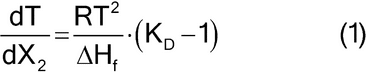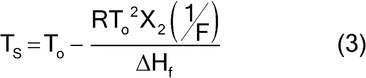INTRODUCTION
Precisely determined thermodynamic events, such as a change of state, can indicate the identity and purity of drugs. Compendial standards have long been established for the melting or boiling temperatures of substances. These transitions occur at characteristic temperatures, and the compendial standards therefore contribute to the identification of the substances. Because impurities affect these changes in predictable ways, the same compendial standards contribute to the control of the purity of the substances.
Thermal analysis in the broadest sense is the measurement of physical–chemical properties of materials as a function of temperature. Instrumental methods have largely supplanted older methods dependent on visual inspection and measurements under fixed or arbitrary conditions, because they are objective, provide more information, afford permanent records, and are generally more sensitive, precise, and accurate. Furthermore, they may provide information on desolvation, dehydration, decomposition, crystal perfection, polymorphism, melting temperature, sublimation, glass transitions, evaporation, pyrolysis, solid-solid interactions, and purity. Such data are useful in the characterization of substances with respect to compatibility, stability, packaging, and quality control. The measurements used most often in thermal analysis, i.e., transition and melting point temperatures by differential scanning calorimetry (DSC), thermogravimetric analysis, hot-stage microscopy, and eutectic impurity analysis, are described here.
TRANSITION AND MELTING POINT TEMPERATURES
As a specimen is heated, transitions can be observed using differential scanning calorimetry (DSC), differential thermal analysis (DTA), or hot-stage microscopy. In heat-flux DSC, the heat differential between the sample and reference material is determined. In power compensation DSC, the sample and reference materials are maintained at the same temperature, using individual heating elements, and the difference in power input to the two heaters is recorded. DTA monitors the difference in temperatures between the sample and the reference. The transitions that may be observed include those shown in Table 1 below. In the case of melting, both an “onset” and a “peak” temperature can be determined objectively and reproducibly, often to within a few tenths of a degree. Although these temperatures are useful for characterizing substances, and the difference between the two temperatures is indicative of purity, the values cannot be directly compared to visual “melting-range” or “melting-point” values or with constants such as the triple point of the pure material.
Furthermore, caution should be used when comparing results obtained by different methods of analysis. Optical methods may measure the melting point as the temperature where the last trace of solid coalesces. In contrast, melting points measured by DSC may refer to the onset temperature or the temperature where the maximum melting rate (vertex) was observed. However, the vertex is sensitive to sample weight, heating rate, and other factors, whereas the onset temperature is less affected by these factors. With thermal techniques, it is necessary to consider the limitations of solid solution formation, insolubility in the melt, polymorphism, and decomposition during the analysis.
Table 1
| Solid to liquid | Melting | Endothermic |
| Liquid to gas | Vaporization | Endothermic |
| Liquid to solid | Freezing | Exothermic |
| Crystallization | Exothermic | |
| Solid to gas | Sublimation | Endothermic |
| Solid to solid | Glass transition | Second order event |
| Desolvation | Endothermic | |
| Amorphous to crystalline | Exothermic | |
| Polymorphic | Endothermic or Exothermic |
Reporting Results of Instrumental Methods—
A complete description of the conditions employed should accompany each thermogram, including make and model of instrument; record of last calibration; specimen size and identification (including previous thermal history); container; identity, flow rate, and pressure of gaseous atmosphere; direction and rate of temperature change; and instrument and recorder sensitivity.
DETERMINATION OF TRANSITION TEMPERATURE (MELT ONSET TEMPERATURE) AND MELTING POINT TEMPERATURE
Apparatus—
Use DTA or DSC instrumentation equipped with a temperature-programming device, thermal detector(s), and a recording system that can be connected to a computer, unless otherwise prescribed by the specific monograph for which this chapter is being used.
