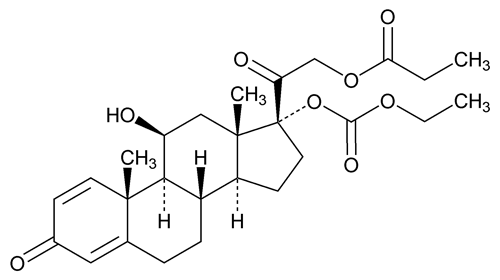
Prednicarbate
(pred'' ni kar' bate).
(1) Pregna-1,4-diene-3,20-dione, 17-[(ethoxycarbonyl)oxy]-11-hydroxy-21-(1-oxopropoxy)-, (11
(2) 11
» Prednicarbate contains not less than 97.0 percent and not more than 102.0 percent of C27H36O8, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed, light-resistant containers.
Identification—
B:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of the Standard preparation, as obtained in the Assay.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 6 hours: it loses not more than 0.5% of its weight.
for 6 hours: it loses not more than 0.5% of its weight.
Chromatographic purity—
Mobile phase, Resolution solution, and Chromatographic system—
Prepare as directed in the Assay.
Test solution—
Use the Assay preparation.
Diluted test solution—
Transfer 1 mL of the Test solution to a 200-mL volumetric flask, dissolve in and dilute with Mobile phase to volume, and mix.
Procedure—
Separately inject equal volumes (about 20 µL) of the Test solution and the Diluted test solution into the chromatograph, record the chromatograms, and measure the response for the prednicarbate peak obtained from the Diluted test solution. Obtain from the Test solution the peak responses for prednicarbate related compound A and for all peaks other than prednicarbate. Continue the chromatography for twice the retention time of prednicarbate. Calculate the percentage of the related compound and all the impurities in the portion of Prednicarbate taken by the formula:
0.5(rT / rDT)
in which rT is the peak response for each individual impurity peak obtained from the Test solution; and rDT is the peak response of the main peak in the chromatogram of the Diluted test solution: not more than 1.0% of prednicarbate related compound A is found; not more than 0.5% of any other individual impurity is found, with the exception of the main peak and the peak corresponding to prednicarbate related compound A; and not more than 2.0% of total impurities is found. Disregard any peak (0.0125%) with an area less than 0.025 times the area of the main peak in the chromatogram obtained from the Diluted test solution.
Assay—
Mobile phase—
Prepare a filtered and degassed mixture of water and acetonitrile (60:50). Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
Resolution solution—
Dissolve suitable quantities of USP Prednicarbate Related Compound A RS and Prednicarbate in Mobile phase to obtain a solution containing about 3 µg of each per mL. [note—Prepare all solutions just prior to use. ]
Standard preparation—
Dissolve an accurately weighed quantity of USP Prednicarbate RS in Mobile phase, and dilute quantitatively, and stepwise if necessary, with Mobile phase to obtain a solution having a known concentration of about 0.6 mg per mL.
Assay preparation—
Transfer about 30 mg of Prednicarbate, accurately weighed, to a 50-mL volumetric flask, dissolve in and dilute with Mobile phase to volume, and mix.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 243-nm detector and a 4-mm × 12.5-cm column that contains packing L1. The flow rate is about 0.7 mL per minute. Chromatograph the Resolution solution, and record the peak responses as directed for Procedure: the relative retention times are about 1.1 for prednicarbate related compound A and 1.0 for prednicarbate; and the resolution, R, between prednicarbate and prednicarbate related compound A is not less than 3.0.
)—
The liquid chromatograph is equipped with a 243-nm detector and a 4-mm × 12.5-cm column that contains packing L1. The flow rate is about 0.7 mL per minute. Chromatograph the Resolution solution, and record the peak responses as directed for Procedure: the relative retention times are about 1.1 for prednicarbate related compound A and 1.0 for prednicarbate; and the resolution, R, between prednicarbate and prednicarbate related compound A is not less than 3.0.
Procedure—
Separately inject equal volumes (about 20 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the prednicarbate peaks. Continue the chromatography for twice the retention time of prednicarbate. Calculate the quantity, in mg, of C27H3608 in the portion of Prednicarbate taken by the formula:
50C(rU / rS)
in which C is the concentration, in mg per mL, of USP Prednicarbate RS in the Standard preparation; and rU and rS are the peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Mary S. Waddell
Scientific Liaison 1-301-816-8124 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4395
Pharmacopeial Forum: Volume No. 31(5) Page 1398