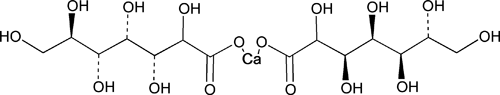
Calcium Gluceptate
C14H26CaO16 (anhydrous)490.42
Glucoheptonic acid, calcium salt (2:1);
Calcium glucoheptonate (1:2)

 [29039-00-7].
[29039-00-7].
(kal' see um gloo sep' tate).
C14H26CaO16 (anhydrous)490.42
Glucoheptonic acid, calcium salt (2:1);
Calcium glucoheptonate (1:2)
DEFINITION
Calcium Gluceptate is anhydrous or contains varying amounts of water of hydration. It consists of the calcium salt of the alpha epimer of glucoheptonic acid or of a mixture of the alpha and beta epimers of glucoheptonic acid. It contains NLT 95.0% and NMT 102.0% of calcium gluceptate (C14H26CaO16), calculated on the dried basis.
IDENTIFICATION
• B. Identification Tests—General, Calcium  191
191 : A 20-mg/mL solution meets the requirements.
: A 20-mg/mL solution meets the requirements.
ASSAY
• Procedure
Sample: 800 mg of Calcium Gluceptate
Blank: 150 mL of water containing 2 mL of 3 N hydrochloric acid
Titrimetric system
(See Titrimetry  541
541 .)
.)
Mode: Direct titration
Titrant: 0.05 M edetate disodium VS
Endpoint detection: Visual
Analysis: Dissolve the Sample with 150 mL of water containing 2 mL of 3 N hydrochloric acid. While stirring, preferably with a magnetic stirrer, add 25 mL of Titrant from the titration buret. Add 15 mL of 1 N sodium hydroxide and 300 mg of hydroxy naphthol blue, and continue the titration to a blue endpoint. Perform the Blank determination.
Calculate the percentage of calcium gluceptate (C14H26CaO16) in the Sample taken:
Result = {[(VS  VB) × M × F]/W} × 100
VB) × M × F]/W} × 100
| VS | = | = Titrant volume consumed by the Sample (mL) |
| VB | = | = Titrant volume consumed by the Blank (mL) |
| M | = | = actual molarity of the Titrant (mM/mL) |
| F | = | = equivalency factor, 490.4 mg/mM |
| W | = | = Sample weight (mg) |
Acceptance criteria: 95.0%–102.0% on the dried basis
IMPURITIES
• Chloride and Sulfate, Chloride  221
221
Standard: 1.0 mL of 0.020 N hydrochloric acid
Sample: 1.0 g
Acceptance criteria: NMT 0.07%
• Chloride and Sulfate, Sulfate  221
221
Standard: 1.0 mL of 0.020 N sulfuric acid
Sample: 2.0 g
Acceptance criteria: NMT 0.05%
• Reducing Sugars
Sample: 0.50 g
Analysis: Dissolve the Sample in 10 mL of hot water, add 2 mL of 3 N hydrochloric acid, boil for about 2 min, and cool. Add 5 mL of sodium carbonate TS, allow to stand for 5 min, dilute with water to 20 mL, and filter. Add 5 mL of the clear filtrate to 2 mL of alkaline cupric tartrate TS, and boil for 1 min.
Acceptance criteria: No red precipitate is formed immediately.
SPECIFIC TESTS
• Loss on Drying  731
731
(See Thermal Analysis  891
891 .)
.)
[Note—The quantity taken for the determination may be adjusted, if necessary, for instrument sensitivity. Weight loss occurring at temperatures above about 160 , indicative of decomposition, is not to be interpreted as Loss on Drying. ]
, indicative of decomposition, is not to be interpreted as Loss on Drying. ]
Sample: 10–25 mg
Analysis: Determine the percentage of volatile substances by thermogravimetric analysis on an appropriately calibrated instrument. Heat the specimen under test at a rate of 5 /min in an atmosphere of nitrogen, at a flow rate of 40 mL/min. Record the thermogram to 150
/min in an atmosphere of nitrogen, at a flow rate of 40 mL/min. Record the thermogram to 150 .
.
Acceptance criteria: See Table 1.
Table 1
| Form | Loss on Drying (%) |
|---|---|
| Anhydrous | NMT 1.0 |
| 2H2O (dihydrate) | NMT 6.9 |
| 31/2H2O | NMT 11.4 |
ADDITIONAL REQUIREMENTS
• Packaging and Storage: Preserve in well-closed containers.
• Labeling: Label to indicate whether hydrous or anhydrous; if hydrous, label to indicate also the degree of hydration.
Auxiliary Information— Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Huy T. Dinh, M.S. Scientific Liaison 1-301-816-8594 | (DS2010) Monographs - Dietary Supplements |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2452
Pharmacopeial Forum: Volume No. 27(6) Page 3257Chromatographic Column—
Chromatographic columns text is not derived from, and not part of, USP 35 or NF 30.