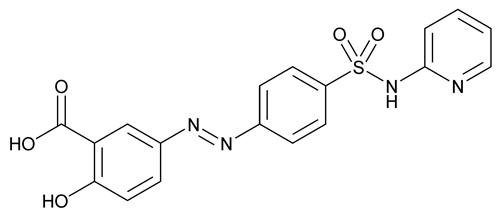
Sulfasalazine
C18H14N4O5S 398.39
Benzoic acid, 2-hydroxy-5-[[4-[(2-pyridinylamino)sulfonyl]phenyl]azo]-;
5-[[p-(2-Pyridylsulfamoyl)phenyl]azo]salicylic acid

 [599-79-1].
[599-79-1].
(sul'' fa sal' a zeen).
C18H14N4O5S 398.39
Benzoic acid, 2-hydroxy-5-[[4-[(2-pyridinylamino)sulfonyl]phenyl]azo]-;
5-[[p-(2-Pyridylsulfamoyl)phenyl]azo]salicylic acid
DEFINITION
Sulfasalazine contains NLT 97.0% and NMT 101.5% of C18H14N4O5S, calculated on the dried basis.
IDENTIFICATION
• B. Procedure
Standard solution:
Use the Standard solution, prepared as directed in the Assay.
Sample solution:
Use the Sample solution, prepared as directed in the Assay.
Acceptance criteria:
The visible absorption spectrum of the Sample solution corresponds to that of the Standard solution, as obtained from the Assay.
ASSAY
• Procedure
Sample stock solution:
1.5 mg/mL of Sulfasalazine in 0.1 N sodium hydroxide
Sample solution:
Transfer 5.0 mL of the Sample stock solution to a 1000-mL volumetric flask containing 750 mL of water, mix, add 20.0 mL of 0.1 N acetic acid, and dilute with water to volume.
Standard solution:
7.5 µg/mL of USP Sulfasalazine RS in the same medium as the Sample solution
Spectrometric conditions
Mode:
UV-Vis
Analytical wavelength:
Maximum at about 359 nm
Blank:
Water
Analysis
Samples:
Standard solution, Sample solution, and Blank
Concomitantly determine the absorbances of the Samples. Calculate the percentage of C18H14N4O5S in the portion of Sulfasalazine taken:
Result = (AU/AS) × (CS/CU) × 100
| AU | = | = absorbance of the Sample solution |
| AS | = | = absorbance of the Standard solution |
| CS | = | = concentration of USP Sulfasalazine RS in the Standard solution (µg/mL) |
| CU | = | = concentration of Sulfasalazine in the Sample solution (µg/mL) |
Acceptance criteria:
97.0%–101.5% on the dried basis
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.5%
:
NMT 0.5%
• Chloride and Sulfate, Chloride  221
221
Analysis:
Digest 2.0 g of Sulfasalazine with 100 mL of water at 70 for 5 min. Cool immediately to room temperature, and filter. Transfer a 25-mL portion of the filtrate to a 50-mL beaker (retain the remainder of this filtrate for the Sulfate test). Add 1 mL of nitric acid, mix, and allow to stand for 5 min. Pass through a fine texture, retentive filter paper (Whatman No. 42, or equivalent).
for 5 min. Cool immediately to room temperature, and filter. Transfer a 25-mL portion of the filtrate to a 50-mL beaker (retain the remainder of this filtrate for the Sulfate test). Add 1 mL of nitric acid, mix, and allow to stand for 5 min. Pass through a fine texture, retentive filter paper (Whatman No. 42, or equivalent).
Acceptance criteria:
The filtrate shows no more chloride than corresponds to 0.10 mL of 0.020 N hydrochloric acid (0.014%).
• Chloride and Sulfate, Sulfate  221
221
Analysis:
Transfer a 25-mL portion of the filtrate from the Chloride test to a 50-mL beaker. Add 1 mL of 3 N hydrochloric acid, mix, and allow to stand for 5 min. Pass through a fine texture, retentive filter paper (Whatman No. 42, or equivalent).
Acceptance criteria:
The filtrate shows no more sulfate than corresponds to 0.20 mL of 0.020 N sulfuric acid (0.04%).
• Heavy Metals, Method II  231
231 :
NMT 20 ppm
:
NMT 20 ppm
Organic Impurities
• Procedure
Standard stock solution:
10 mg/mL of USP Sulfasalazine RS in a mixture of alcohol and 2 M ammonium hydroxide (4:1)
Standard solutions:
Dilute aliquots of the Standard stock solution stepwise with the same medium to obtain solutions having concentrations of 200, 150, 100, and 20 µg/mL, corresponding to 2.0%, 1.5%, 1.0%, and 0.2%, respectively (Standard solutions A, B, C, and D).
Sample solution:
10 mg/mL of Sulfasalazine in a mixture of alcohol and 2 M ammonium hydroxide (4:1)
Chromatographic system
Mode:
TLC
Adsorbent:
0.25-mm layer of chromatographic silica gel mixture
Application volume:
10 µL
Developing solvent system:
Chloroform, acetone, and formic acid (12:6:1)
Analysis
Samples:
Standard solution and Sample solution
Proceed as directed in the chapter. Allow the spots to dry, and develop the chromatogram in an unequilibrated chamber until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the chamber, mark the solvent front, dry with the aid of a current of hot air, and examine the plate under short-wavelength UV light.
Acceptance criteria:
The RF value of the principal spot of the Sample solution corresponds to that of the Standard solution. No spots, other than the principal spot, in the chromatogram of the Sample solution are larger or more intense than the principal spot of Standard solution A (2%), and the sum of the intensities of any secondary spots detected does not exceed 4%.
SPECIFIC TESTS
• Loss on Drying  731
731 :
Dry a sample at 105
:
Dry a sample at 105 for 2 h: it loses NMT 1.0% of its weight.
for 2 h: it loses NMT 1.0% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight, light-resistant containers.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Leonel M. Santos, Ph.D.
Senior Scientific Liaison 1-301-816-8168 |
(SM12010) Monographs - Small Molecules 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4722
Pharmacopeial Forum: Volume No. 35(6) Page 1478