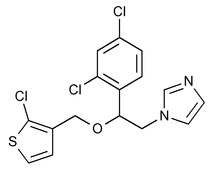
Tioconazole
C16H13Cl3N2OS 387.71
1H-Imidazole, 1-[2-[(2-chloro-3-thienyl)methoxy]-2-(2,4-dichlorophenyl)ethyl]-;
1-[2,4-Dichloro-[ -(2-chloro-3-thenyl)-oxy]phenethyl]imidazole
-(2-chloro-3-thenyl)-oxy]phenethyl]imidazole


 [65899-73-2].
[65899-73-2].
(tye'' oh kon' a zole).
C16H13Cl3N2OS 387.71
1H-Imidazole, 1-[2-[(2-chloro-3-thienyl)methoxy]-2-(2,4-dichlorophenyl)ethyl]-;
1-[2,4-Dichloro-[
DEFINITION
Tioconazole contains NLT 97.0% and NMT 103.0% of C16H13Cl3N2OS.
IDENTIFICATION
• B. Procedure
Standard solution:
50 mg/mL of USP Tioconazole RS in methanol
Sample solution:
50 mg/mL of Tioconazole in methanol
Developing solvent system:
Chloroform, methanol, and glacial acetic acid (40:5:1)
Visualizing solution:
Dissolve 0.85 g of bismuth subnitrate in 10 mL of glacial acetic acid, and dilute with water to 50 mL. Mix 10 mL of this solution, 50 mL of potassium iodide solution (2 in 25), and 20 mL of glacial acetic acid, and dilute to 100 mL.
Chromatographic system
Mode:
TLC
Adsorbent:
0.25-mm layer of chromatographic silica gel mixture
Application volume:
10 µL
Analysis
Samples:
Standard solution and Sample solution
Develop the chromatogram until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber and locate the spots on the plate by viewing under short- and long-wavelength UV light after drying the plate at 80 for 5 min. Spray the plate with Visualizing solution, air-dry for 2 min, and overspray with sodium nitrite solution (1 in 20). Air-dry the plate for 5 min, and examine it for brown spots on a pale yellow background.
for 5 min. Spray the plate with Visualizing solution, air-dry for 2 min, and overspray with sodium nitrite solution (1 in 20). Air-dry the plate for 5 min, and examine it for brown spots on a pale yellow background.
Acceptance criteria:
The RF value of the principal spot from the Sample solution corresponds to that from the Standard solution.
• C.
The retention time of the major peak for tioconazole of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
ASSAY
• Procedure
Mobile phase:
Acetonitrile, methanol, and water (44:40:28). Degas the solution, and add 2.0 mL of ammonium hydroxide. [Note—Prepare the Mobile phase fresh daily. ]
Standard solution:
200 µg/mL of USP Tioconazole RS in methanol
Sample solution:
200 µg/mL of Tioconazole in methanol
Chromatographic system
Mode:
LC
Detector:
UV 219 nm
Column:
5-mm × 25-cm; packing L1 with a precolumn containing packing L4 installed between the pump and the injector. [Note—Replace the precolumn daily. ]
Flow rate:
Adjust to obtain a retention time of between 12 and 17 min for tioconazole.
Injection size:
20 µL
System suitability
Sample:
Standard solution
Suitability requirements
Column efficiency:
NLT 1000 theoretical plates, determined from the analyte peak
Tailing factor:
NMT 2.0 for the analyte peak
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C16H13Cl3N2OS in the Tioconazole taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Tioconazole RS in the Standard solution (mg/mL) |
| CU | = | = concentration of the Sample solution (mg/mL) |
Acceptance criteria:
97.0%–103.0%
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.2%
:
NMT 0.2%
• Chloride and Sulfate, Chloride  221
221 :
A 0.7-g portion dissolved in methanol shows no more chloride than corresponds to 0.50 mL of 0.020 N hydrochloric acid (0.05%).
:
A 0.7-g portion dissolved in methanol shows no more chloride than corresponds to 0.50 mL of 0.020 N hydrochloric acid (0.05%).
• Heavy Metals, Method II  231
231 :
NMT 50 ppm
:
NMT 50 ppm
Organic Impurities
• Procedure
Mobile phase and Chromatographic system:
Proceed as directed in the Assay.
Standard solution:
Dissolve 1 mg each of USP Tioconazole Related Compound A RS, USP Tioconazole Related Compound B RS, and USP Tioconazole Related Compound C RS in 15.0 mL of methanol, and shake until the contents are completely dissolved.
Sample solution:
Dissolve 100 mg of Tioconazole in 15.0 mL of methanol, and shake until the substance is completely dissolved.
Analysis
Samples:
Standard solution and Sample solution
Calculate, in turn, the percentages of 1-[2,4-dichloro- -[(3-thenyl)-oxy]phenethyl]imidazole hydrochloride (tioconazole related compound A), 1-[2,4-dichloro-
-[(3-thenyl)-oxy]phenethyl]imidazole hydrochloride (tioconazole related compound A), 1-[2,4-dichloro- -[(2,5-dichloro-3-thenyl)-oxy]phenethyl]imidazole hydrochloride (tioconazole related compound B), and 1-[2,4-dichloro-
-[(2,5-dichloro-3-thenyl)-oxy]phenethyl]imidazole hydrochloride (tioconazole related compound B), and 1-[2,4-dichloro- -[(5-bromo-2-chloro-3-thenyl)-oxy]phenethyl]imidazole hydrochloride (tioconazole related compound C) in the portion of Tioconazole taken:
-[(5-bromo-2-chloro-3-thenyl)-oxy]phenethyl]imidazole hydrochloride (tioconazole related compound C) in the portion of Tioconazole taken:
Result = (rU/rS) × (WI/WU) × 100
| rU | = | = peak response of the related compound from the Sample solution |
| rS | = | = peak response of the related compound from the Standard solution |
| WI | = | = weight of the respective USP Reference Standard taken to prepare the Standard solution (mg) |
| WU | = | = weight of Tioconazole taken to prepare the Sample solution (mg) |
Acceptance criteria:
The limit of each related compound is NMT 1.0%.
SPECIFIC TESTS
• Water Determination, Method I  921
921 :
NMT 0.5%
:
NMT 0.5%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers.
• USP Reference Standards  11
11
USP Tioconazole Related Compound A RS 
1-[2,4-Dichloro- -[(3-thenyl)-oxy]phenethyl]imidazole hydrochloride.
-[(3-thenyl)-oxy]phenethyl]imidazole hydrochloride.
C16H14Cl2 N2OS·HCl 389.73

1-[2,4-Dichloro-
C16H14Cl2 N2OS·HCl 389.73
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Leonel M. Santos, Ph.D.
Senior Scientific Liaison 1-301-816-8168 |
(SM12010) Monographs - Small Molecules 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4875
Pharmacopeial Forum: Volume No. 35(4) Page 875