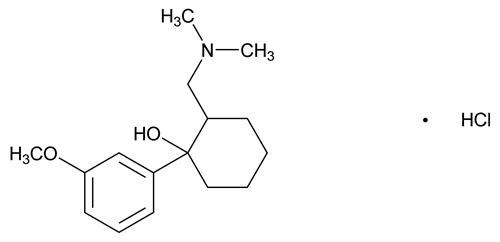
Tramadol Hydrochloride
C16H25NO2·HCl 299.84
(±)-cis-2-[(Dimethylamino)methyl]-1-(3-methoxyphenyl) cyclohexanol hydrochloride;
(±)-cis-2-[(Dimethylamino)methyl]-1-(m-methoxyphenyl) cyclohexanol hydrochloride

 [36282-47-0].
[36282-47-0].
(tram' a dol hye'' droe klor' ide).
C16H25NO2·HCl 299.84
(±)-cis-2-[(Dimethylamino)methyl]-1-(3-methoxyphenyl) cyclohexanol hydrochloride;
(±)-cis-2-[(Dimethylamino)methyl]-1-(m-methoxyphenyl) cyclohexanol hydrochloride
DEFINITION
Tramadol Hydrochloride contains NLT 98.0% and NMT 102.0% of C16H25NO2·HCl, calculated on the anhydrous basis.
IDENTIFICATION
• B. Identification Tests—General, Chloride  191
191 :
An aqueous solution (1 in 100) meets the requirements.
:
An aqueous solution (1 in 100) meets the requirements.
ASSAY
• Procedure
Solution A:
Dissolve 0.5 mL of trifluoroacetic acid in 1000 mL of water.
Mobile phase:
Acetonitrile and Solution A (30:70)
System suitability solution:
0.05 mg/mL each of USP Tramadol Hydrochloride RS and USP Tramadol Hydrochloride Related Compound A RS in Mobile phase
Standard solution:
1.5 mg/mL USP Tramadol Hydrochloride RS in Mobile phase
Sample solution:
1.5 mg/mL tramadol hydrochloride in Mobile phase
Chromatographic system
Mode:
LC
Detector:
UV 270 nm
Column:
4.6-mm × 25-cm; 5-µm packing L1
Flow rate:
1 mL/min
Injection size:
20 µL
System suitability
Sample:
System suitability solution
[Note—The relative retention times for tramadol related compound A and tramadol are about 0.9 and 1.0, respectively. ]
Suitability requirements
Resolution:
NLT 2.0 between tramadol related compound A and tramadol
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C16H25NO2·HCl in the portion of Tramadol Hydrochloride taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Tramadol Hydrochloride in the Standard solution (mg/mL) |
| CU | = | = nominal concentration of Tramadol Hydrochloride in the Sample solution (mg/mL) |
Acceptance criteria:
98.0%–102.0% on the anhydrous basis
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.1%
:
NMT 0.1%
• Heavy Metals, Method I  231
231 :
20 ppm
:
20 ppm
• Content of Chloride
Sample solution:
150 mg of Tramadol Hydrochloride in 40 mL of water
Analysis:
To the Sample solution add, with stirring, 7.5 mL of 4 N nitric acid and 15.0 mL of 0.1 N silver nitrate, and titrate with 0.1 N ammonium thiocynate VS, determining the endpoint potentiometrically, and using a silver–glass electrode system. Each mL of 0.1 N ammonium thiocynate is equivalent to 3.545 mg of chloride.
Acceptance criteria:
11.6%–12.1% of chloride is found.
Organic Impurities
• Procedure 1: Limit of Tramadol Related Compound B (2-(dimethylaminomethyl)-1-cyclohexanone hydrochloride)
Adsorbent:
0.25-mm layer of chromatographic silica gel mixture
Standard solution:
0.1 mg/mL USP Tramadol Related Compound B RS in methanol
Sample solution:
50 mg/mL Tramadol Hydrochloride in methanol
Developing solvent system:
Toluene, isopropyl alcohol, and 25% ammonia water (80:19:1)
Sodium nitrite solution:
50 mg/mL sodium nitrite in water
Analysis:
Separately apply about 10 µL each of the Sample solution and the Standard solution to the plate, and develop the plate until the solvent front is 10 cm above the line of application. Remove the plate, spray with Dragendorff's TS, and air-dry for 5 min. Spray the dried plate with Sodium nitrite solution until the spot from tramadol related compound B in the Standard solution is visible. Any secondary spot from the Sample solution corresponding to tramadol related compound B is not more intense than a corresponding spot from the Standard solution.
Acceptance criteria:
NMT 0.2%
• Procedure 2
Mobile phase, System suitability solution, Sample solution, Chromatographic system, and System suitability:
Proceed as directed in the Assay.
Analysis
Sample:
Sample solution
Calculate the percentage of each impurity in the portion of Tramadol Hydrochloride taken:
Result = (rU/rT) × 100
| rU | = | = peak response of each impurity from the Sample solution |
| rT | = | = sum of all the peak responses from the Sample solution |
Acceptance criteria
Tramadol related compound A:
NMT 0.2%
Individual impurities:
NMT 0.1%
Total impurities:
NMT 0.4%
SPECIFIC TESTS
• Water Determination, Method Ia  921
921 :
NMT 0.5%
:
NMT 0.5%
• Acidity
Sample solution:
500 mg of Tramadol Hydrochloride in 10 mL of water
Analysis:
To the Sample solution add 0.2 mL of methyl red TS and 0.2 mL of 0.01 N hydrochloric acid VS, and titrate with 0.01 N sodium hydroxide VS.
Acceptance criteria:
NMT 0.4 mL of 0.01 N sodium hydroxide is required to produce a yellow color.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers, and store at controlled room temperature.
• USP Reference Standards  11
11
USP Tramadol Related Compound A RS
RS,SR-1-(3-Methoxyphenyl)-2-(dimethylaminomethyl-cyclohexanol hydrochloride.
RS,SR-1-(3-Methoxyphenyl)-2-(dimethylaminomethyl-cyclohexanol hydrochloride.
USP Tramadol Related Compound B RS
2-(Dimethylaminomethyl)-1-cyclohexanone hydrochloride.
2-(Dimethylaminomethyl)-1-cyclohexanone hydrochloride.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Clydewyn M. Anthony, Ph.D.
Senior Scientific Liaison 1-301-816-8139 |
(SM22010) Monographs - Small Molecules 2 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4904
Pharmacopeial Forum: Volume No. 34(5) Page 1200