Calibration—
Calibrate instrumentation for temperature and enthalpy changes, using indium or another suitable certified material. Temperature calibration is performed by heating a standard through the melting transition and comparing the extrapolated onset of melting point of the standard to the certified onset of melting point. The temperature calibration should be conducted at the same heating rate as the experiment. Enthalpy calibration is performed by heating a standard through the melting transition and comparing the calculated heat of fusion to the theoretical value.
Procedure—
Accurately weigh an appropriate quantity of the substance to be examined in the sample pan, as described in the specific monograph. Set initial temperature, heating rate, direction of temperature change, and final temperature as specified in the monograph. If not specified in the monograph, these parameters are determined as follows: make a preliminary examination over a wide range of temperatures (typically, room temperature to decomposition temperature, or about 10 to 20
to 20 above the melting point) and over a wide range of heating rates (1
above the melting point) and over a wide range of heating rates (1 to 20
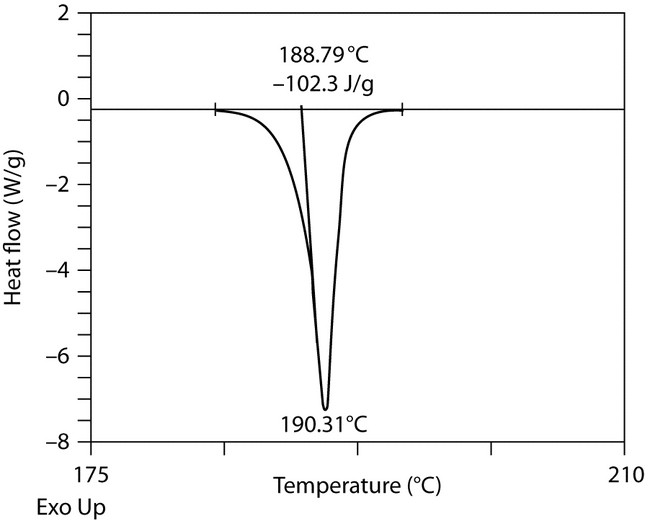
to 20 per minute) to reveal any unexpected effects. Then determine a lower heating rate such that decomposition is minimized and the transition temperature is not compromised. Determine a temperature range bracketing the transition of interest such that the baseline can be extended to intersect with the tangent of the melt (see Figure 1).
per minute) to reveal any unexpected effects. Then determine a lower heating rate such that decomposition is minimized and the transition temperature is not compromised. Determine a temperature range bracketing the transition of interest such that the baseline can be extended to intersect with the tangent of the melt (see Figure 1).
In examining pure crystalline materials, rates as low as 1 per minute may be appropriate, whereas rates of up to 20
per minute may be appropriate, whereas rates of up to 20 per minute are more appropriate for polymeric and other semicrystalline materials. Begin the analysis, and record the differential thermal analysis curve with the temperature on the x-axis and the energy change on the y-axis. The melting temperature (melt onset temperature) is the intersection (188.79
per minute are more appropriate for polymeric and other semicrystalline materials. Begin the analysis, and record the differential thermal analysis curve with the temperature on the x-axis and the energy change on the y-axis. The melting temperature (melt onset temperature) is the intersection (188.79 ) of the extension of the baseline with the tangent at the point of greatest slope (inflexion point) of the curve (see Figure 1). The vertex is the temperature at the peak of the curve (190.31
) of the extension of the baseline with the tangent at the point of greatest slope (inflexion point) of the curve (see Figure 1). The vertex is the temperature at the peak of the curve (190.31 ). The enthalpy of the event is proportional to the area under the curve after application of a baseline correction.
). The enthalpy of the event is proportional to the area under the curve after application of a baseline correction.
