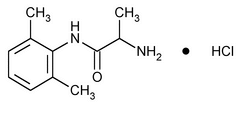Tocainide Hydrochloride
(toe' ka nide hye'' droe klor' ide).
Propanamide, 2-amino-N-(2,6-dimethylphenyl)-, hydrochloride, (±)-.
(±)-2-Amino-2¢,6¢-propionoxylidide hydrochloride.
» Tocainide Hydrochloride contains not less than 98.0 percent and not more than 101.0 percent of C11H16N2O·HCl, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
USP Reference standards  11
11 —
—
USP Tocainide Hydrochloride RS
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 2 hours: it loses not more than 0.5% of its weight.
for 2 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method II  231
231 :
0.002%.
:
0.002%.
Chromatographic purity—
Adsorbent:
0.25-mm layer of chromatographic silica gel mixture coating on a thin-layer chromatographic plate, previously washed with methanol.
Test solution:
100 mg per mL, in methanol.
Standard solutions:
1.0, 0.5, 0.25, and 0.1 mg per mL in methanol to obtain Standard solutions A, B, C, and D, respectively.
Application volume:
20 µL.
Developing solvent system:
a freshly prepared mixture of toluene and alcohol (4:1) in a paper-lined equilibrated tank in an atmosphere of ammonia vapors.
Procedure—
Proceed as directed for Thin-Layer Chromatography under Chromatography  621
621 . Examine the plate under short-wavelength UV light. Expose the plate to iodine vapors, and observe again under white light: the chromatograms show principal spots at about the same RF value. Estimate the concentration of any spot observed in the chromatogram of the Test solution, other than the principal spot and that observed at the origin (which may appear because of the presence of ammonium chloride), by comparison with the principal spots in the chromatograms of Standard solutions B, C, and D: the intensity of any secondary spot is not greater than that of the principal spot obtained from Standard solution B (0.5%), and the sum of all secondary spots is not greater than the intensity of the principal spot obtained from Standard solution A (1.0%).
. Examine the plate under short-wavelength UV light. Expose the plate to iodine vapors, and observe again under white light: the chromatograms show principal spots at about the same RF value. Estimate the concentration of any spot observed in the chromatogram of the Test solution, other than the principal spot and that observed at the origin (which may appear because of the presence of ammonium chloride), by comparison with the principal spots in the chromatograms of Standard solutions B, C, and D: the intensity of any secondary spot is not greater than that of the principal spot obtained from Standard solution B (0.5%), and the sum of all secondary spots is not greater than the intensity of the principal spot obtained from Standard solution A (1.0%).
Assay—
Dissolve about 180 mg of Tocainide Hydrochloride, accurately weighed, in about 40 mL of glacial acetic acid and 15 mL of a 6 in 100 solution of mercuric acetate in glacial acetic acid, and titrate with 0.1 N perchloric acid VS, determining the endpoint potentiometrically, using a platinum ring electrode and a sleeve-type calomel electrode containing 0.1 N lithium perchlorate in acetic anhydride (see Titrimetry  541
541 ). Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 22.87 mg of C11H16N2O·HCl.
). Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 22.87 mg of C11H16N2O·HCl.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Sujatha Ramakrishna, Ph.D.
Senior Scientific Liaison 1-301-816-8349 |
(SM22010) Monographs - Small Molecules 2 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4887