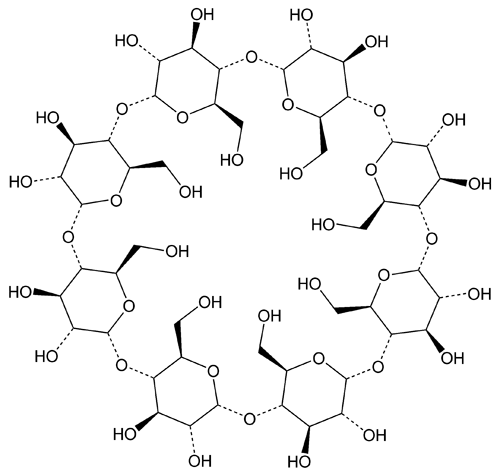
Gamma Cyclodextrin
DEFINITION
Gamma Cyclodextrin is composed of 8 alpha-(1–4) linked d-glycopyranosyl units. It contains NLT 98.0% and NMT 102.0% of cyclooctaamylose (C6H10O5)8, calculated on the dried basis.
IDENTIFICATION
• B.
The retention time of the major peak from the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
• C.
It meets the requirements of the test for Specific Rotation.
ASSAY
• Procedure
Mobile phase:
Methanol and water (7:93)
System suitability solution:
Prepare an aqueous solution containing 0.5 mg/mL each of USP Alpha Cyclodextrin RS, USP Beta Cyclodextrin RS, and USP Gamma Cyclodextrin RS.
Standard solution:
1.0 mg/mL of USP Gamma Cyclodextrin RS
Sample stock solution:
Transfer 250 mg of Gamma Cyclodextrin to a 25-mL volumetric flask, and dissolve in water, with the aid of heat if necessary. Cool, and dilute with water to volume.
Sample solution:
1.0 mg/mL of Gamma Cyclodextrin, prepared from the Sample stock solution
Chromatographic system
Mode:
LC
Detector:
Refractive index
Column:
4.6-mm × 15-cm; 5-µm packing L1
Temperature
Detector:
40
Column:
30
Flow rate:
1.5 mL/min
Injection size:
50 µL
System suitability
Sample:
System suitability solution
[Note—The relative retention times for gamma cyclodextrin, alfadex, and betadex are 0.8, 1.0, and 1.9, respectively. ]
Suitability requirements
Resolution:
NLT 1.5 between the gamma cyclodextrin and alfadex peaks
Tailing factors:
0.8–2.0 for the three cyclodextrins
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of gamma cyclodextrin [(C6H10O5)8] in the portion of sample taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of the Standard solution (mg/mL) |
| CU | = | = concentration of the Sample solution (mg/mL) corrected for water found in Specific Tests, Loss on Drying |
Acceptance criteria:
98.0%–102.0% on the dried basis
IMPURITIES
• Residue on Ignition  281
281 :
NMT 0.1%, determined on a 1.0-g specimen
:
NMT 0.1%, determined on a 1.0-g specimen
• Heavy Metals, Method II  231
231 :
NMT 5 ppm
:
NMT 5 ppm
• Related Compounds
Mobile phase, System suitability solution, and Chromatographic system:
Proceed as directed in the Assay.
Standard solution:
Transfer 5.0 mL of System suitability solution into a 50-mL volumetric flask, and dilute with water to volume.
Sample solution:
Use the Sample stock solution, prepared as directed in the Assay.
Analysis
Samples:
Standard solution and Sample solution
Acceptance criteria:
For the Sample solution, the areas of any peaks corresponding to alfadex (alpha cyclodextrin) or to betadex (beta cyclodextrin) are not greater than the area of the corresponding peaks in the chromatogram of the Standard solution (0.5%); and the sum of the areas of all the peaks, excluding the principal peak, the peaks corresponding to alfadex or to betadex, and artifact peaks, is not greater than the area of the peak corresponding to gamma cyclodextrin in the chromatogram of the Standard solution (0.5%).
• Reducing Substances
Dextrose standard solution:
10.0 mg/mL of USP Dextrose RS, calculated on the anhydrous basis
Analysis:
Transfer a quantity of Gamma Cyclodextrin, equivalent to 1.0 g on the dried basis, to a 500-mL conical flask. Dissolve in 10 mL of water, and add 25 mL of alkaline cupric citrate TS2. Cover the flask with aluminum foil, and boil the solution for 5 min. Cool in an ice bath to room temperature. Add 25 mL of 0.6 N acetic acid, 10 mL of 3 N hydrochloric acid, and 10 mL of 0.1 N iodine solution. [Note—The addition of these solutions must be in the order given. ]
Titrate the solution with 0.1 N sodium thiosulfate VS, and determine the endpoint potentiometrically. Perform a blank determination (see Titrimetry  541
541 , Residual Titrations). Calculate the difference in volumes required.
, Residual Titrations). Calculate the difference in volumes required.
Create a calibration curve by similarly titrating 0.25, 0.5, 0.75, and 1.0 mL of Dextrose standard solution. Plot the amount, in mg, of dextrose in each titrated Dextrose standard solution versus the volume consumed, in mL, of 0.1 N sodium thiosulfate VS in the titration, and draw a straight line through the four points. From the line so obtained and the volume of 0.1 N sodium thiosulfate VS required in the titration of Gamma Cyclodextrin, determine the weight, W, in mg, of the reducing substances as dextrose in the portion of Gamma Cyclodextrin taken.
Calculate the percentage of the reducing substances in the portion of Gamma Cyclodextrin taken:
Result = (W/WG) × F × 100
| W | = | = weight of the reducing substances as dextrose in the portion of Gamma Cyclodextrin taken (mg) |
| WG | = | = weight of Gamma Cyclodextrin taken (g) |
| F | = | = conversion factor, 10-3 g/mg |
Acceptance criteria:
NMT 0.5%
SPECIFIC TESTS
• Microbial Enumeration Tests  61
61 and Tests for Specified Microorganisms
and Tests for Specified Microorganisms  62
62 :
It meets the requirements of the tests for the absence of Salmonella species and Escherichia coli. The total aerobic microbial count does not exceed 1000 cfu/g, and the total combined molds and yeasts count does not exceed 100 cfu/g.
:
It meets the requirements of the tests for the absence of Salmonella species and Escherichia coli. The total aerobic microbial count does not exceed 1000 cfu/g, and the total combined molds and yeasts count does not exceed 100 cfu/g.
• Color and Clarity of Solution
Sample solution:
Transfer a quantity of Gamma Cyclodextrin, equivalent to 2.5 g on the dried basis, into a 25-mL volumetric flask, dissolve in and dilute with water that has been previously boiled and cooled to room temperature to volume, and mix.
Analysis:
Determine the absorbance of the Sample solution in a 1-cm cell at 420 nm, with a suitable spectrophotometer, after correcting for the blank.
Acceptance criteria:
At 420 nm, the absorbance is not greater than 0.20, and the solution is clear.
• Loss on Drying  731
731 :
Dry a sample at 105
:
Dry a sample at 105 for 2 h: it loses NMT 11.0% of its weight.
for 2 h: it loses NMT 11.0% of its weight.
• Optical Rotation, Specific Rotation  781S
781S
Sample solution:
10 mg/mL
Analysis:
Proceed as directed in the chapter.
Acceptance criteria:
+174 to +180
to +180
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in well-closed containers, and store at room temperature.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 1776
Pharmacopeial Forum: Volume No. 33(4) Page 707