THERMOGRAVIMETRIC ANALYSIS
Thermogravimetric analysis involves the determination of the mass of a specimen as a function of temperature, or time of heating, or both. It is often used to investigate dehydration/desolvation processes and compound decomposition. When thermogravimetry is properly applied, it provides more useful information than loss on drying at fixed temperature, often for a fixed time and in what is usually an ill-defined atmosphere. Usually, loss of surface-absorbed solvent can be distinguished from solvent in the crystal lattice and from degradation losses. The measurements can be carried out in atmospheres having controlled humidity and oxygen concentration to reveal interactions with the drug substance, between drug substances, and between active substances and excipients or packaging materials.
Apparatus—
While the details depend on the manufacturer, the essential features of the equipment are a recording balance and a programmable heat source. Equipment differs in the ability to handle specimens of various sizes, the means of sensing specimen temperature, and the range of atmosphere control.
Calibration—
Calibration is required with all systems: that is, the mass scale is calibrated by the use of standard weights, and calibration of the temperature scale involves the use of standard materials, because it is assumed that the specimen temperature is the furnace temperature. Weight calibration is conducted by measuring the mass of a certified or standard weight and comparing the measured mass with the certified value. Temperature calibration is performed by analyzing a high-purity magnetic standard such as nickel for its curie temperature and comparing the measured value to the theoretical value.
Procedure—
Apply the method to the sample, using the conditions described in the monograph, and calculate the mass gain or loss, expressing the change in mass as percentage. Alternatively, place a suitable quantity of material in the sample holder, and record the mass. Because the test atmosphere is critical, the pressure or flow rate and the composition of the gas are specified. Set the initial temperature, heating rate, and final temperature according to the manufacturer’s instructions, and initiate the temperature increase. Alternatively, conduct an examination of the thermogram over a wide range of temperatures (typically, from room temperature to the decomposition temperature, or 10 to 20
to 20 above the melting point at a heating rate of 1
above the melting point at a heating rate of 1 to 20
to 20 per minute). Calculate the mass gain or loss, expressing the change in mass as percentage.
per minute). Calculate the mass gain or loss, expressing the change in mass as percentage.
HOT-STAGE MICROSCOPY
Hot-stage microscopy is an analytical technique that involves monitoring the optical properties of the sample using a microscope as a function of temperature. Hot-stage microscopy may be used as a complementary technique to other thermal analysis techniques such as DSC, DTA, and variable temperature X-ray powder diffraction for the solid-state characterization of pharmaceutical compounds. It is useful to confirm transitions such as melts, recrystallizations, and solid-state transformations using a visual technique. The hot-stage microscope must be calibrated for temperature.
EUTECTIC IMPURITY ANALYSIS
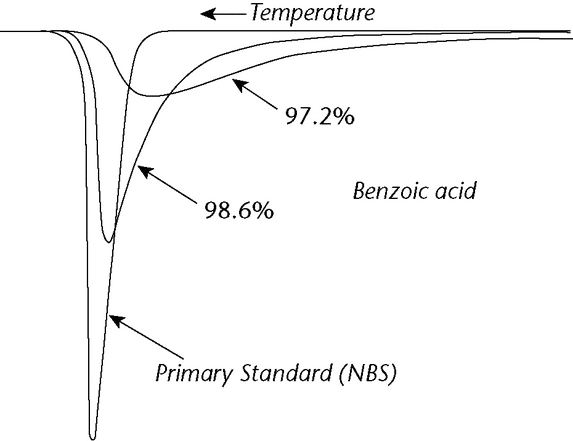
The basis of any calorimetric purity method is the relationship between the melting and freezing point depression and the level of impurity. The melting of a compound is characterized by the absorption of latent heat of fusion, DHf, at a specific temperature, To. In theory, a melting transition for an absolutely pure crystalline compound should occur within an infinitely narrow range. A broadening of the melting range, due to impurities, provides a sensitive criterion of purity. The effect is apparent visually by examination of thermograms of specimens differing by a few tenths percent in impurity content. A material that is 99% pure is about 20% molten at 3 below the melting point of the pure material (see Figure 2).
below the melting point of the pure material (see Figure 2).
