between pyridoxine and p-hydroxybenzoic acid
Relative standard deviation: NMT 3.0% for the ratios of the pyridoxine peak area response to the internal standard peak area response
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of pyridoxine hydrochloride (C8H11NO3·HCl) in the portion of Pyridoxine Hydrochloride taken:
Result = (RU/RS) × (CS/CU) × 100
| RU | = | = internal standard ratio (peak response of pyridoxine/peak response of the internal standard) from the Sample solution |
| RS | = | = internal standard ratio (peak response of pyridoxine/peak response of the internal standard) from the Standard solution |
| CS | = | = concentration of USP Pyridoxine Hydrochloride RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Pyridoxine Hydrochloride in the Sample solution (mg/mL) |
Acceptance criteria: 98.0%–102.0% on the dried basis
IMPURITIES
• Residue on Ignition  281
281 : NMT 0.1%
: NMT 0.1%
• Heavy Metals, Method II  231
231 : NMT 30 ppm
: NMT 30 ppm
SPECIFIC TESTS
• Content of Chloride
Sample: 500 mg of Pyridoxine Hydrochloride
Blank: 50 mL of methanol
Titrimetric system
(See Titrimetry  541
541 .)
.)
Mode: Direct titration
Titrant: 0.1 N silver nitrate VS
Endpoint detection: Visual
Analysis: Dissolve the Sample in 50 mL of methanol. Add 5 mL of glacial acetic acid and 2–3 drops of eosin Y TS. Titrate with the Titrant. Perform a Blank determination.
Calculate the percentage of chloride (Cl) in the Sample taken:
Result = {[(VS  VB) × N × F]/W} × 100
VB) × N × F]/W} × 100
| VS | = | = Titrant volume consumed by the Sample (mL) |
| VB | = | = Titrant volume consumed by the Blank (mL) |
| N | = | = actual normalilty of the Titrant (mEq/mL) |
| F | = | = equivalency factor, 35.45 mg/mEq |
| W | = | = Sample weight (mg) |
Acceptance criteria: 16.9%–17.6% on the dried basis
• Loss on Drying  731
731 : Dry a sample in vacuum over silica gel for 4 h: it loses NMT 0.5% of its weight.
: Dry a sample in vacuum over silica gel for 4 h: it loses NMT 0.5% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage: Preserve in tight, light-resistant containers.
Auxiliary Information— Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Huy T. Dinh, M.S. Scientific Liaison 1-301-816-8594 | (DS2010) Monographs - Dietary Supplements |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4491
Chromatographic Column—
Chromatographic columns text is not derived from, and not part of, USP 35 or NF 30.