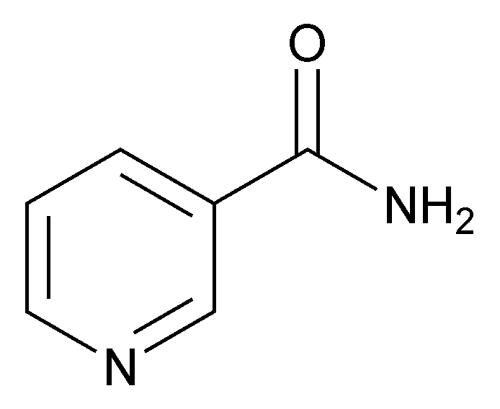
Niacinamide
(nye'' a sin' a mide).
DEFINITION
Niacinamide contains NLT 98.5% and NMT 101.5% of niacinamide (C6H6N2O), calculated on the dried basis.
IDENTIFICATION
• B. Ultraviolet Absorption  197U
197U
Sample solution: 20 µg/mL in water
Acceptance criteria: It meets the requirements in the chapter, and the A245/A262 ratio is 0.63–0.67.
ASSAY
• Procedure
Mobile phase: Methanol and 0.005 M sodium 1-heptanesulfonate (30:70)
Standard solution: Transfer 50 mg of USP Niacinamide RS to a 100-mL volumetric flask. Add 3 mL of water to dissolve, and dilute with Mobile phase to volume. Dilute with Mobile phase to 0.04 mg/mL.
Niacin standard solution Prepare as directed in the Standard solution, using USP Niacin RS instead of USP Niacinamide RS.
Sample solution: Prepare as directed in the Standard solution, using Niacinamide instead of USP Niacinamide RS.
System suitability solution: Mix equal volumes of the Standard solution and Niacin standard solution.
Chromatographic system
Mode: LC
Detector: UV 254 nm
Column: 3.9-mm × 30-cm; packing L1
Flow rate: 2 mL/min
Injection size: 20 µL
System suitability
Samples: Standard solution and System suitability solution
Suitability requirements
Resolution: NLT 3.0 between niacin and niacinamide, System suitability solution
Relative standard deviation: NMT 2.0%, Standard solution
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of niacinamide (C6H6N2O) in the portion of Niacinamide taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak area from the Sample solution |
| rS | = | = peak area from the Standard solution |
| CS | = | = concentration of USP Niacinamide RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Niacinamide in the Sample solution (mg/mL) |
Acceptance criteria: 98.5%–101.5% on the dried basis
IMPURITIES
• Residue on Ignition  281
281 : NMT 0.1%
: NMT 0.1%
• Heavy Metals, Method II  231
231 : NMT 30 ppm
: NMT 30 ppm
• Readily Carbonizable Substances  271
271
Sample solution: Dissolve 200 mg of Niacinamide in 5 mL of sulfuric acid.
Acceptance criteria: The Sample solution has no more color than Matching Fluid A.
SPECIFIC TESTS
• Melting Range or Temperature  741
741 : 128
: 128 –131
–131
• Loss on Drying  731
731 : Dry a sample over silica gel for 4 h: it loses NMT 0.5% of its weight.
: Dry a sample over silica gel for 4 h: it loses NMT 0.5% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage: Preserve in tight containers.
Auxiliary Information— Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Huy T. Dinh, M.S. Scientific Liaison 1-301-816-8594 | (DS2010) Monographs - Dietary Supplements |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4043
Pharmacopeial Forum: Volume No. 34(5) Page 1176Chromatographic Column—
Chromatographic columns text is not derived from, and not part of, USP 35 or NF 30.