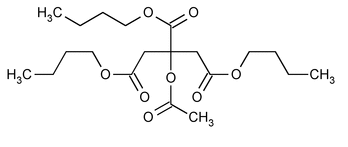
Acetyltributyl Citrate
(a seet'' il trye bue' til sit' rate).
DEFINITION
Acetyltributyl Citrate contains NLT 99.0% of C20H34O8, calculated on the anhydrous basis.
IDENTIFICATION
• B.
The retention time of the Sample solution corresponds to that of a similar preparation of USP Acetyltriethyl Citrate RS, as obtained in the Assay.
ASSAY
• Procedure
System suitability solution:
30 mg/mL each of USP Acetyltributyl Citrate RS and USP Tributyl Citrate RS in toluene
Sample solution:
30 mg/mL of Acetyltributyl Citrate in toluene
Chromatographic system
Mode:
GC
Detector:
Flame ionization
Column:
0.32-mm × 30-m, bonded with a 0.5-µm layer of phase G42
Temperature
Injector:
240
Detector:
280
Column:
See the temperature program table below.
| Initial Temperature ( |
Temperature Ramp ( |
Final Temperature ( |
Hold Time at Final Temperature (min) |
|---|---|---|---|
| 80 | — | 80 | 0 |
| 80 | 20 | 230 | 15 |
Flow rate:
1.9 mL/min
Carrier gas:
Helium
Injection type:
Split, 30:1
Injection size:
1 µL
System suitability
Sample:
System suitability solution
[Note—The relative retention times for tributyl citrate and acetyl tributyl citrate are 0.9 and 1.0, respectively. ]
Suitability requirements
Resolution:
NLT 1.5 between tributyl citrate and acetyl tributyl citrate
Relative standard deviation:
NMT 2.0% determined from both the tributyl citrate and acetyl tributyl citrate peaks, based on area percent calculation
Analysis
Sample:
Sample solution
[Note—Measure all of the peak areas, excluding the solvent peak. ]
Calculate the percentage of C20H34O8 in the portion of Acetyltributyl Citrate taken:
Result = (rU/rT) ×100
| rU | = | = peak area of the Sample solution |
| rT | = | = sum of all the peak areas |
Acceptance criteria:
NLT 99.0% on the anhydrous basis
IMPURITIES
Inorganic Impurities
• Heavy Metals, Method II  231
231 :
NMT 10 ppm
:
NMT 10 ppm
SPECIFIC TESTS
• Specific Gravity  841
841 :
1.045–1.055
:
1.045–1.055
• Refractive Index  831
831 :
1.4410–1.4425
:
1.4410–1.4425
• Acidity
Neutralized isopropyl alcohol:
To a suitable quantity of isopropyl alcohol add 2–3 drops of bromothymol blue TS and just sufficient 0.10 N sodium hydroxide dropwise to produce a faint blue color. [Note—Prepare Neutralized isopropyl alcohol just before use. ]
Sample solution:
32.0 g of Acetyltributyl Citrate in 30 mL of Neutralized isopropyl alcohol
Analysis:
Add bromothymol blue TS. Titrate with 0.10 N sodium hydroxide to a faint blue endpoint.
Acceptance criteria:
NMT 1.0 mL of 0.10 N sodium hydroxide is required.
• Water Determination, Method I  921
921 :
NMT 0.25%
:
NMT 0.25%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 1683
Pharmacopeial Forum: Volume No. 35(6) Page 1495