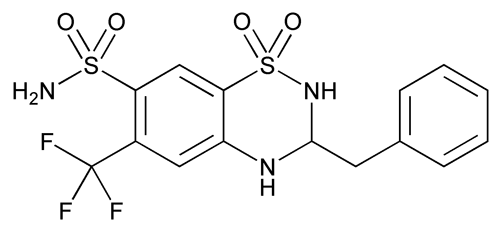
Bendroflumethiazide
C15H14F3N3O4S2 421.41
2H-1,2,4-Benzothiadiazine-7-sulfonamide, 3,4-dihydro-3-(phenylmethyl)-6-(trifluoromethyl)-, 1,1-dioxide, (±)-;
(±)-3-Benzyl-3,4-dihydro-6-(trifluoromethyl)-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide

 [73-48-3].
[73-48-3].
(ben'' droe floo'' me thye' a zide).
C15H14F3N3O4S2 421.41
2H-1,2,4-Benzothiadiazine-7-sulfonamide, 3,4-dihydro-3-(phenylmethyl)-6-(trifluoromethyl)-, 1,1-dioxide, (±)-;
(±)-3-Benzyl-3,4-dihydro-6-(trifluoromethyl)-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide
DEFINITION
Bendroflumethiazide contains NLT 98.0% and NMT 102.0% of C15H14F3N3O4S2, calculated on the anhydrous basis.
IDENTIFICATION
• A. Infrared Absorption  197K
197K
Sample:
Previously dried over silica gel for 4 h
Acceptance criteria:
Meets the requirements
• B. Ultraviolet Absorption  197U
197U
Analytical wavelength:
271 nm
Sample solution:
10 µg/mL in methanol
Acceptance criteria:
Absorptivities, calculated on the anhydrous basis, do not differ by more than 4.0%.
• C.
Sample solution:
Mix 5 mL of dilute hydrochloric acid (50% v/v) with 20 mg of Bendroflumethiazide, boil gently for 1 min, and cool in an ice bath.
Analysis:
To the Sample solution add, in succession, 0.5 mL of sodium nitrite solution (1 mg/mL), 0.5 mL of ammonium sulfamate solution (5 mg/mL), and 0.5 mL of N-(1-naphthyl)ethylenediamine dihydrochloride solution (1 mg/mL).
Acceptance criteria:
A deep red color is produced.
ASSAY
• Procedure
Sample:
190 mg of Bendroflumethiazide
Analysis:
Dissolve the Sample in 80 mL of pyridine in a tall-form, 250-mL beaker in a well-ventilated hood. Add 3 drops of a saturated solution of azo violet in methanol, cover the beaker, and gently bubble nitrogen through the solution for 5 min, being careful to avoid any contact between the solution and the cover. Raise the nitrogen delivery tube above the solution surface and, maintaining a gentle flushing with nitrogen and stirring with a magnetic or mechanical stirring device, add 0.1 N sodium methoxide VS from a 10-mL buret inserted through an opening in the cover. Titrate to a blue endpoint, approaching the endpoint at a rate of 1 or 2 drop/s. Perform a blank determination, and make any necessary correction (see Titrimetry  541
541 ). Each mL of 0.1 N sodium methoxide is equivalent to 21.07 mg of C15H14F3N3O4S2.
). Each mL of 0.1 N sodium methoxide is equivalent to 21.07 mg of C15H14F3N3O4S2.
Acceptance criteria:
98.0%–102.0% on the anhydrous basis
IMPURITIES
• Residue on Ignition  281
281 :
NMT 0.2%
:
NMT 0.2%
• Heavy Metals, Method II  231
231 :
20 ppm
:
20 ppm
• Selenium  291
291
Sample:
100 mg of Bendroflumethiazide and 100 mg of magnesium oxide
Acceptance criteria:
The absorbance from the Test solution is NMT one-half that from the Standard solution (NMT 30 ppm).
• Limit of 2,4-Disulfamyl-5-trifluoromethylaniline
[Note—Use low-actinic glassware for the Standard solution and the Sample solution. ]
Mobile phase:
Dissolve 5.62 g of sodium chloride and 1.97 g of anhydrous sodium sulfate in 1000 mL of water in a 2-L volumetric flask. Add 4.0 mL of glacial acetic acid and 800 mL of methanol, and dilute with water to volume.
Standard solution:
0.75 µg/mL of USP 2,4-Disulfamyl-5-trifluoromethylaniline RS in methanol
Sample solution:
50 µg/mL of Bendroflumethiazide in methanol
Chromatographic system
Mode:
LC
Detector:
UV 270 nm
Column:
4.6-mm × 30-cm; packing L11
Column temperature:
35 ± 5
± 5
Flow rate:
1.5 mL/min
Injection size:
20 µL
System suitability
Sample:
Standard solution
Suitability requirements
Resolution:
NLT 1.4 between the methanol and 2,4-disulfamyl-5-trifluoromethylaniline peaks
Relative standard deviation:
NMT 3.0% for five replicate injections
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of 2,4-disulfamyl-5-trifluoromethylaniline in the portion of Bendroflumethiazide taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP 2,4-Disulfamyl-5-trifluoromethylaniline RS in the Standard solution (µg/mL) |
| CU | = | = concentration of Bendroflumethiazide in the Sample solution (µg/mL) |
Acceptance criteria:
NMT 1.5%
SPECIFIC TESTS
• Water Determination, Method I  921
921 :
NMT 0.5%
:
NMT 0.5%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers.
• USP Reference Standards  11
11
USP 2,4-Disulfamyl-5-trifluoromethylaniline RS
C7H8F3N3O4S2 319.29
C7H8F3N3O4S2 319.29
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Sujatha Ramakrishna, Ph.D.
Senior Scientific Liaison 1-301-816-8349 |
(SM22010) Monographs - Small Molecules 2 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2315