The parameters of melting (melting range, DHf, and calculated eutectic purity) are readily obtained from the thermogram of a single melting event using a small test specimen, and the method does not require multiple, precise actual temperature measurements. Thermogram units are directly convertible to heat transfer, millicalories per second.
The lowering of the freezing point in dilute solutions by molecules of nearly equal size is expressed by a modified van't Hoff equation:
in which T = absolute temperature in kelvins; X2 = mole fraction of minor component (solute, impurity); DHf = molar heat of fusion of the major component in Joules per mol: R = gas constant in Joules per mol × kelvins; and KD = distribution ratio of solute between the solid and liquid phases.
Assuming that the temperature range is small and that no solid solutions are formed (KD = 0).
Integration of the van't Hoff equation yields the following relationship between the mole fraction of impurity and the melting-point depression:
in which To = melting point of the pure compound, in kelvins, and Tm = melting point of the test specimen, in kelvins.
With no solid solution formation, the concentration of impurity in the liquid phase at any temperature during the melting is inversely proportional to the fraction melted at that temperature, and the melting-point depression is directly proportional to the mole fraction of impurity. A plot of the observed test specimen temperature, Ts, versus the reciprocal of the fraction melted, 1/F, at temperature Ts, should yield a straight line with the slope equal to the melting-point depression (To  Tm). The theoretical melting point of the pure compound is obtained by extrapolation to 1/F = 0:
Tm). The theoretical melting point of the pure compound is obtained by extrapolation to 1/F = 0:
Substituting the experimentally obtained values for To  Tm, DHf, and To in equation (2) yields the mole fraction of the total eutectic impurity, which, when multiplied by 100, gives the mole percentage of total eutectic impurities.
Tm, DHf, and To in equation (2) yields the mole fraction of the total eutectic impurity, which, when multiplied by 100, gives the mole percentage of total eutectic impurities.
Deviations from the theoretical linear plot also may be due to solid solution formation (KD 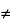 0), so that care must be taken in interpreting the data.
0), so that care must be taken in interpreting the data.
To observe the linear effect of the impurity concentration on the melting-point depression, the impurity must be soluble in the liquid phase or melt of the compound, but insoluble in the solid phase, i.e., no solid solutions are formed. Some chemical similarities are necessary for solubility in the melt. For example, the presence of ionic compounds in neutral organic compounds and the occurrence of thermal decomposition may not be reflected in purity estimates. The extent of these theoretical limitations has been only partially explored.
Impurities present from the synthetic route often are similar to the end product, hence there usually is no problem of solubility in the melt. Impurities consisting of molecules of the same shape, size, and character as those of the major component can fit into the matrix of the major component without disruption of the lattice, forming solid solutions or inclusions; such impurities are not detectable by DSC. Purity estimates are too high in such cases. This is more common with less-ordered crystals as indicated by low heats of fusion.
In addition, the method is reliable when the purity of the major component is greater than 98.5 mol% and the materials are not decomposed during the melting phase.
Impurity levels calculated from thermograms are reproducible and generally reliable within 0.1% for ideal compounds.
Compounds that exist in polymorphic form cannot be used in purity determination unless the compound is completely converted to one form. On the other hand, DSC and DTA are inherently useful for detecting, and therefore monitoring, polymorphism.
Procedure—
The actual procedure and the calculations to be employed for eutectic impurity analysis are dependent on the particular instrument used. Consult the manufacturer's literature and/or the thermal analysis literature for the most appropriate technique for a given instrument. In any event, it is imperative to keep in mind the limitations of solid solution formation, insolubility in the melt, polymorphism, and decomposition during the analysis.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| General Chapter | Antonio Hernandez-Cardoso, M.Sc.
Senior Scientific Liaison 1-301-816-8308 |
(GCPA2010) General Chapters - Physical Analysis |
USP35–NF30 Page 417
Pharmacopeial Forum: Volume No. 36(1) Page 289